InVivoMAb anti-mouse CTLA-4 (CD152)
Product Description
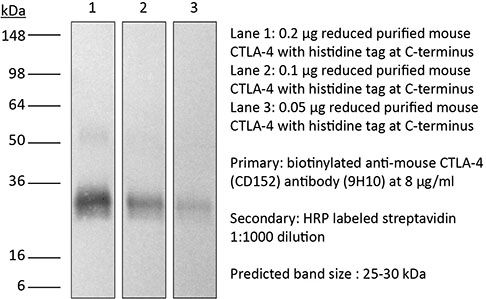
Specifications
| Isotype | Syrian hamster IgG |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb polyclonal Syrian hamster IgG |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse CTLA-4-human IgG1 fusion protein |
| Reported Applications |
in vivo CTLA-4 neutralization in vitro CTLA-4 neutralization Western blot |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_10950184 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Ariyan, C. E., et al (2018). "Robust Antitumor Responses Result from Local Chemotherapy and CTLA-4 Blockade" Cancer Immunol Res 6(2): 189-200.
PubMed
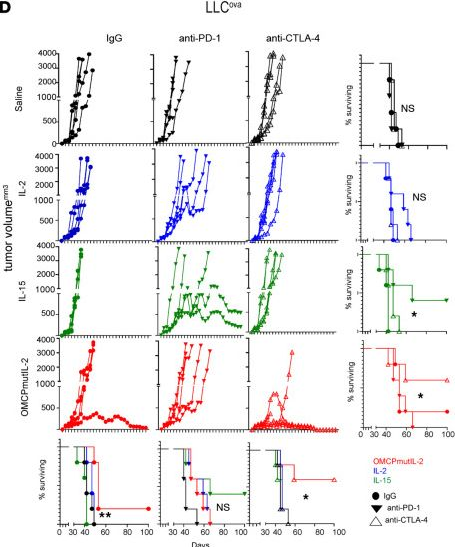
Clinical responses to immunotherapy have been associated with augmentation of preexisting immune responses, manifested by heightened inflammation in the tumor microenvironment. However, many tumors have a noninflamed microenvironment, and response rates to immunotherapy in melanoma have been <50%. We approached this problem by utilizing immunotherapy (CTLA-4 blockade) combined with chemotherapy to induce local inflammation. In murine models of melanoma and prostate cancer, the combination of chemotherapy and CTLA-4 blockade induced a shift in the cellular composition of the tumor microenvironment, with infiltrating CD8(+) and CD4(+) T cells increasing the CD8/Foxp3 T-cell ratio. These changes were associated with improved survival of the mice. To translate these findings into a clinical setting, 26 patients with advanced melanoma were treated locally by isolated limb infusion with the nitrogen mustard alkylating agent melphalan followed by systemic administration of CTLA-4 blocking antibody (ipilimumab) in a phase II trial. This combination of local chemotherapy with systemic checkpoint blockade inhibitor resulted in a response rate of 85% at 3 months (62% complete and 23% partial response rate) and a 58% progression-free survival at 1 year. The clinical response was associated with increased T-cell infiltration, similar to that seen in the murine models. Together, our findings suggest that local chemotherapy combined with checkpoint blockade-based immunotherapy results in a durable response to cancer therapy.
