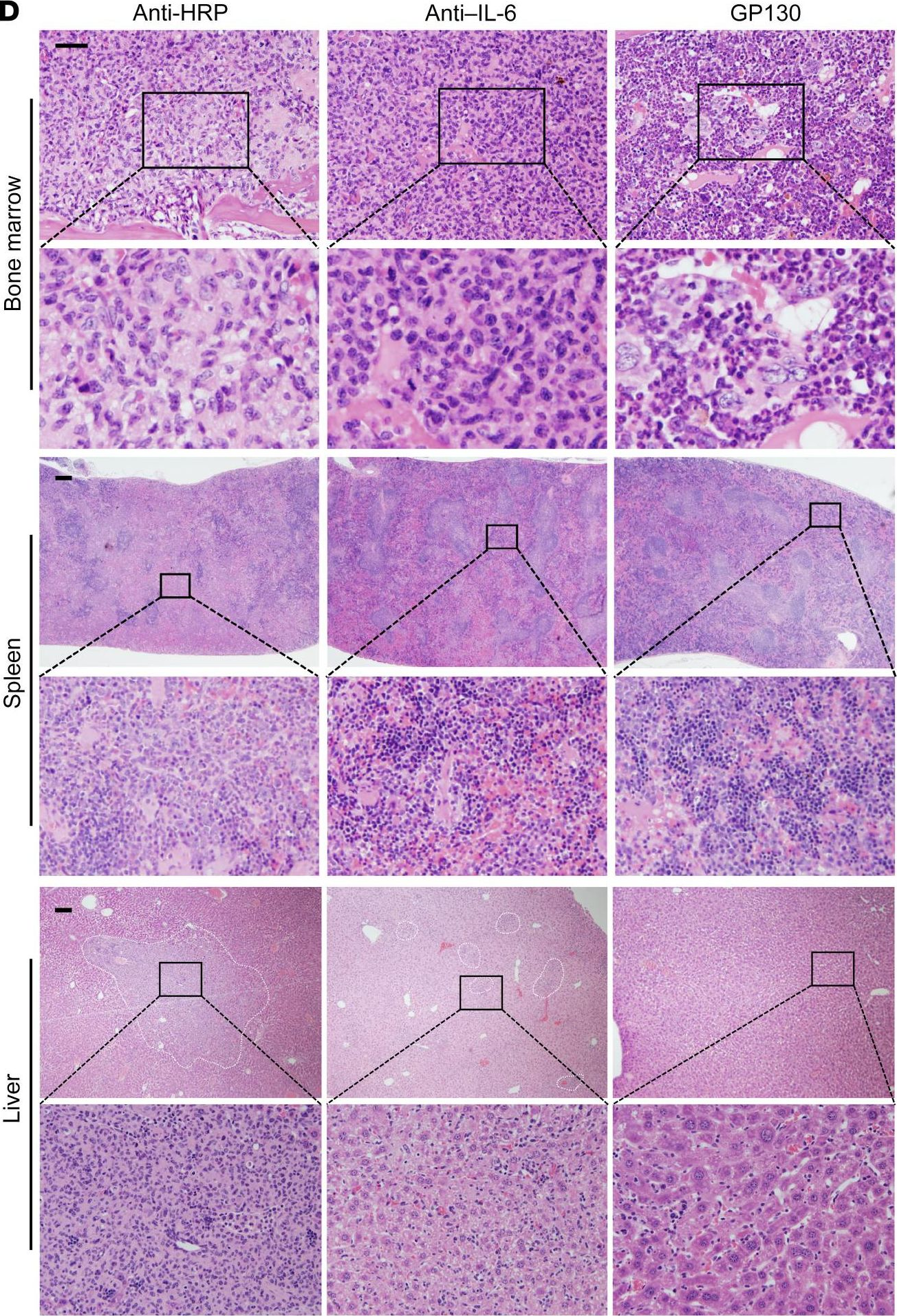
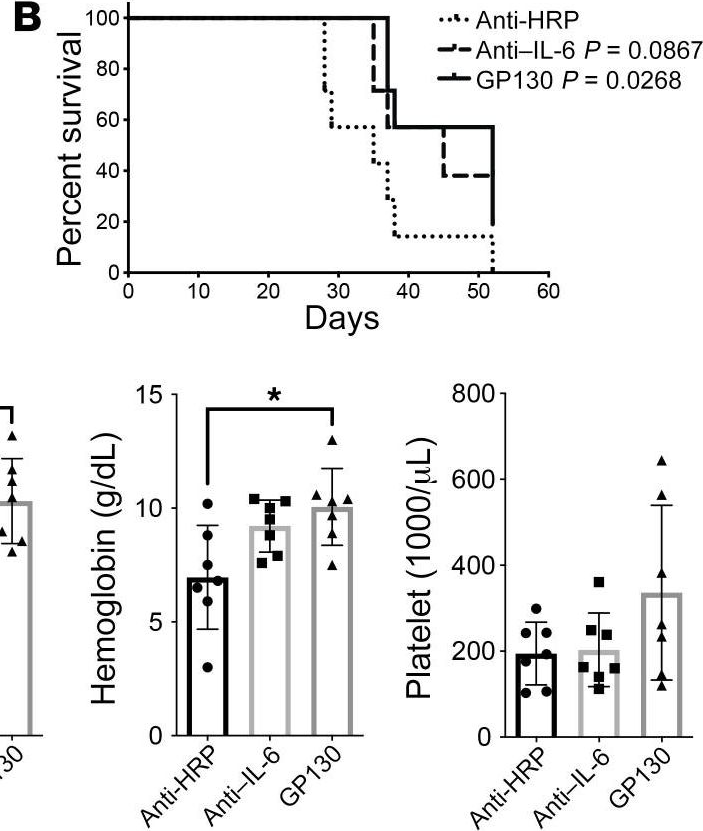
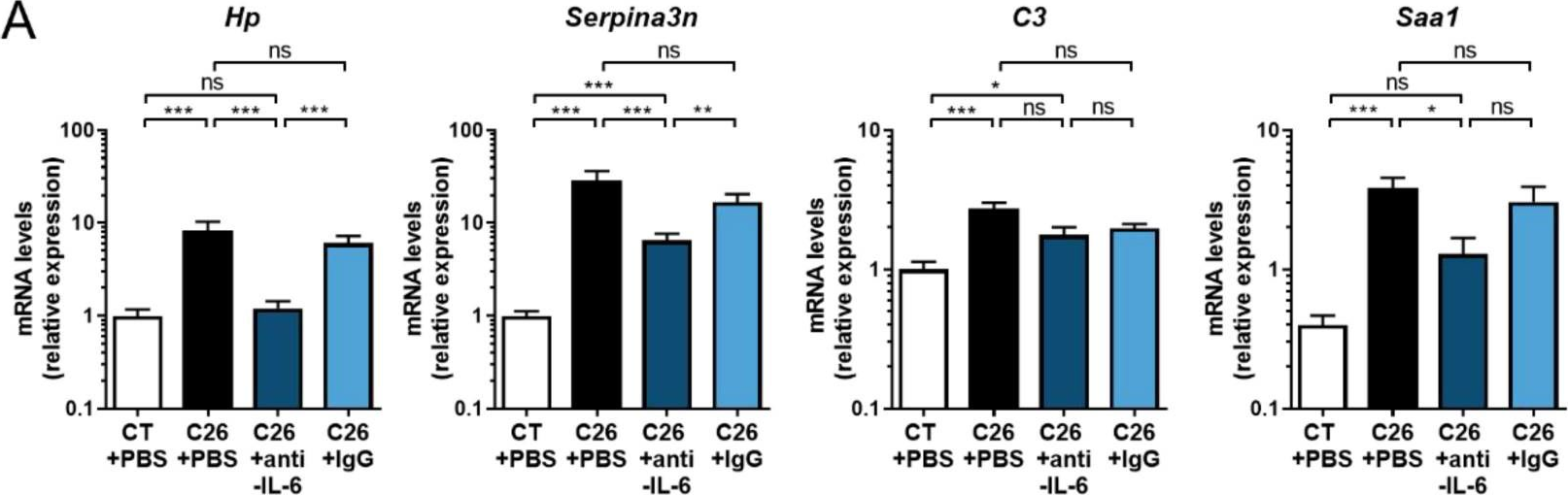
InVivoMAb rat IgG1 isotype control, anti-horseradish peroxidase
Product Description
Specifications
| Isotype | Rat IgG1, κ |
|---|---|
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107775 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Goschl, L., et al (2018). "A T cell-specific deletion of HDAC1 protects against experimental autoimmune encephalomyelitis" J Autoimmun 86: 51-61.
