InVivoMAb mouse IgG1 isotype control, unknown specificity
Product Description
Specifications
| Isotype | Mouse IgG1, κ |
|---|---|
| Recommended Dilution Buffer | InVivoPure pH 6.5 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Formulation |
PBS, pH 6.5 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107784 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Faraco, G., et al (2018). "Dietary salt promotes neurovascular and cognitive dysfunction through a gut-initiated TH17 response" Nat Neurosci 21(2): 240-249.
PubMed
A diet rich in salt is linked to an increased risk of cerebrovascular diseases and dementia, but it remains unclear how dietary salt harms the brain. We report that, in mice, excess dietary salt suppresses resting cerebral blood flow and endothelial function, leading to cognitive impairment. The effect depends on expansion of TH17 cells in the small intestine, resulting in a marked increase in plasma interleukin-17 (IL-17). Circulating IL-17, in turn, promotes endothelial dysfunction and cognitive impairment by the Rho kinase-dependent inhibitory phosphorylation of endothelial nitric oxide synthase and reduced nitric oxide production in cerebral endothelial cells. The findings reveal a new gut-brain axis linking dietary habits to cognitive impairment through a gut-initiated adaptive immune response compromising brain function via circulating IL-17. Thus, the TH17 cell-IL-17 pathway is a putative target to counter the deleterious brain effects induced by dietary salt and other diseases associated with TH17 polarization.
-
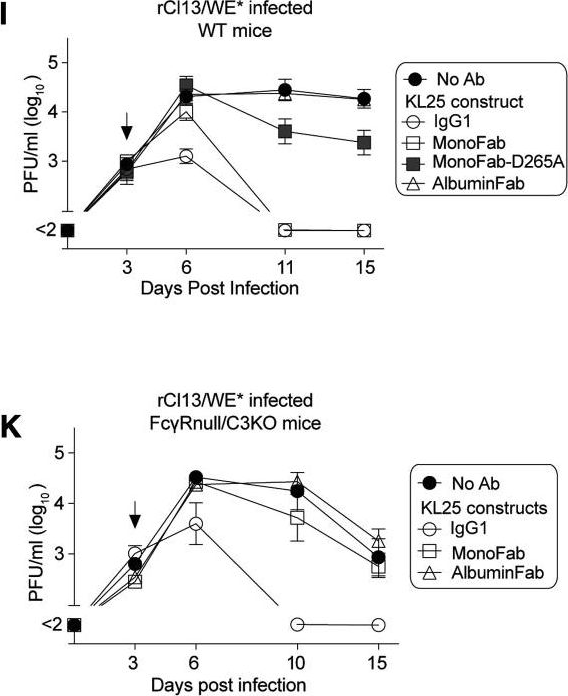
Macal, M., et al (2018). "Self-Renewal and Toll-like Receptor Signaling Sustain Exhausted Plasmacytoid Dendritic Cells during Chronic Viral Infection" Immunity 48(4): 730-744 e735.
PubMed
Although characterization of T cell exhaustion has unlocked powerful immunotherapies, the mechanisms sustaining adaptations of short-lived innate cells to chronic inflammatory settings remain unknown. During murine chronic viral infection, we found that concerted events in bone marrow and spleen mediated by type I interferon (IFN-I) and Toll-like receptor 7 (TLR7) maintained a pool of functionally exhausted plasmacytoid dendritic cells (pDCs). In the bone marrow, IFN-I compromised the number and the developmental capacity of pDC progenitors, which generated dysfunctional pDCs. Concurrently, exhausted pDCs in the periphery were maintained by self-renewal via IFN-I- and TLR7-induced proliferation of CD4(-) subsets. On the other hand, pDC functional loss was mediated by TLR7, leading to compromised IFN-I production and resistance to secondary infection. These findings unveil the mechanisms sustaining a self-perpetuating pool of functionally exhausted pDCs and provide a framework for deciphering long-term exhaustion of other short-lived innate cells during chronic inflammation.
-
Sell, S., et al (2015). "Control of murine cytomegalovirus infection by gammadelta T cells" PLoS Pathog 11(2): e1004481.
