InVivoMAb anti-mouse VEGFR-2
Product Description
Specifications
| Isotype | Rat IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG1 isotype control, anti-horseradish peroxidase |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse VEGFR-2-SEAPs soluble receptor |
| Reported Applications |
in vivo blocking of VEGF/VEGFR-2 signaling in vitro blocking of VEGFR signaling Western blot |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107766 |
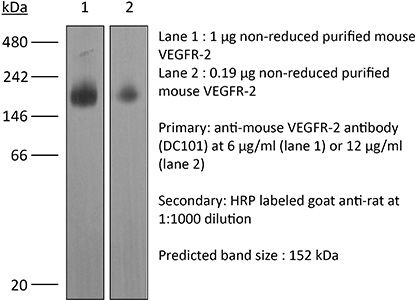
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Arulanandam, R., et al (2015). "VEGF-Mediated Induction of PRD1-BF1/Blimp1 Expression Sensitizes Tumor Vasculature to Oncolytic Virus Infection" Cancer Cell 28(2): 210-224.
PubMed
Oncolytic viruses designed to attack malignant cells can in addition infect and destroy tumor vascular endothelial cells. We show here that this expanded tropism of oncolytic vaccinia virus to the endothelial compartment is a consequence of VEGF-mediated suppression of the intrinsic antiviral response. VEGF/VEGFR2 signaling through Erk1/2 and Stat3 leads to upregulation, nuclear localization, and activation of the transcription repressor PRD1-BF1/Blimp1. PRD1-BF1 does not contribute to the mitogenic effects of VEGF, but directly represses genes involved in type I interferon (IFN)-mediated antiviral signaling. In vivo suppression of VEGF signaling diminishes PRD1-BF1/Blimp1 expression in tumor vasculature and inhibits intravenously administered oncolytic vaccinia delivery to and consequent spread within the tumor.
-
Ding, X., et al (2015). "Distinct functions of epidermal and myeloid-derived VEGF-A in skin tumorigenesis mediated by HPV8" Cancer Res 75(2): 330-343.
PubMed
Beta human papillomaviruses (HPV) have been suspected to be carcinogenic in nonmelanoma skin cancers (NMSC), but the basis for potential viral contributions to these cancers is poorly understood. In particular, it is unresolved how HPV-infected keratinocytes escape cell-cycle control and whether their cross-talk with immune cells is critical for tumorigenesis. In nonviral preclinical models, the angiogenic cytokine VEGF-A has been identified as a critical regulator of NMSC. In this study, we dissected the contribution of epidermal versus myeloid cell-derived VEGF-A in HPV-mediated skin cancer by interbreeding an HPV8 transgenic mouse model with a conditional disruption of VEGF-A restricted to either epidermal or myeloid cells. Although only epidermal-derived VEGF-A was essential for initiation of skin tumor development, both spontaneously and UV-light triggered, both epidermal and myeloid cell-derived VEGF-A contributed to regeneration-induced tumorigenesis upon HPV8 overexpression, partly not only through a paracrine effect on endothelial cells, but also most probably through an additional autocrine effect on epidermal cells. Our findings offer new mechanistic insights into distinct functions of epidermal versus myeloid cell-derived VEGF-A during HPV-mediated tumorigenesis, with possible implications for preventing this disease.
-
Lee, H. J., et al (2015). "Inhibition of vascular endothelial growth factor A and hypoxia-inducible factor 1alpha maximizes the effects of radiation in sarcoma mouse models through destruction of tumor vasculature" Int J Radiat Oncol Biol Phys 91(3):
PubMed
PURPOSE: To examine the addition of genetic or pharmacologic inhibition of hypoxia-inducible factor 1alpha (HIF-1alpha) to radiation therapy (RT) and vascular endothelial growth factor A (VEGF-A) inhibition (ie trimodality therapy) for soft-tissue sarcoma. METHODS AND MATERIALS: Hypoxia-inducible factor 1alpha was inhibited using short hairpin RNA or low metronomic doses of doxorubicin, which blocks HIF-1alpha binding to DNA. Trimodality therapy was examined in a mouse xenograft model and a genetically engineered mouse model of sarcoma, as well as in vitro in tumor endothelial cells (ECs) and 4 sarcoma cell lines. RESULTS: In both mouse models, any monotherapy or bimodality therapy resulted in tumor growth beyond 250 mm(3) within the 12-day treatment period, but trimodality therapy with RT, VEGF-A inhibition, and HIF-1alpha inhibition kept tumors at <250 mm(3) for up to 30 days. Trimodality therapy on tumors reduced HIF-1alpha activity as measured by expression of nuclear HIF-1alpha by 87% to 95% compared with RT alone, and cytoplasmic carbonic anhydrase 9 by 79% to 82%. Trimodality therapy also increased EC-specific apoptosis 2- to 4-fold more than RT alone and reduced microvessel density by 75% to 82%. When tumor ECs were treated in vitro with trimodality therapy under hypoxia, there were significant decreases in proliferation and colony formation and increases in DNA damage (as measured by Comet assay and gammaH2AX expression) and apoptosis (as measured by cleaved caspase 3 expression). Trimodality therapy had much less pronounced effects when 4 sarcoma cell lines were examined in these same assays. CONCLUSIONS: Inhibition of HIF-1alpha is highly effective when combined with RT and VEGF-A inhibition in blocking sarcoma growth by maximizing DNA damage and apoptosis in tumor ECs, leading to loss of tumor vasculature.