PubMed
Multiple sclerosis (MS) is a human neurodegenerative disease characterized by the invasion of autoreactive T cells from the periphery into the CNS. Application of pan-histone deacetylase inhibitors (HDACi) ameliorates experimental autoimmune encephalomyelitis (EAE), an animal model for MS, suggesting that HDACi might be a potential therapeutic strategy for MS. However, the function of individual HDAC members in the pathogenesis of EAE is not known. In this study we report that mice with a T cell-specific deletion of HDAC1 (using the Cd4-Cre deleter strain; HDAC1-cKO) were completely resistant to EAE despite the ability of HDAC1cKO CD4(+) T cells to differentiate into Th17 cells. RNA sequencing revealed STAT1 as a prominent upstream regulator of differentially expressed genes in activated HDAC1-cKO CD4(+) T cells and this was accompanied by a strong increase in phosphorylated STAT1 (pSTAT1). This suggests that HDAC1 controls STAT1 activity in activated CD4(+) T cells. Increased pSTAT1 levels correlated with a reduced expression of the chemokine receptors Ccr4 and Ccr6, which are important for the migration of T cells into the CNS. Finally, EAE susceptibility was restored in WT:HDAC1-cKO mixed BM chimeric mice, indicating a cell-autonomous defect. Our data demonstrate a novel pathophysiological role for HDAC1 in EAE and provide evidence that selective inhibition of HDAC1 might be a promising strategy for the treatment of MS.
-
Clemente-Casares, X., et al (2016). "Expanding antigen-specific regulatory networks to treat autoimmunity" Nature 530(7591): 434-440.
PubMed
Regulatory T cells hold promise as targets for therapeutic intervention in autoimmunity, but approaches capable of expanding antigen-specific regulatory T cells in vivo are currently not available. Here we show that systemic delivery of nanoparticles coated with autoimmune-disease-relevant peptides bound to major histocompatibility complex class II (pMHCII) molecules triggers the generation and expansion of antigen-specific regulatory CD4(+) T cell type 1 (TR1)-like cells in different mouse models, including mice humanized with lymphocytes from patients, leading to resolution of established autoimmune phenomena. Ten pMHCII-based nanomedicines show similar biological effects, regardless of genetic background, prevalence of the cognate T-cell population or MHC restriction. These nanomedicines promote the differentiation of disease-primed autoreactive T cells into TR1-like cells, which in turn suppress autoantigen-loaded antigen-presenting cells and drive the differentiation of cognate B cells into disease-suppressing regulatory B cells, without compromising systemic immunity. pMHCII-based nanomedicines thus represent a new class of drugs, potentially useful for treating a broad spectrum of autoimmune conditions in a disease-specific manner.
-
Sell, S., et al (2015). "Control of murine cytomegalovirus infection by gammadelta T cells" PLoS Pathog 11(2): e1004481.
