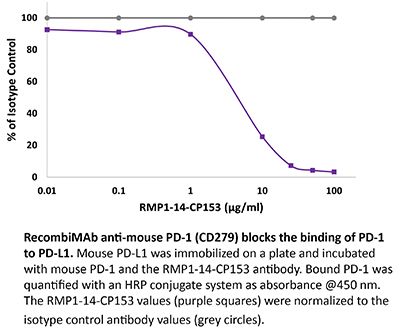
RecombiMAb anti-mouse PD-1 (CD279) (LALA-PG)
(switched from rat IgG2a)
Product Description
Specifications
| Isotype | Mouse IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | RecombiMAb mouse IgG2a (LALA-PG) isotype control, anti-hen egg lysozyme |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Mutations | LALA-PG |
| Immunogen | Syrian Hamster BKH cells transfected with mouse PD-1 cDNA |
| Reported Applications |
in vivo blocking of PD-1/PD-L signaling* *Reported for the original rat IgG2a RMP1-14 antibody |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<1EU/mg (<0.001EU/μg) Determined by LAL assay |
| Aggregation |
<5% Determined by DLS |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from CHO cell supernatant in an animal free facility |
| Purification | Protein G |
| Molecular Weight | 150 kDa |
| Murine Pathogen Tests |
Ectromelia/Mousepox Virus: Negative Hantavirus: Negative K Virus: Negative Lactate Dehydrogenase-Elevating Virus: Negative Lymphocytic Choriomeningitis virus: Negative Mouse Adenovirus: Negative Mouse Cytomegalovirus: Negative Mouse Hepatitis Virus: Negative Mouse Minute Virus: Negative Mouse Norovirus: Negative Mouse Parvovirus: Negative Mouse Rotavirus: Negative Mycoplasma Pulmonis: Negative Pneumonia Virus of Mice: Negative Polyoma Virus: Negative Reovirus Screen: Negative Sendai Virus: Negative Theiler’s Murine Encephalomyelitis: Negative |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Product Citations
-
Combination of a novel TCR Vβ chain-directed selective T cell activator with standard of care therapy for head and neck cancer improves antitumor responses and promotes regression of checkpoint-refractory tumor models.
In J Immunother Cancer on 28 October 2025 by Rosato, F., Santiago-Sánchez, G., et al.
PubMed
Immune checkpoint blockade (ICB) treatment, alone or in combination with standard anticancer therapies, has led to important progress in the treatment of head and neck squamous cell carcinoma (HNSCC). Yet, a significant proportion of patients with carcinogen-associated HNSCC (human papillomavirus (HPV)-) develop disease relapse or progression. Effective treatments for patients who have failed standard of care (SOC) treatment are lacking. STAR0602, a selective, bifunctional T cell agonist comprising an antibody targeting Vβ6 and Vβ10 T cell receptors fused to human interleukin-2, has demonstrated clinical activity in anti-programmed death-ligand 1 resistant tumors. A murine surrogate molecule, mSTAR1302, has been shown to induce tumor regression in multiple syngeneic tumor models and enhance antitumor activity in ICB-refractory settings. This study investigates the therapeutic benefit of mSTAR1302 combined with SOC in the mouse oral carcinoma (MOC)1 and MOC2 HNSCC tumor models.
-
Combination of HDAC inhibition and cytokine enhances therapeutic HPV vaccine therapy.
In J Immunother Cancer on 2 May 2025 by Poppe, L. K., Roller, N., et al.
PubMed
Human papillomavirus (HPV)-associated malignancies continue to present a major health concern despite the development of prophylactic vaccines. Standard therapies offer limited benefit to patients with advanced-stage disease. Despite improved outcomes with programmed cell death protein-1 (PD-1) targeted therapies, treatment resistance and modest response rates highlight a significant unmet need to develop novel therapies for these patients. PDS0101 (designated HPV vaccine) is a liposomal nanoparticle HPV16-specific therapeutic vaccine that has been shown to generate strong HPV-specific responses in preclinical and clinical studies. Here we assess the efficacy of this HPV vaccine in combination with the tumor-targeting immunocytokine NHS-IL12 (PDS01ADC), plus either αPD-1 or the class I histone deacetylase inhibitor Entinostat.