-
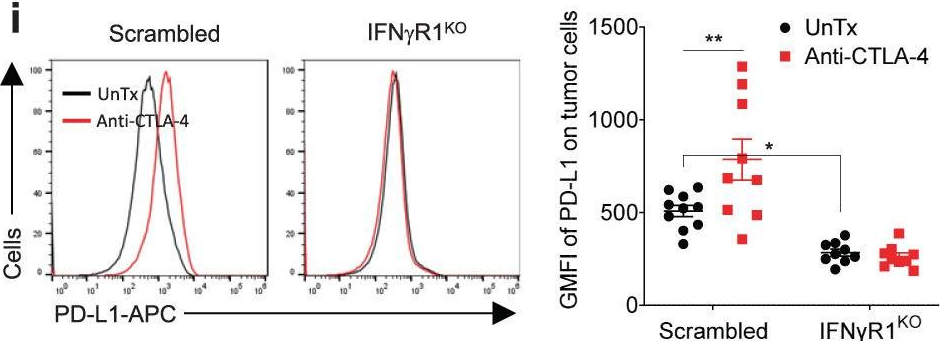
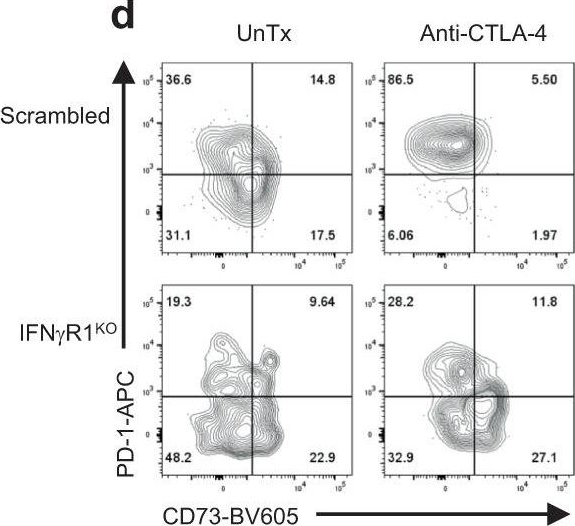
Gao, J., et al (2016). "Loss of IFN-gamma Pathway Genes in Tumor Cells as a Mechanism of Resistance to Anti-CTLA-4 Therapy" Cell 167(2): 397-404 e399.
PubMed
Antibody blockade of the inhibitory CTLA-4 pathway has led to clinical benefit in a subset of patients with metastatic melanoma. Anti-CTLA-4 enhances T cell responses, including production of IFN-gamma, which is a critical cytokine for host immune responses. However, the role of IFN-gamma signaling in tumor cells in the setting of anti-CTLA-4 therapy remains unknown. Here, we demonstrate that patients identified as non-responders to anti-CTLA-4 (ipilimumab) have tumors with genomic defects in IFN-gamma pathway genes. Furthermore, mice bearing melanoma tumors with knockdown of IFN-gamma receptor 1 (IFNGR1) have impaired tumor rejection upon anti-CTLA-4 therapy. These data highlight that loss of the IFN-gamma signaling pathway is associated with primary resistance to anti-CTLA-4 therapy. Our findings demonstrate the importance of tumor genomic data, especially IFN-gamma related genes, as prognostic information for patients selected to receive treatment with immune checkpoint therapy.
-
Bartkowiak, T., et al (2015). "Unique potential of 4-1BB agonist antibody to promote durable regression of HPV+ tumors when combined with an E6/E7 peptide vaccine" Proc Natl Acad Sci U S A 112(38): E5290-5299.