PubMed
Infections with cytomegalovirus (CMV) can cause severe disease in immunosuppressed patients and infected newborns. Innate as well as cellular and humoral adaptive immune effector functions contribute to the control of CMV in immunocompetent individuals. None of the innate or adaptive immune functions are essential for virus control, however. Expansion of gammadelta T cells has been observed during human CMV (HCMV) infection in the fetus and in transplant patients with HCMV reactivation but the protective function of gammadelta T cells under these conditions remains unclear. Here we show for murine CMV (MCMV) infections that mice that lack CD8 and CD4 alphabeta-T cells as well as B lymphocytes can control a MCMV infection that is lethal in RAG-1(-/-) mice lacking any T- and B-cells. gammadelta T cells, isolated from infected mice can kill MCMV infected target cells in vitro and, importantly, provide long-term protection in infected RAG-1(-/-) mice after adoptive transfer. gammadelta T cells in MCMV infected hosts undergo a prominent and long-lasting phenotypic change most compatible with the view that the majority of the gammadelta T cell population persists in an effector/memory state even after resolution of the acute phase of the infection. A clonotypically focused Vgamma1 and Vgamma2 repertoire was observed at later stages of the infection in the organs where MCMV persists. These findings add gammadelta T cells as yet another protective component to the anti-CMV immune response. Our data provide clear evidence that gammadelta T cells can provide an effective control mechanism of acute CMV infections, particularly when conventional adaptive immune mechanisms are insufficient or absent, like in transplant patient or in the developing immune system in utero. The findings have implications in the stem cell transplant setting, as antigen recognition by gammadelta T cells is not MHC-restricted and dual reactivity against CMV and tumors has been described.
-
Grinberg-Bleyer, Y., et al (2015). "Cutting edge: NF-kappaB p65 and c-Rel control epidermal development and immune homeostasis in the skin" J Immunol 194(6): 2472-2476.
PubMed
Psoriasis is an inflammatory skin disease in which activated immune cells and the proinflammatory cytokine TNF are well-known mediators of pathogenesis. The transcription factor NF-kappaB is a key regulator of TNF production and TNF-induced proinflammatory gene expression, and both the psoriatic transcriptome and genetic susceptibility further implicate NF-kappaB in psoriasis etiopathology. However, the role of NF-kappaB in psoriasis remains controversial. We analyzed the function of canonical NF-kappaB in the epidermis using CRE-mediated deletion of p65 and c-Rel in keratinocytes. In contrast to animals lacking p65 or c-Rel alone, mice lacking both subunits developed severe dermatitis after birth. Consistent with its partial histological similarity to human psoriasis, this condition could be prevented by anti-TNF treatment. Moreover, regulatory T cells in lesional skin played an important role in disease remission. Our results demonstrate that canonical NF-kappaB in keratinocytes is essential for the maintenance of skin immune homeostasis and is protective against spontaneous dermatitis.
Product Citations
-
Colonic Engyodontium fungus triggers neutrophil antimicrobial activity to suppress Lactobacillus johnsonii-derived glutamic acid-maintained Tregs.
In J Clin Invest on 15 April 2026 by Wang, X., Sun, H., et al.
PubMed
Isolating commensal fungi from mouse intestines has been challenging, limiting our understanding of their role in intestinal immune homeostasis and diseases. Using an Fc fusion protein of the C-type lectin receptor Dectin-2, we successfully purified the commensal Ascomycota fungus Engyodontium sp. from mouse feces. Engyodontium enhances the antimicrobial activity of colonic neutrophils via the CARD9 pathway and exacerbates colitis by impairing the colonization of intestinal Lactobacillus johnsonii WXY strain. L. johnsonii produces high levels of l-glutamic acid by expressing the glutaminase-encoding gene glsA to facilitate Treg expansion via enhancing IL-2 receptor signaling. Patients with Crohn disease (CD) and ulcerative colitis harbored increased Engyodontium and decreased L. johnsonii abundance. Engyodontium directly induced calprotectin in human colonic neutrophils, and patients with CD had lower levels of l-glutamic acid, which also promoted human Treg expansion. These findings highlight the Engyodontium-calprotectin axis against the Lactobacillus-glutamate-Treg cascade to aggravate colitis, suggesting commensal Engyodontium-triggered signaling as a therapeutic target for mucosal inflammatory diseases.
