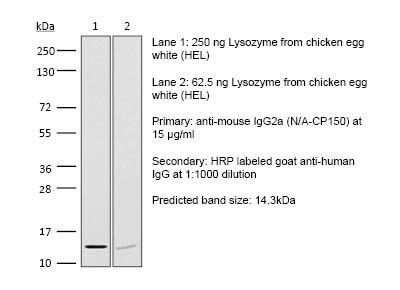
RecombiMAb mouse IgG2a (D265A) isotype control, anti-hen egg lysozyme
Product Description
Specifications
| Isotype | Mouse IgG2a |
|---|---|
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Mutations | D265A |
| Immunogen | Hen egg lysozyme (HEL) |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤0.5EU/mg (≤0.0005EU/μg) Determined by LAL assay |
| Aggregation |
<5% Determined by SEC |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from CHO cell supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_2927524 |
| Molecular Weight | 150 kDa |
| Murine Pathogen Tests |
Ectromelia/Mousepox Virus: Negative Hantavirus: Negative K Virus: Negative Lactate Dehydrogenase-Elevating Virus: Negative Lymphocytic Choriomeningitis virus: Negative Mouse Adenovirus: Negative Mouse Cytomegalovirus: Negative Mouse Hepatitis Virus: Negative Mouse Minute Virus: Negative Mouse Norovirus: Negative Mouse Parvovirus: Negative Mouse Rotavirus: Negative Mycoplasma Pulmonis: Negative Pneumonia Virus of Mice: Negative Polyoma Virus: Negative Reovirus Screen: Negative Sendai Virus: Negative Theiler’s Murine Encephalomyelitis: Negative |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Product Citations
-
Anti-CSF-1R therapy with combined immuno-chemotherapy coordinate an adaptive immune response to eliminate macrophage enriched triple negative breast cancers.
In Nat Commun on 3 January 2026 by Pedroza, D. A., Yuan, X., et al.
PubMed
Women diagnosed with metastatic triple negative breast cancer (mTNBC) have limited treatment options, are more prone to develop resistance and are associated with high mortality. A cold tumor immune microenvironment (TIME) characterized by low T cells and high tumor associated macrophages (TAMs) in mTNBC is associated with the failure of standard-of-care chemotherapy and immune checkpoint blockade (ICB) treatment. We demonstrate that the combination of immunomodulatory low-dose Cyclophosphamide (CTX) coupled with anti-CSF-1R antibody targeted therapy (SNDX-ms6352) and anti-PD-1 (ICB), was highly effective against aggressive metastatic Trp53 null TNBC transplantable syngeneic models that present with high macrophage infiltration. Mechanistically, CSF-1R inhibition along with CTX disrupted the M-CSF/CSF-1R axis which upregulated IL-17, IL-5 and type II interferon resulting in elevated B- and T cell infiltration. Addition of an anti-PD-1 maintenance dose helped overcome de novo PD-L1 intra-tumoral heterogeneity (ITH) associated recurrence in lung and liver mTNBC.
-
Immature monocytic cells within tumors differentiate into immunosuppressive cells in resistant tumors to immunotherapy.
In iScience on 15 August 2025 by Levin, S., Benguigui, M., et al.
PubMed
Immune checkpoint inhibitors (ICIs) have improved outcomes in advanced cancers, yet resistance remains a major obstacle. Here, we investigated the role of myeloid cells in shaping the immunosuppressive tumor microenvironment that contributes to ICI resistance. Using mutagenized ICI-sensitive and resistant 4T1 breast cancer clones, we performed single-cell RNA sequencing to characterize immune cell populations post-ICI therapy. We identified monocytic dendritic progenitors (MDPs) and common monocytic progenitors (cMOPs) enriched in sensitive tumors, which may differentiate into immunosuppressive cells in resistant tumors. Analysis of public datasets confirmed the presence of MDP-cMOPs in tumors and blood of patients with breast, lung, and colorectal cancer. We found high expression of CXCR4 and IL6R in MDP-cMOPs, and inhibiting these pathways blocked their recruitment and differentiation. Combined targeting of CXCR4 and IL6 pathway with ICI improved responses in resistant tumors, highlighting MDP-cMOPs as contributors to immunotherapy resistance and potential therapeutic targets.
-
CREB-binding protein/P300 bromodomain inhibition reduces neutrophil accumulation and activates antitumor immunity in triple-negative breast cancer.
In JCI Insight on 17 September 2024 by Yuan, X., Hao, X., et al.
PubMed
Tumor-associated neutrophils (TANs) have been shown to promote immunosuppression and tumor progression, and a high TAN frequency predicts poor prognosis in triple-negative breast cancer (TNBC). Dysregulation of CREB-binding protein (CBP)/P300 function has been observed with multiple cancer types. The bromodomain (BRD) of CBP/P300 has been shown to regulate its activity. In this study, we found that IACS-70654, a selective CBP/P300 BRD inhibitor, reduced TANs and inhibited the growth of neutrophil-enriched TNBC models. In the bone marrow, CBP/P300 BRD inhibition reduced the tumor-driven abnormal differentiation and proliferation of neutrophil progenitors. Inhibition of CBP/P300 BRD also stimulated the immune response by inducing an IFN response and MHCI expression in tumor cells and increasing tumor-infiltrated cytotoxic T cells. Moreover, IACS-70654 improved the response of a neutrophil-enriched TNBC model to docetaxel and immune checkpoint blockade. This provides a rationale for combining a CBP/P300 BRD inhibitor with standard-of-care therapies in future clinical trials for neutrophil-enriched TNBC.