PubMed
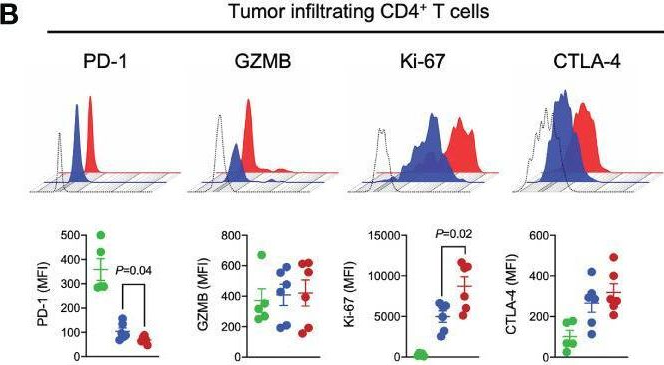
Antibody modulation of T-cell coinhibitory (e.g., CTLA-4) or costimulatory (e.g., 4-1BB) receptors promotes clinical responses to a variety of cancers. Therapeutic cancer vaccination, in contrast, has produced limited clinical benefit and no curative therapies. The E6 and E7 oncoproteins of human papilloma virus (HPV) drive the majority of genital cancers, and many oropharyngeal tumors. We discovered 15-19 amino acid peptides from HPV-16 E6/E7 for which induction of T-cell immunity correlates with disease-free survival in patients treated for high-grade cervical neoplasia. We report here that intranasal vaccination with these peptides and the adjuvant alpha-galactosylceramide elicits systemic and mucosal T-cell responses leading to reduced HPV(+) TC-1 tumor growth and prolonged survival in mice. We hypothesized that the inability of these T cells to fully reject established tumors resulted from suppression in the tumor microenvironment which could be ameliorated through checkpoint modulation. Combining this E6/E7 peptide vaccine with checkpoint blockade produced only modest benefit; however, coadministration with a 4-1BB agonist antibody promoted durable regression of established genital TC-1 tumors. Relative to other therapies tested, this combination of vaccine and