InVivoMAb human IgG2 isotype control
Product Description
Specifications
| Isotype | Human IgG2, λ |
|---|---|
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from human myeloma serum |
| Purification | Protein A |
| RRID | AB_2715459 |
| Molecular Weight | 150 kDa |
| Human Pathogen Test Results |
Hepatitis B Surface Antigen: Negative Human Immunodeficiency Virus 1 antibodies: Negative Human Immunodeficiency Virus 2 antibodies: Negative Hepatitis C Virus antibodies: Negative * These tests cannot guarantee the absence of infective agents |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Product Citations
-
Scope of Microbial Transglutaminase for Site-Specific and Oriented Immobilization of Native Antibodies from Various Host Species.
In Langmuir on 10 March 2026 by Beitello, E., Osei, K., et al.
PubMed
Modification of antibodies to chemically couple labels or immobilization reagents is essential for developing biosensors. Typically, conjugation occurs through chemical methods that leverage reactive amines and thiols on native antibodies; however, this nonspecific approach can interfere with antibody function. Microbial transglutaminase (mTG) is an enzyme that has been used for site-specific conjugation of chemical modifiers to the Fc region of native antibodies, but thus far mTG-mediated conjugation has been limited to production of antibody-drug conjugates with human IgGs. Here, we assessed the scope and versatility of mTG to target IgGs, with the goal of site-specific conjugation to facilitate oriented immobilization. A fluorescently labeled peptide was conjugated to several IgG host species and subclasses commonly used to produce monoclonal (e.g., mouse IgG1 and rat IgG1) and polyclonal (e.g., rabbit IgG and goat IgG) antibodies. SDS-PAGE confirmed site-specific conjugation of the peptide to each of these IgG subclasses. In addition, NH2-PEG4-biotin was chemo-enzymatically installed on the Fc region of each tested IgG, as confirmed by Western blot analysis. Site-specific biotinylated antibody was immobilized on a streptavidin-coated substrate to evaluate antigen binding activity in a functional assay. The site-specific conjugation of biotin enabled the formation of an oriented capture antibody layer to enhance antigen binding when compared to the performance of a functional assay constructed by immobilizing a randomly biotinylated antibody prepared by conventional chemical conjugation. These results highlight the broad scope of mTG to site-specifically conjugate native antibodies to improve analytical performance of biosensing platforms.
-
-
Cancer Research
A commonly inherited human PCSK9 germline variant drives breast cancer metastasis via LRP1 receptor.
In Cell on 23 January 2025 by Mei, W., Faraj Tabrizi, S., et al.
PubMed
Identifying patients at risk for metastatic relapse is a critical medical need. We identified a common missense germline variant in proprotein convertase subtilisin/kexin type 9 (PCSK9) (rs562556, V474I) that is associated with reduced survival in multiple breast cancer patient cohorts. Genetic modeling of this gain-of-function single-nucleotide variant in mice revealed that it causally promotes breast cancer metastasis. Conversely, host PCSK9 deletion reduced metastatic colonization in multiple breast cancer models. Host PCSK9 promoted metastatic initiation events in lung and enhanced metastatic proliferative competence by targeting tumoral low-density lipoprotein receptor related protein 1 (LRP1) receptors, which repressed metastasis-promoting genes XAF1 and USP18. Antibody-mediated therapeutic inhibition of PCSK9 suppressed breast cancer metastasis in multiple models. In a large Swedish early-stage breast cancer cohort, rs562556 homozygotes had a 22% risk of distant metastatic relapse at 15 years, whereas non-homozygotes had a 2% risk. Our findings reveal that a commonly inherited genetic alteration governs breast cancer metastasis and predicts survival-uncovering a hereditary basis underlying breast cancer metastasis.
-
-
-
Cancer Research
-
Endocrinology and Physiology
-
Immunology and Microbiology
-
Immunohistochemistry-immunofluorescence
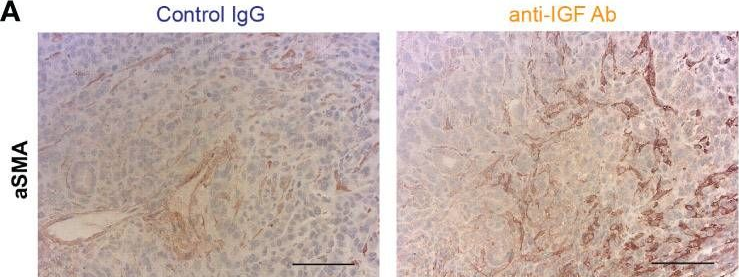
Inhibition of insulin-like growth factors increases production of CXCL9/10 by macrophages and fibroblasts and facilitates CD8+ cytotoxic T cell recruitment to pancreatic tumours.
