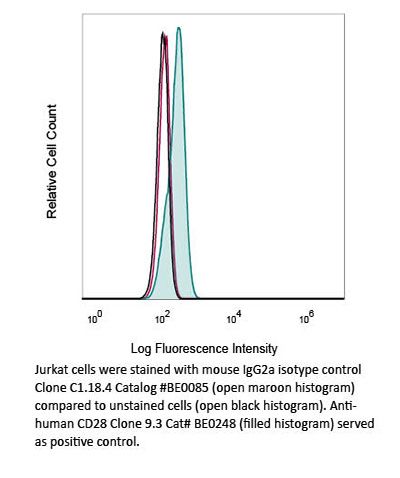
InVivoMAb mouse IgG2a isotype control, unknown specificity
Product Description
Specifications
| Isotype | Mouse IgG2a, κ |
|---|---|
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107771 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Bulliard, Y., et al (2013). "Activating Fc gamma receptors contribute to the antitumor activities of immunoregulatory receptor-targeting antibodies" J Exp Med 210(9): 1685-1693.
PubMed
Fc gamma receptor (FcgammaR) coengagement can facilitate antibody-mediated receptor activation in target cells. In particular, agonistic antibodies that target tumor necrosis factor receptor (TNFR) family members have shown dependence on expression of the inhibitory FcgammaR, FcgammaRIIB. It remains unclear if engagement of FcgammaRIIB also extends to the activities of antibodies targeting immunoregulatory TNFRs expressed by T cells. We have explored the requirement for activating and inhibitory FcgammaRs for the antitumor effects of antibodies targeting the TNFR glucocorticoid-induced TNFR-related protein (GITR; TNFRSF18; CD357) expressed on activated and regulatory T cells (T reg cells). We found that although FcgammaRIIB was dispensable for the in vivo efficacy of anti-GITR antibodies, in contrast, activating FcgammaRs were essential. Surprisingly, the dependence on activating FcgammaRs extended to an antibody targeting the non-TNFR receptor CTLA-4 (CD152) that acts as a negative regulator of T cell immunity. We define a common mechanism that correlated with tumor efficacy, whereby antibodies that coengaged activating FcgammaRs expressed by tumor-associated leukocytes facilitated the selective elimination of intratumoral T cell populations, particularly T reg cells. These findings may have broad implications for antibody engineering efforts aimed at enhancing the therapeutic activity of immunomodulatory antibodies.
-
Carmi, Y., et al (2015). "Allogeneic IgG combined with dendritic cell stimuli induce antitumour T-cell immunity" Nature 521(7550): 99-104.
PubMed
Whereas cancers grow within host tissues and evade host immunity through immune-editing and immunosuppression, tumours are rarely transmissible between individuals. Much like transplanted allogeneic organs, allogeneic tumours are reliably rejected by host T cells, even when the tumour and host share the same major histocompatibility complex alleles, the most potent determinants of transplant rejection. How such tumour-eradicating immunity is initiated remains unknown, although elucidating this process could provide the basis for inducing similar responses against naturally arising tumours. Here we find that allogeneic tumour rejection is initiated in mice by naturally occurring tumour-binding IgG antibodies, which enable dendritic cells (DCs) to internalize tumour antigens and subsequently activate tumour-reactive T cells. We exploited this mechanism to treat autologous and autochthonous tumours successfully. Either systemic administration of DCs loaded with allogeneic-IgG-coated tumour cells or intratumoral injection of allogeneic IgG in combination with DC stimuli induced potent T-cell-mediated antitumour immune responses, resulting in tumour eradication in mouse models of melanoma, pancreas, lung and breast cancer. Moreover, this strategy led to eradication of distant tumours and metastases, as well as the injected primary tumours. To assess the clinical relevance of these findings, we studied antibodies and cells from patients with lung cancer. T cells from these patients responded vigorously to autologous tumour antigens after culture with allogeneic-IgG-loaded DCs, recapitulating our findings in mice. These results reveal that tumour-binding allogeneic IgG can induce powerful antitumour immunity that can be exploited for cancer immunotherapy.
-
Lamere, M. W., et al (2011). "Regulation of antinucleoprotein IgG by systemic vaccination and its effect on influenza virus clearance" J Virol 85(10): 5027-5035.
PubMed
Seasonal influenza epidemics recur due to antigenic drift of envelope glycoprotein antigens and immune evasion of circulating viruses. Additionally, antigenic shift can lead to influenza pandemics. Thus, a universal vaccine that protects against multiple influenza virus strains could alleviate the continuing impact of this virus on human health. In mice, accelerated clearance of a new viral strain (cross-protection) can be elicited by prior infection (heterosubtypic immunity) or by immunization with the highly conserved internal nucleoprotein (NP). Both heterosubtypic immunity and NP-immune protection require antibody production. Here, we show that systemic immunization with NP readily accelerated clearance of a 2009 pandemic H1N1 influenza virus isolate in an antibody-dependent manner. However, human immunization with trivalent inactivated influenza virus vaccine (TIV) only rarely and modestly boosted existing levels of anti-NP IgG. Similar results were observed in mice, although the reaction could be enhanced with adjuvants, by adjusting the stoichiometry among NP and other vaccine components, and by increasing the interval between TIV prime and boost. Importantly, mouse heterosubtypic immunity that had waned over several months could be enhanced by injecting purified anti-NP IgG or by boosting with NP protein, correlating with a long-lived increase in anti-NP antibody titers. Thus, current immunization strategies poorly induce NP-immune antibody that is nonetheless capable of contributing to long-lived cross-protection. The high conservation of NP antigen and the known longevity of antibody responses suggest that the antiviral activity of anti-NP IgG may provide a critically needed component of a universal influenza vaccine.