-
Kizhatil, K., et al (2014). "Schlemm’s canal is a unique vessel with a combination of blood vascular and lymphatic phenotypes that forms by a novel developmental process" PLoS Biol 12(7): e1001912.
PubMed
Schlemm’s canal (SC) plays central roles in ocular physiology. These roles depend on the molecular phenotypes of SC endothelial cells (SECs). Both the specific phenotype of SECs and development of SC remain poorly defined. To allow a modern and extensive analysis of SC and its origins, we developed a new whole-mount procedure to visualize its development in the context of surrounding tissues. We then applied genetic lineage tracing, specific-fluorescent reporter genes, immunofluorescence, high-resolution confocal microscopy, and three-dimensional (3D) rendering to study SC. Using these techniques, we show that SECs have a unique phenotype that is a blend of both blood and lymphatic endothelial cell phenotypes. By analyzing whole mounts of postnatal mouse eyes progressively to adulthood, we show that SC develops from blood vessels through a newly discovered process that we name “canalogenesis.” Functional inhibition of KDR (VEGFR2), a critical receptor in initiating angiogenesis, shows that this receptor is required during canalogenesis. Unlike angiogenesis and similar to stages of vasculogenesis, during canalogenesis tip cells divide and form branched chains prior to vessel formation. Differing from both angiogenesis and vasculogenesis, during canalogenesis SECs express Prox1, a master regulator of lymphangiogenesis and lymphatic phenotypes. Thus, SC development resembles a blend of vascular developmental programs. These advances define SC as a unique vessel with a combination of blood vascular and lymphatic phenotypes. They are important for dissecting its functions that are essential for ocular health and normal vision.
Product Citations
-
IDO1 regulating ROS rhythm reveals glycogenolysis/PPP as a cancer treatment target.
In Nat Chem Biol on 13 March 2026 by Zhou, N., Ling, Z., et al.
PubMed
Reactive oxygen species (ROS) dynamics exhibits rhythmic oscillations in cancer cells but how this rhythm influences tumorigenesis and therapeutic responses remains unclear. Here we found coexistence of ROS rhythmicity and rhythm loss in tumor samples. Under low-ROS conditions, indoleamine 2,3-dioxygenase 1 (IDO1), an immune-checkpoint molecule, binds to KEAP1 for proteasomal degradation in the nucleus. In contrast, elevated ROS levels drive IDO1 translocation into the cytosol, where it binds mitochondria-released heme to form an active holoenzyme. This holoenzyme catalyzes tryptophan to kynurenine that allosterically activates glucose-6-phosphate dehydrogenase, enhancing NADPH production and promoting ROS clearance. However, in hypoxic tumor microenvironments, ROS rhythmicity is lost. Compensating for this, hypoxic tumor cells mobilize the sulfenylated aryl hydrocarbon receptor (AhR)-mediated glycogenolysis pathway to manage disordered ROS accumulation, maintaining elevated ROS levels that favor tumor growth. Dual inhibition of IDO1 and AhR significantly prolongs survival of NSG mice, highlighting enforced disruption of ROS rhythm as a common therapeutic strategy.
-
The influence of VEGFR-2 blockade and focused ultrasound blood-brain barrier opening on the glioma-immune landscape.
In Neurooncol Adv on 10 February 2026 by Nowak, K. M., Hoch, M. R., et al.
PubMed
Glioblastoma (GBM) is an aggressive brain cancer with limited treatment options and high recurrence rates. The blood-brain barrier (BBB) impedes therapeutic delivery for the brain, limiting systemic treatment efficacy. Focused ultrasound (FUS) combined with microbubbles (MBs) can transiently open the BBB (BBBO), enhancing drug delivery and modulating the tumor immune microenvironment (TME). However, the disorganized and leaky vasculature in GBM limits the effectiveness of FUS-mediated BBBO. Vascular normalization using antiangiogenic therapy may enhance both immune modulation and delivery. This study aimed to investigate whether vascular normalization via VEGFR-2 blockade with DC101, alone or in combination with FUS+MBs, improves TME remodeling in a murine GBM model.
-
Murine vascular organoids are responsive and adaptable 3D systems with cellular heterogeneity and dynamic plasticity.
In Sci Adv on 24 October 2025 by Guelfi, S., Skandha Gopalan, K., et al.
PubMed
Blood vascular organoids (BVOs) have emerged as powerful tools for interrogating endothelial heterogeneity, function, and organ-specific cues during development, homeostasis, and disease. Here, we describe the stepwise generation and characterization of murine BVOs from different embryonic stem cells and introduce a fluorescent tracer system to follow their formation and maturation, visualizing functional and perfused vessels after transplantations into mice. Single-cell RNA sequencing, FACS, and whole-mount immunohistochemistry revealed a heterogeneous and dynamic trajectory of multiple endothelial, mesenchymal, and macrophage clusters in mBVOs at growth spike and full maturation. Endothelial cells inferred high plasticity toward arterial, venous, capillary, and tip/stalk cells and a neural-like identity at full maturation state. mBVOs responded to inflammatory cues and VEGF blockade in vitro and could be assembled with tumor spheroids in which tumor cells mimicked the same vascular growth pattern as observed in vivo. Our results highlight mBVOs as adaptable in vitro platforms, unlocking various applications and avenues for discovery in vascular biology.
-
VEGFR2 blockade converts thermally ablative focused ultrasound into a potent driver of T cell-dependent anti-tumor immunity
In bioRxiv on 24 October 2025 by Schwartz, M. R., Anwar, N. Z., et al.