alpha4-1BB promoted the highest CD8(+) versus regulatory FoxP3(+) T-cell ratios, elicited 2- to 5-fold higher infiltration by E7-specific CTL, and evoked higher densities of highly cytotoxic TcEO (T cytotoxic Eomesodermin) CD8 (>70-fold) and ThEO (T helper Eomesodermin) CD4 (>17-fold) T cells. These findings have immediate clinical relevance both in terms of the direct clinical utility of the vaccine studied and in illustrating the potential of 4-1BB antibody to convert therapeutic E6/E7 vaccines already in clinical trials into curative therapies.
-
Twyman-Saint Victor, C., et al (2015). "Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer" Nature 520(7547): 373-377.
PubMed
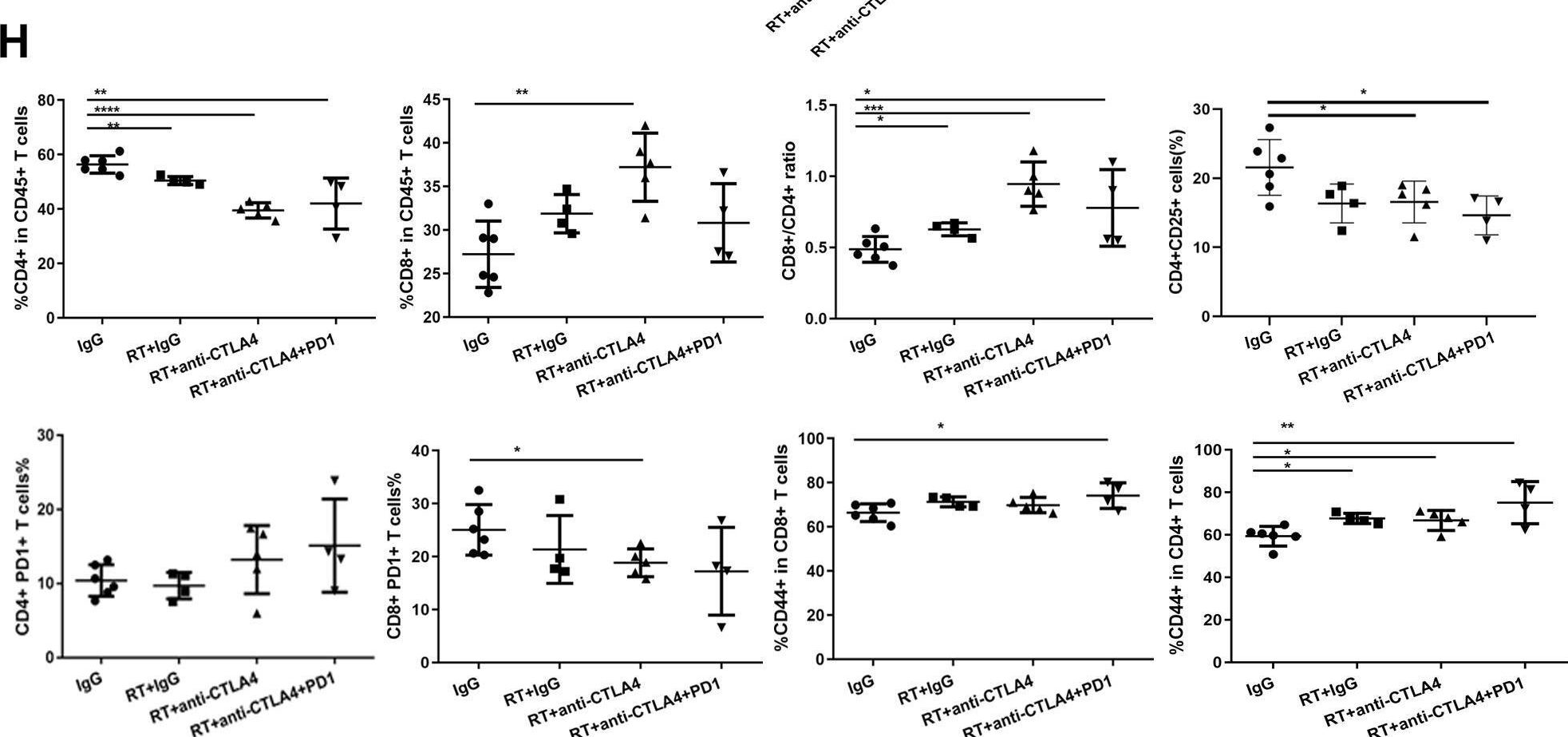
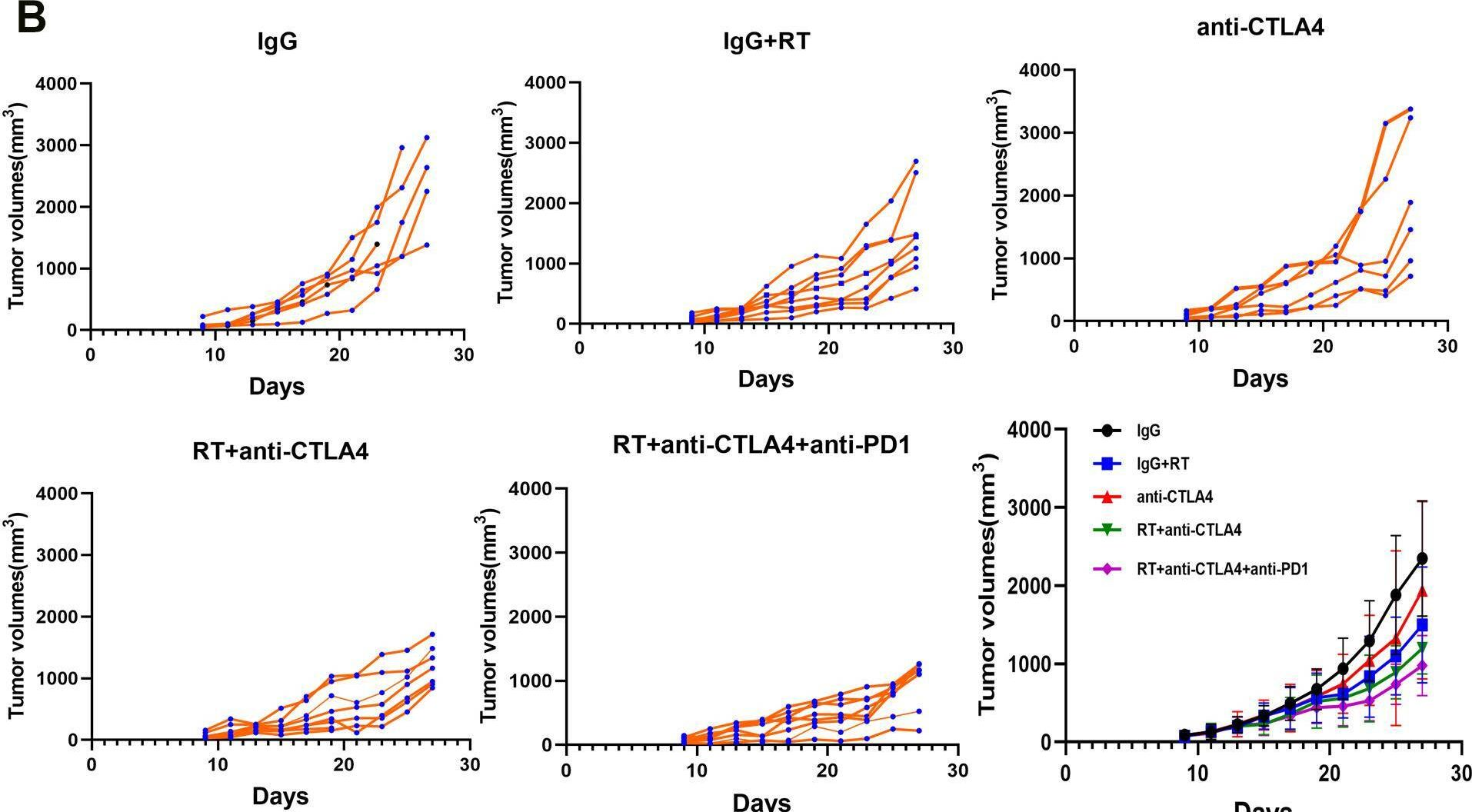
Immune checkpoint inhibitors result in impressive clinical responses, but optimal results will require combination with each other and other therapies. This raises fundamental questions about mechanisms of non-redundancy and resistance. Here we report major tumour regressions in a subset of patients with metastatic melanoma treated with an anti-CTLA4 antibody (anti-CTLA4) and radiation, and reproduced this effect in mouse models. Although combined treatment improved responses in irradiated and unirradiated tumours, resistance was common. Unbiased analyses of mice