-
Nakatsukasa, H., et al (2015). "The DNA-binding inhibitor Id3 regulates IL-9 production in CD4(+) T cells" Nat Immunol 16(10): 1077-1084.
PubMed
The molecular mechanisms by which signaling via transforming growth factor-beta (TGF-beta) and interleukin 4 (IL-4) control the differentiation of CD4(+) IL-9-producing helper T cells (TH9 cells) remain incompletely understood. We found here that the DNA-binding inhibitor Id3 regulated TH9 differentiation, as deletion of Id3 increased IL-9 production from CD4(+) T cells. Mechanistically, TGF-beta1 and IL-4 downregulated Id3 expression, and this process required the kinase TAK1. A reduction in Id3 expression enhanced binding of the transcription factors E2A and GATA-3 to the Il9 promoter region, which promoted Il9 transcription. Notably, Id3-mediated control of TH9 differentiation regulated anti-tumor immunity in an experimental melanoma-bearing model in vivo and also in human CD4(+) T cells in vitro. Thus, our study reveals a previously unrecognized TAK1-Id3-E2A-GATA-3 pathway that regulates TH9 differentiation.
Product Citations
-
Macrophages eat more after disruption of cis interactions between CD47 and the checkpoint receptor SIRPα.
In Journal of Cell Science on 1 January 2020 by Hayes, B. H., Tsai, R. K., et al.
PubMed
The macrophage checkpoint receptor SIRPα signals against phagocytosis by binding CD47 expressed on all cells - including macrophages. Here, inhibiting cis interactions between SIRPα and CD47 on the same macrophage increases eating approximately the same as inhibiting trans interactions. Antibody blockade of CD47, as pursued in clinical trials against cancer, is applied separately to human-derived macrophages and to red blood cell (RBC) targets for phagocytosis, and both scenarios produce surprisingly similar increases in RBC engulfment. Blockade of both macrophages and targets results in hyper-phagocytosis, and knockdown of macrophage-CD47 likewise increases eating of 'foreign' cells and particles, decreases SIRPα's baseline inhibitory signaling, and linearly increases binding of soluble-CD47 in trans, consistent with cis-trans competition. Many cell types express both SIRPα and CD47, including mouse melanoma B16 cells, and CRISPR-mediated deletions modulate B16 phagocytosis consistent with cis-trans competition. Additionally, soluble SIRPα binding to human-CD47 displayed on Chinese hamster ovary (CHO) cells is suppressed by SIRPα co-display, and atomistic computations confirm SIRPα bends and binds CD47 in cis. Safety and efficacy profiles for CD47-SIRPα blockade might therefore reflect a disruption of both cis and trans interactions. © 2020. Published by The Company of Biologists Ltd.
-
Triggering the TCR Developmental Checkpoint Activates a Therapeutically Targetable Tumor Suppressive Pathway in T-cell Leukemia.
In Cancer Discovery on 1 September 2016 by Trinquand, A., dos Santos, N. R., et al.
PubMed
Cancer onset and progression involves the accumulation of multiple oncogenic hits, which are thought to dominate or bypass the physiologic regulatory mechanisms in tissue development and homeostasis. We demonstrate in T-cell acute lymphoblastic leukemia (T-ALL) that, irrespective of the complex oncogenic abnormalities underlying tumor progression, experimentally induced, persistent T-cell receptor (TCR) signaling has antileukemic properties and enforces a molecular program resembling thymic negative selection, a major developmental event in normal T-cell development. Using mouse models of T-ALL, we show that induction of TCR signaling by high-affinity self-peptide/MHC or treatment with monoclonal antibodies to the CD3ε chain (anti-CD3) causes massive leukemic cell death. Importantly, anti-CD3 treatment hampered leukemogenesis in mice transplanted with either mouse- or patient-derived T-ALLs. These data provide a strong rationale for targeted therapy based on anti-CD3 treatment of patients with TCR-expressing T-ALL and demonstrate that endogenous developmental checkpoint pathways are amenable to therapeutic intervention in cancer cells. T-ALLs are aggressive malignant lymphoid proliferations of T-cell precursors characterized by high relapse rates and poor prognosis, calling for the search for novel therapeutic options. Here, we report that the lineage-specific TCR/CD3 developmental checkpoint controlling cell death in normal T-cell progenitors remains switchable to induce massive tumor cell apoptosis in T-ALL and is amenable to preclinical therapeutic intervention. Cancer Discov; 6(9); 972-85. ©2016 AACR.See related commentary by Lemonnier and Mak, p. 946This article is highlighted in the In This Issue feature, p. 932. ©2016 American Association for Cancer Research.
-
Anti-cancer immune priming with β-radioligand therapy using a novel high affinity antibody selectively targeting the 4Ig-Isoform of B7-H3.
In Theranostics on 17 April 2026 by Glazer, S. E., Sutton, M. N., et al.
PubMed
Radioligand therapy (RLT) is an emerging oncologic strategy that uses molecularly targeted therapeutic radioisotopes to reduce tumor burden and improve survival in patients with advanced cancers. Expanding RLT to new targets and understanding its systemic immune effects could enhance its clinical impact. B7-H3 (CD276) is an attractive target due to its high differential expression in solid tumors compared to normal tissues. However, the presence of two isoforms, 4Ig-B7-H3 (tumor-associated) and 2Ig-B7-H3 (soluble decoy), poses challenges for selective targeting, especially in the context of RLT.
-
NK cells initiate an IFN-γ–dependent innate-to-adaptive immune cascade driving antitumor immunity upon ferroptosis induction
In Research Square on 2 April 2026 by Li, L., Zhang, Y., et al.