In Front Immunol on 21 August 2024 by Freeman, P., Bellomo, G., et al.
PubMed
Pancreatic ductal adenocarcinoma (PDAC) is a highly lethal malignancy with an urgent unmet clinical need for new therapies. Using a combination of in vitro assays and in vivo preclinical models we demonstrate that therapeutic inhibition of the IGF signalling axis promotes the accumulation of CD8+ cytotoxic T cells within the tumour microenvironment of PDAC tumours. Mechanistically, we show that IGF blockade promotes macrophage and fibroblast production of the chemokines CXCL9 and CXCL10 to facilitate CD8+ T cell recruitment and trafficking towards the PDAC tumour. Exploring this pathway further, we show that IGF inhibition leads to increased STAT1 transcriptional activity, correlating with a downregulation of the AKT/STAT3 signalling axis, in turn promoting Cxcl9 and Cxcl10 gene transcription. Using patient derived tumour explants, we also demonstrate that our findings translate into the human setting. PDAC tumours are frequently described as "immunologically cold", therefore bolstering CD8+ T cell recruitment to PDAC tumours through IGF inhibition may serve to improve the efficacy of immune checkpoint inhibitors which rely on the presence of CD8+ T cells in tumours.
-
-
-
In vitro experiments
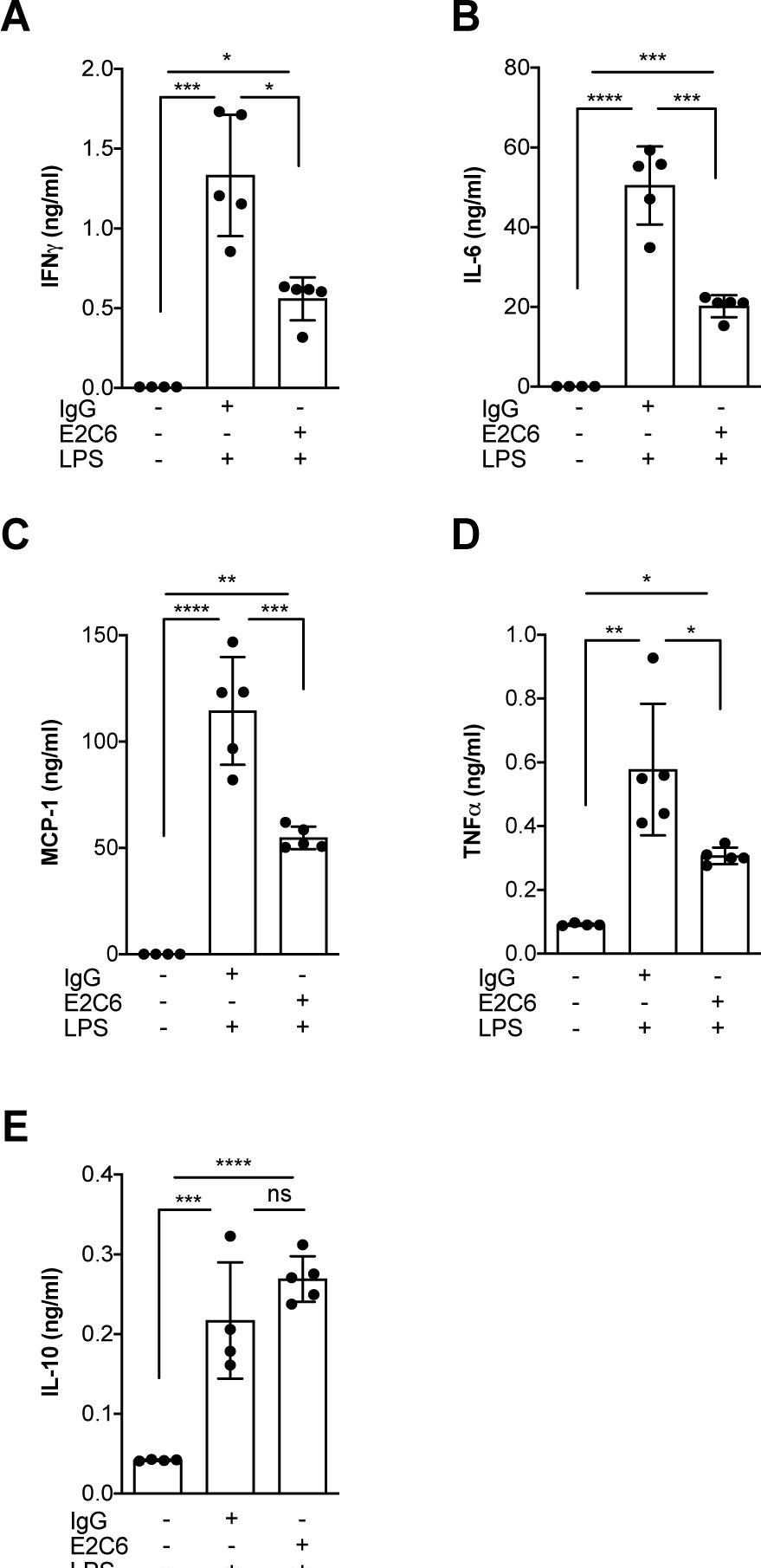
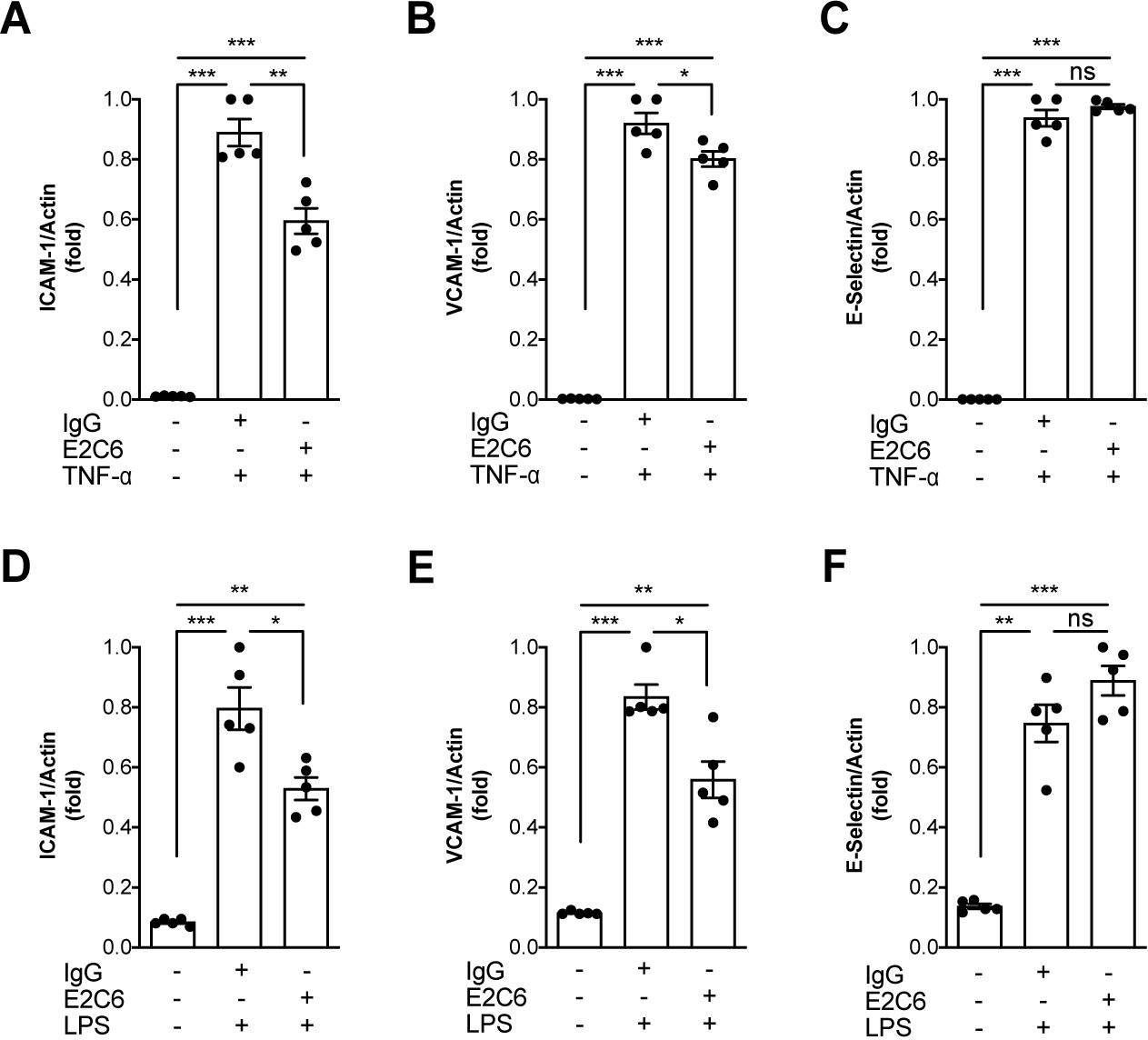
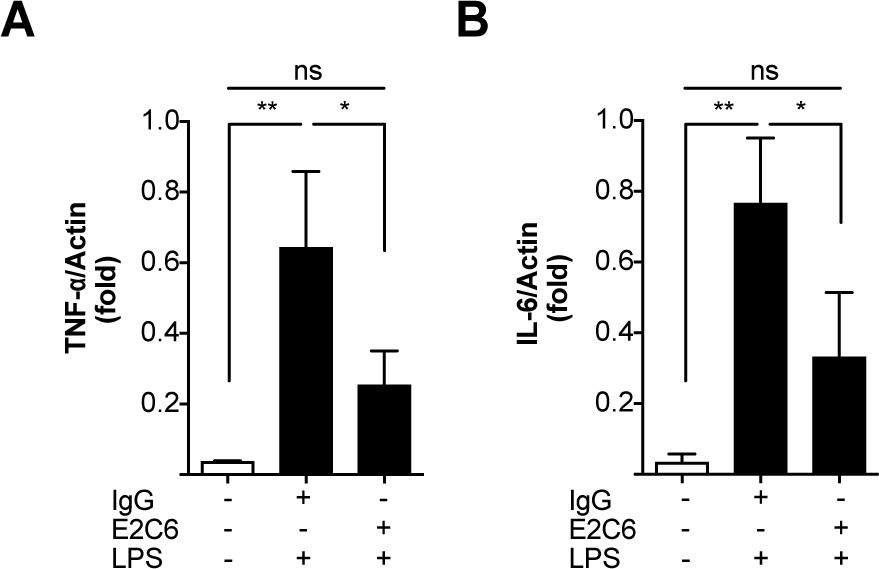
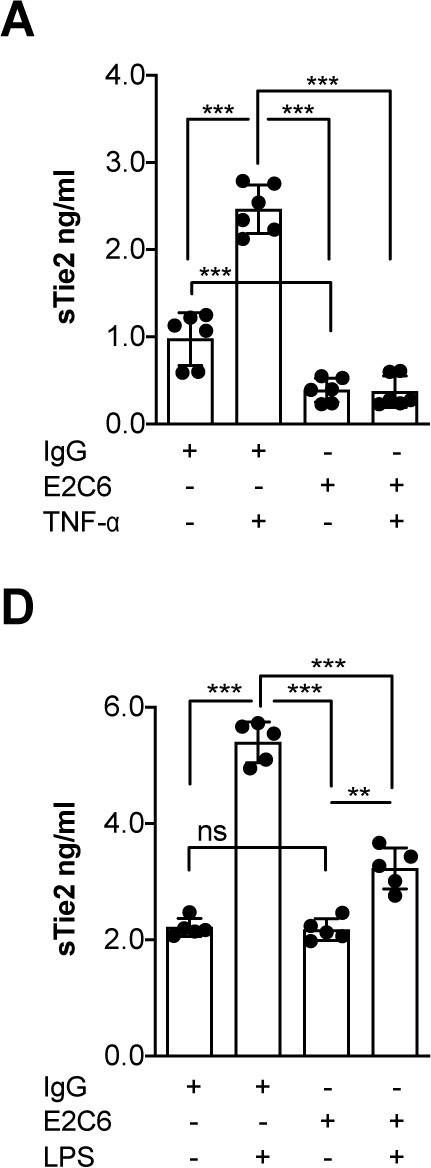
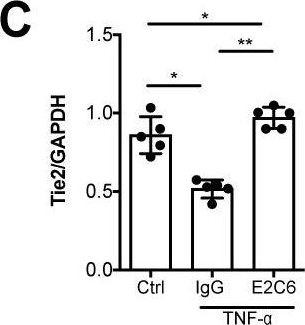
Identification of specific Tie2 cleavage sites and therapeutic modulation in experimental sepsis.
In Elife on 24 August 2020 by Idowu, T. O., Etzrodt, V., et al.
PubMed
Endothelial Tie2 signaling plays a pivotal role in vascular barrier maintenance at baseline and after injury. We previously demonstrated that a sharp drop in Tie2 expression observed across various murine models of critical illnesses is associated with increased vascular permeability and mortality. Matrix metalloprotease (MMP)-14-mediated Tie2 ectodomain shedding has recently been recognized as a possible mechanism for Tie2 downregulation in sepsis. Here, we identified the exact MMP14-mediated Tie2 ectodomain cleavage sites and could show that pharmacological MMP14 blockade in experimental murine sepsis exerts barrier protective and anti-inflammatory effects predominantly through the attenuation of Tie2 cleavage to improve survival both in a pre-treatment and rescue approach. Overall, we show that protecting Tie2 shedding might offer a new therapeutic opportunity for the treatment of septic vascular leakage.
-