PubMed
Infections with cytomegalovirus (CMV) can cause severe disease in immunosuppressed patients and infected newborns. Innate as well as cellular and humoral adaptive immune effector functions contribute to the control of CMV in immunocompetent individuals. None of the innate or adaptive immune functions are essential for virus control, however. Expansion of gammadelta T cells has been observed during human CMV (HCMV) infection in the fetus and in transplant patients with HCMV reactivation but the protective function of gammadelta T cells under these conditions remains unclear. Here we show for murine CMV (MCMV) infections that mice that lack CD8 and CD4 alphabeta-T cells as well as B lymphocytes can control a MCMV infection that is lethal in RAG-1(-/-) mice lacking any T- and B-cells. gammadelta T cells, isolated from infected mice can kill MCMV infected target cells in vitro and, importantly, provide long-term protection in infected RAG-1(-/-) mice after adoptive transfer. gammadelta T cells in MCMV infected hosts undergo a prominent and long-lasting phenotypic change most compatible with the view that the majority of the gammadelta T cell population persists in an effector/memory state even after resolution of the acute phase of the infection. A clonotypically focused Vgamma1 and Vgamma2 repertoire was observed at later stages of the infection in the organs where MCMV persists. These findings add gammadelta T cells as yet another protective component to the anti-CMV immune response. Our data provide clear evidence that gammadelta T cells can provide an effective control mechanism of acute CMV infections, particularly when conventional adaptive immune mechanisms are insufficient or absent, like in transplant patient or in the developing immune system in utero. The findings have implications in the stem cell transplant setting, as antigen recognition by gammadelta T cells is not MHC-restricted and dual reactivity against CMV and tumors has been described.
-
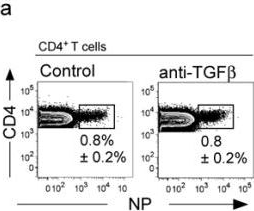
Manlove, L. S., et al (2015). "Adaptive Immunity to Leukemia Is Inhibited by Cross-Reactive Induced Regulatory T Cells" J Immunol .
PubMed
BCR-ABL+ acute lymphoblastic leukemia patients have transient responses to current therapies. However, the fusion of BCR to ABL generates a potential leukemia-specific Ag that could be a target for immunotherapy. We demonstrate that the immune system can limit BCR-ABL+ leukemia progression although ultimately this immune response fails. To address how BCR-ABL+ leukemia escapes immune surveillance, we developed a peptide: MHC class II tetramer that labels endogenous BCR-ABL-specific CD4+ T cells. Naive mice harbored a small population of BCR-ABL-specific T cells that proliferated modestly upon immunization. The small number of naive BCR-ABL-specific T cells was due to negative selection in the thymus, which depleted BCR-ABL-specific T cells. Consistent with this observation, we saw that BCR-ABL-specific T cells were cross-reactive with an endogenous peptide derived from ABL. Despite this cross-reactivity, the remaining population of BCR-ABL reactive T cells proliferated upon immunization with the BCR-ABL fusion peptide and adjuvant. In response to BCR-ABL+ leukemia, BCR-ABL-specific T cells proliferated and converted into regulatory T (Treg) cells, a process that was dependent on cross-reactivity with self-antigen, TGF-beta1, and MHC class II Ag presentation by leukemic cells. Treg cells were critical for leukemia progression in C57BL/6 mice, as transient Treg cell ablation led to extended survival of leukemic mice. Thus, BCR-ABL+ leukemia actively suppresses antileukemia immune responses by converting cross-reactive leukemia-specific T cells into Treg cells.
Product Citations
-
Nasal CD4+ tissue-resident memory T cells provide cross-protective immunity to influenza.
In J Exp Med on 4 May 2026 by Mathew, N., Gailleton, R., et al.
PubMed
CD4 tissue-resident memory T cells (TRM) are crucial adaptive immune components involved in preventing influenza A virus (IAV) infection. Despite their importance, their physiological role in the upper respiratory tract, the first site of contact with IAV, remains unclear. Here, we find that, after IAV infection, antigen-specific CD4 TRM persist in the nasal tissue (NT) compartment after infection and provide protection upon heterosubtypic challenge. Single-cell RNA-sequencing analysis reveals that NT CD4 TRM are heterogeneous and transcriptionally distinct as compared with their lung counterparts. Mechanistically, we demonstrate that the CXCR6-CXCL16 axis promotes CD4 TRM residency in the NT. Furthermore, we show that the NT of mice and humans contains a high frequency of Th17 CD4 TRM that aid in local viral clearance and in reducing tissue damage. Collectively, our results support a robust physiological role for NT CD4 TRM in local protection during heterosubtypic IAV infection.
-
Splenic erythrophagocytosis is regulated by ALX/FPR2 signaling.