revealed that resistance was due to upregulation of PD-L1 on melanoma cells and associated with T-cell exhaustion. Accordingly, optimal response in melanoma and other cancer types requires radiation, anti-CTLA4 and anti-PD-L1/PD-1. Anti-CTLA4 predominantly inhibits T-regulatory cells (Treg cells), thereby increasing the CD8 T-cell to Treg (CD8/Treg) ratio. Radiation enhances the diversity of the T-cell receptor (TCR) repertoire of intratumoral T cells. Together, anti-CTLA4 promotes expansion of T cells, while radiation shapes the TCR repertoire of the expanded peripheral clones. Addition of PD-L1 blockade reverses T-cell exhaustion to mitigate depression in the CD8/Treg ratio and further encourages oligoclonal T-cell expansion. Similarly to results from mice, patients on our clinical trial with melanoma showing high PD-L1 did not respond to radiation plus anti-CTLA4, demonstrated persistent T-cell exhaustion, and rapidly progressed. Thus, PD-L1 on melanoma cells allows tumours to escape anti-CTLA4-based therapy, and the combination of radiation, anti-CTLA4 and anti-PD-L1 promotes response and immunity through distinct mechanisms.
Product Citations
-
Flt3L-mediated tumor cDC1 expansion enhances immunotherapy by priming stem-like CD8+ T cells in lymph nodes.
In Nat Immunol on 1 March 2026 by Lai, J., Chan, C. W., et al.
