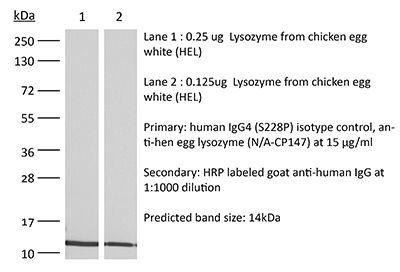
RecombiMAb human IgG4 (S228P) isotype control, anti-hen egg lysozyme
Product Description
Note: This product was previously sold as catalog number BE0349 and is identical to the product previously sold as BE0349.
Specifications
| Isotype | Human IgG4, κ |
|---|---|
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Mutations | S228P |
| Immunogen | Hen egg lysozyme (HEL) |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤0.5EU/mg (≤0.0005EU/μg) Determined by LAL assay |
| Aggregation |
<5% Determined by SEC |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from CHO cell supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_2894768 |
| Molecular Weight | 150 kDa |
| Murine Pathogen Tests |
Ectromelia/Mousepox Virus: Negative Hantavirus: Negative K Virus: Negative Lactate Dehydrogenase-Elevating Virus: Negative Lymphocytic Choriomeningitis virus: Negative Mouse Adenovirus: Negative Mouse Cytomegalovirus: Negative Mouse Hepatitis Virus: Negative Mouse Minute Virus: Negative Mouse Norovirus: Negative Mouse Parvovirus: Negative Mouse Rotavirus: Negative Mycoplasma Pulmonis: Negative Pneumonia Virus of Mice: Negative Polyoma Virus: Negative Reovirus Screen: Negative Sendai Virus: Negative Theiler’s Murine Encephalomyelitis: Negative |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Product Citations
-
Inflammatory arthritis irAE may represent a unique autoimmune disease primarily driven by T cells but likely not autoantibodies.
In Sci Adv on 3 April 2026 by Zhu, X., Yu, Y., et al.
PubMed
The underlying immunopathogenesis of inflammatory arthritis (IA) immune-related adverse event (irAE) remains obscure. Unlike rheumatoid arthritis (RA), where autoantibodies and B cell dysfunction are central features, the contribution of humoral immunity to IA-irAE is unclear. Here, we performed immunophenotyping of peripheral blood from patients with IA-irAE and compared them with patients with seronegative RA, immune checkpoint inhibition-treated patients without irAE, and healthy controls. IA-irAE was marked with increased cytotoxic gene expression and metabolic activation in T cells and reduced CXCR3 and CCR6 expression in CD4+ T cells. Contrary to seronegative RA, patients with IA-irAE displayed no substantial elevation in autoantibody levels or atypical CD11c+CD21- B cells. IA-irAE was further characterized by elevated levels of interleukin-6 (IL-6), IL-12, and type I interferon, which correlated with the T cell activation phenotypes. Together, our findings define IA-irAE as a disease with certain immunological features distinctive from RA, representing a potentially T cell-driven, autoantibody-independent autoimmunity. These results offer insights into immune tolerance breakdown and therapeutic targeting in irAEs.
-
Ibrutinib and PD-1 Blockade Potentiate Mesothelin-Targeting CAR T-cell Therapy in Preclinical Models of Pancreatic Cancer.
In Clin Cancer Res on 4 February 2026 by Armstrong, A., van der Plancke, G., et al.
PubMed
Pancreatic ductal adenocarcinoma (PDAC) remains refractory to chimeric antigen receptor (CAR) T-cell therapies because of its immunosuppressive microenvironment and a dense extracellular matrix deposited by cancer-associated fibroblasts (CAF), which impair CAR T-cell infiltration. To address these barriers, we previously developed a dual-targeting CAR-TEAM platform in which mesothelin-specific CAR T cells secrete a fibroblast activation protein (FAP)-targeting T-cell engager antibody molecule (TEAM) to simultaneously kill tumor cells and CAF. In this study, we leveraged mesothelin-targeting CAR T cells and tested rational drug combinations and optimal delivery strategies to enhance therapeutic efficacy and guide potential combinations that could be incorporated into a clinical study.
-
Novel humanized anti-Nav1.7 antibodies with long-lasting, side-effect-free analgesic effects
In bioRxiv on 27 August 2025 by Yoneda, S., Uta, D., et al.
-
The lncRNA EPIC1 suppresses dsRNA-induced type I IFN signaling and is a therapeutic target to enhance TNBC response to PD-1 inhibition.
In Sci Signal on 12 August 2025 by Pattarayan, D., Wang, Y., et al.
PubMed
Increases in retroelement-derived double-stranded RNAs (dsRNAs) in various types of cancer cells facilitate the activation of antitumor immune responses. The long noncoding RNA EPIC1 interacts with the histone methyltransferase EZH2 and contributes to tumor immune evasion. Here, we found that EPIC1 in tumor cells suppressed cytoplasmic dsRNA accumulation, type I interferon (IFN) responses, and antitumor immunity. In various cancer cell lines, knockdown of EPIC1 stimulated the production of dsRNA from retroelements and an antiviral-like type I IFN response that activated immune cells. EPIC1 inhibited the expression of LINE, SINE, and LTR retroelements that were also repressed by EZH2, suggesting a potential role for the EPIC1-EZH2 interaction in regulating dsRNA production. In a humanized mouse model, in vivo delivery of EPIC1-targeting oligonucleotides enhanced dsRNA accumulation in breast cancer xenografts, reduced tumor growth, and increased the infiltration of T cells and inflammatory macrophages into tumors. Furthermore, EPIC1 knockdown improved the therapeutic efficacy of the immunotherapy drug pembrolizumab, a PD-1 inhibitor, in the humanized mouse model. Together, our findings establish EPIC1 as a key regulator of dsRNA-mediated type I IFN responses and highlight its potential as a therapeutic target to improve the efficacy of immunotherapy.