In Haematologica on 1 May 2026 by Asplund, H., Dreyer, H. H., et al.
PubMed
Maintaining a healthy pool of circulating red blood cells (RBC) is essential for adequate perfusion, as even minor changes in the population can impair oxygen delivery, resulting in serious health complications including tissue ischemia and organ dysfunction. This responsibility largely falls to specialized macrophages in the spleen, known as red pulp macrophages, which efficiently take up and recycle damaged RBC. However, questions remain regarding how these macrophages are acutely activated to accommodate increased demand. Proresolving lipid mediators stimulate macrophage phagocytosis and efferocytosis but their role in erythrophagocytosis has only recently been described. To investigate the role of lipid mediators in red pulp macrophage function, we targeted the ALX/FPR2 signaling pathway, as this receptor binds multiple lipid mediator ligands eliciting potent macrophage responses. We found that mice with Fpr2 deletion exhibited disrupted erythrocyte homeostasis resulting in an aged RBC pool, decreased markers of splenic RBC turnover, and altered splenic macrophage phenotype characterized by changes in heme metabolism. Upon activation of on-demand erythrophagocytosis, production of the ALX/FPR2 ligand, lipoxin A4 (LXA4), was induced in the spleen while receptor-deficient animals were unable to efficiently clear damaged RBC, a defect that was conserved in mice with myeloid-specific FPR2 deletion. Similarly, mice lacking the LXA4 biosynthetic enzyme displayed defective erythrophagocytosis that was rescued with LXA4 administration. These results indicate that the ALX/FPR2 signaling axis is necessary for maintenance of RBC homeostasis and that LXA4 activation is a critical aspect of the red pulp macrophage response to acute erythroid stress.
-
Senescence-like cells recruit γδ T cells to drive prolonged hyposmia after SARS-CoV-2 infection in mice.
In EMBO Rep on 10 April 2026 by Tsuji, S., Nakano, S., et al.
PubMed
Persistent hyposmia is a hallmark of post COVID-19 conditions, yet the mechanisms sustaining olfactory dysfunction after viral clearance remain poorly understood. Here, using mouse models of SARS-CoV-2 infection, we show that virus-induced senescence-like changes in uninfected olfactory mucosal fibroblasts persist long after viral clearance and drive prolonged olfactory dysfunction. These senescence-like cells secrete SASP factors, including IFNγ, CXCL9, and CXCL11, thereby recruiting γδ T cells to the olfactory mucosa. The accumulated γδ T cells produce excessive IL-17A, which acts on IL-17 receptor A expressed on olfactory sensory neurons, leading to sustained impairment of their function. Genetic ablation of senescence pathways (p16/p21 double knockout), pharmacological elimination of senescent cells with the senolytic drug ABT263, or olfactory neuron-specific deletion of IL-17 receptor A each significantly alleviate prolonged olfactory dysfunction. These findings identify a senescence-γδ T cell-IL-17A axis as a key driver of prolonged hyposmia following SARS-CoV-2 infection in mice.
-
Peripheral immune-inducer dendritic cells drive early-life allergic inflammation.
In Nature on 1 April 2026 by Xing, Y., Reznikov, I., et al.
PubMed
Atopic diseases associated with allergens, as well as allergic diseases, frequently arise early in life; however, the age-dependent mechanisms governing immune responses to allergens remain poorly understood1. Here we find that in early life, exposure to common allergens triggers a distinct bifurcated immune response, simultaneously triggering type 17 inflammation in the skin and initiating canonical T helper 2 sensitization in the lymph nodes. This early-life γδ type 17-mediated dermatitis primes the exaggerated allergic lung inflammation upon secondary allergen exposure. Mechanistically, we find dendritic cell (DC)-mediated type 17 activation directly in the skin without requiring migration to lymph nodes; we term this state 'peripheral immune inducer' (pii) DC. CD301b+ conventional type 2 DCs acquire allergen, adopt the pii-DC state, produce IL-23 and activate local γδ type 17 cells independently of lymph-node engagement. The pii-DC state is enabled by the immature hypothalamic-pituitary-adrenal axis and physiologically low systemic glucocorticoids characteristic of early life2,3; DC-specific deletion of the glucocorticoid receptor recapitulates the pii-DC phenotype. These findings define a developmental checkpoint, set by neuroendocrine maturation, that enables in situ DC activation and immune induction, thereby shaping age-dependent responses to allergens.