-
Comprehensive immune profiling reveals IFN-γ signaling in T cells mediates parasite phagocytosis in a rodent malaria model.
In MBio on 8 April 2026 by Chen, S. -. s., Yang, Q., et al.
PubMed
Pulmonary manifestation in malaria can be life-threatening. Using a rodent malaria model and comprehensive transcriptomics analyses to illustrate the immune landscape of malaria-associated lung pathology, we identified that IFN-γ signaling in T cells plays an important role in malaria-associated lung pathology. Disruption of IFN-γ signaling in T cells leads to reduced parasite load in the lungs and attenuated lung pathology by enhancing T cell-monocyte interaction. Surprisingly, the stronger interaction leads to an increase in a proinflammatory monocyte subset characterized by CD8 and Ly6C expression, which exhibits prominently elevated phagocytic capacity compared to the CD8- counterpart. Our study illustrates the importance of IFN-γ signaling in T cell-monocyte interaction in malaria-associated lung pathology, highlighting the complex and intricate immune network induced by the Plasmodium infection.IMPORTANCEMalaria-associated lung pathology is a common complication of malaria in adults and often occurs during or even after antimalarial treatment, and current evidence suggests that it is associated with cytokine imbalance and dysregulation of immune responses in the lungs. In this study, we conducted detailed flow cytometry analyses, time-series bulk transcriptomics, and spatial transcriptomics to profile the immune landscape of malaria-associated lung pathology in a mouse malaria model and revealed that IFN-γ signaling in T cells plays a key role in the lung pathology. In addition, we identified a subgroup of CD8-expressing proinflammatory monocytes that exhibit heightened parasite phagocytotic capability.
-
Single-Cell Analysis of Chemotherapy-induced Remodeling Reveals CD276-driven Basal-like Chemoresistance in Pancreatic Cancer.
In Gastroenterology on 1 April 2026 by Zhang, Y., Du, Y., et al.
PubMed
Unresectable advanced pancreatic ductal adenocarcinoma (PDAC) typically requires systematic chemotherapy, but it remains unclear how this treatment remodels tumor cell plasticity and the tumor microenvironment (TME) to influence clinical outcomes.
-
Metabolically reprogrammed eosinophils impair T cell immunity and cause chronic skin infection.
In EMBO Mol Med on 1 April 2026 by Barinberg, D., Sebald, H., et al.
PubMed
Eosinophils exhibit antimicrobial, cytotoxic and immunoregulatory effects, but our knowledge of their transcriptional and functional heterogeneity is still limited, especially in non-intestinal tissues. Here, we used a mouse model of chronic cutaneous inflammation elicited by the protozoan pathogen Leishmania mexicana to investigate the function and transcriptional dynamics of skin eosinophils. Infection of C57BL/6 mice triggered local and systemic eosinophilia that was driven by type 2 innate lymphoid cells and interleukin-5. Genetic and pharmacological eosinophil depletion led to an enhanced Th1 response, polarization towards M1-like macrophages and resolution of clinical disease, despite an unexpected simultaneous upregulation of IL-4. Single-cell transcriptomics revealed a skin-imprinted trajectory of inflammatory eosinophils that strongly expressed the glucose transporter Slc2a3 (GLUT3) These eosinophils impeded the function of Th1 cells by forming a competitive metabolic niche through preferential glucose uptake. Our findings uncover an inflammatory, metabolically reprogrammed eosinophil population that promotes chronic skin inflammation by limiting protective T cell responses.