PubMed
Immune checkpoint blockade (ICB) evokes antitumor immunity through the reinvigoration of T cell responses. T cell differentiation status controls response, with less differentiated cells having an enhanced capacity to proliferate after ICB. Given that conventional type 1 dendritic cells (cDC1) maintain precursor exhausted T cells (TPEX), we hypothesized that expansion of cDC1s with Flt3L could enhance responses to ICB. Here we show that treatment with Fms-related tyrosine kinase 3 ligand (Flt3L) expands CD62L+SLAMF6+CD8+ T cells in the tumor through a mechanism that requires XCR1+ dendritic cells to traffic to the tumor-draining lymph node. The combination of Flt3L and anti-CTLA-4 enhanced therapeutic responses. Combination therapy is associated with the emergence of a CD8+ T cell subset characterized by the expression of Il21r and oligoclonal expansion of CD8+ T cells within tumors through a mechanism that is dependent on lymph node egress.
-
Mesothelioma location influences the tumour microenvironment and immune checkpoint therapy response in preclinical models.
In Sci Rep on 25 February 2026 by Orozco Morales, M. L., Lansley, S. M., et al.
PubMed
Mesothelioma is a cancer derived from mesothelial cells, most commonly arising from the pleura or the peritoneum. Immune checkpoint therapy (ICT) has shown survival benefit for pleural mesothelioma, but little is known about the response in peritoneal mesothelioma. Most preclinical mesothelioma models involve subcutaneous cancer cell implantation, which lacks the relevant tumour microenvironment of peritoneal mesothelioma and does not resemble the clinical presentation. We therefore set out to explore the influence of location on the mesothelioma tumour microenvironment, comparing pleural, peritoneal, and subcutaneous models using identical cell line-derived syngeneic mesotheliomas. We found that the peritoneal location conferred an anti-inflammatory tumour microenvironment, characterised by low IFN, TNFα, and STAT signalling activity, low immune cell infiltration and a gene signature associated with non-response to ICT. ICT was effective in subcutaneous models, but the same cell line-derived tumours were irresponsive when inoculated intraperitoneally. Together, these findings show that peritoneal location is associated with an immune suppressive tumour microenvironment.
-
Programmable iPSC-derived CAR-NK vesicles remodel the immune microenvironment and eradicate tumors.
In Cell Rep Med on 17 February 2026 by Zhang, H., Li, S., et al.
PubMed
Chimeric antigen receptor (CAR) cell therapy transforms hematologic cancer treatment but remains limited in solid tumors due to stromal barriers and an immunosuppressive tumor microenvironment that restricts immune cell infiltration. To address these barriers, we develop a cell-free therapeutic platform based on CAR-engineered induced pluripotent stem cell (iPSC)-derived natural killer (NK) extracellular vesicles (CAR-iNEVs), which retain tumor-targeting capability without reliance on live-cell delivery. CAR-iNEV demonstrates potent antitumor activity and excellent tolerability across multiple xenograft and patient-derived models. Mechanistically, CAR-iNEV directly eliminates tumor cells and remodels the tumor microenvironment by promoting pro-inflammatory macrophage polarization, thereby enhancing host innate antitumor immunity. CAR-iNEV also functions cooperatively with immune checkpoint blockade, and combined treatment with CAR-iNEV and CD47 inhibition increases tumor clearance and induces long-term immunological memory in surviving mice. These findings support the therapeutic potential of CAR-iNEV for solid tumors through coordinated tumor targeting and immune microenvironment modulation.
-
Oral immunization with Listeria monocytogenes vaccine enhances immunotherapy for protective immunity in murine models of colorectal cancer.
In J Immunother Cancer on 5 February 2026 by Lei, X., Yu, Y., et al.
PubMed
Colorectal cancer (CRC) is a leading cause of cancer-related death and remains a significant global health challenge. Cancer vaccines have emerged as a promising immunotherapy for long-term tumor control. While Listeria monocytogenes (Lm)-based intravenous vaccines can generate tumor-reactive CD8 T cells, clinical trial success has been limited. Here, we sought to determine whether in vivo targeting of gastrointestinal tissues with foodborne delivery of Lm-based cancer vaccines controlled tumor growth in murine models of CRC.