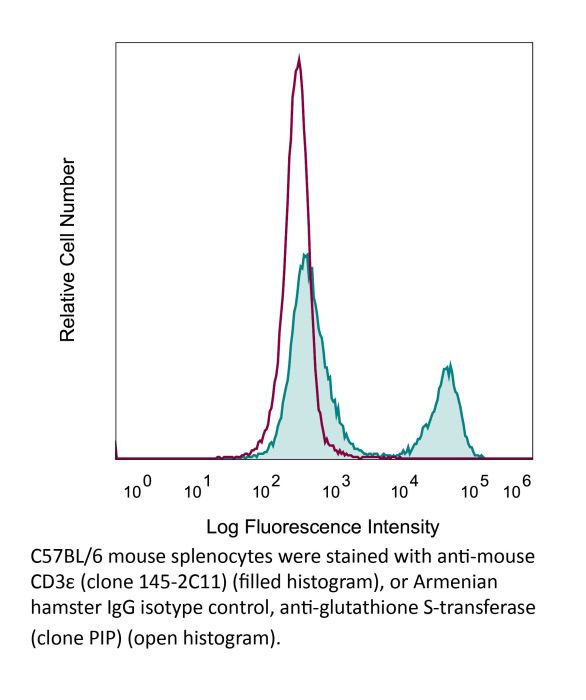
InVivoMAb anti-mouse CD3ε
Product Description
Specifications
| Isotype | Armenian Hamster IgG1 |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb polyclonal Armenian hamster IgG |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse BM10-37 cytotoxic T cells |
| Reported Applications |
in vivo T cell depletion in vitro T cell stimulation/activation Immunofluorescence Flow cytometry Western blot |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_1107634 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Tang, W., et al (2014). "The oncoprotein and transcriptional regulator Bcl-3 governs plasticity and pathogenicity of autoimmune T cells" Immunity 41(4): 555-566.
PubMed
Bcl-3 is an atypical member of the IkappaB family that modulates transcription in the nucleus via association with p50 (NF-kappaB1) or p52 (NF-kappaB2) homodimers. Despite evidence attesting to the overall physiologic importance of Bcl-3, little is known about its cell-specific functions or mechanisms. Here we demonstrate a T-cell-intrinsic function of Bcl-3 in autoimmunity. Bcl-3-deficient T cells failed to induce disease in T cell transfer-induced colitis and experimental autoimmune encephalomyelitis. The protection against disease correlated with a decrease in Th1 cells that produced the cytokines IFN-gamma and GM-CSF and an increase in Th17 cells. Although differentiation into Th1 cells was not impaired in the absence of Bcl-3, differentiated Th1 cells converted to less-pathogenic Th17-like cells, in part via mechanisms involving expression of the RORgammat transcription factor. Thus, Bcl-3 constrained Th1 cell plasticity and promoted pathogenicity by blocking conversion to Th17-like cells, revealing a unique type of regulation that shapes adaptive immunity.
-
Berger, H., et al (2013). "SOCS3 transactivation by PPARgamma prevents IL-17-driven cancer growth" Cancer Res 73(12): 3578-3590.
PubMed
Activation of the transcription factor PPARgamma by the n-3 fatty acid docosahexaenoic acid (DHA) is implicated in controlling proinflammatory cytokine secretion, but the intracellular signaling pathways engaged by PPARgamma are incompletely characterized. Here, we identify the adapter-encoding gene SOCS3 as a critical transcriptional target of PPARgamma. SOCS3 promoter binding and gene transactivation by PPARgamma was associated with a repression in differentiation of proinflammatory T-helper (TH)17 cells. Accordingly, TH17 cells induced in vitro displayed increased SOCS3 expression and diminished capacity to produce interleukin (IL)-17 following activation of PPARgamma by DHA. Furthermore, naive CD4 T cells derived from mice fed a DHA-enriched diet displayed less capability to differentiate into TH17 cells. In two different mouse models of cancer, DHA prevented tumor outgrowth and angiogenesis in an IL-17-dependent manner. Altogether, our results uncover a novel molecular pathway by which PPARgamma-induced SOCS3 expression prevents IL-17-mediated cancer growth.
-
Rabenstein, H., et al (2014). "Differential kinetics of antigen dependency of CD4+ and CD8+ T cells" J Immunol 192(8): 3507-3517.
PubMed
Ag recognition via the TCR is necessary for the expansion of specific T cells that then contribute to adaptive immunity as effector and memory cells. Because CD4+ and CD8+ T cells differ in terms of their priming APCs and MHC ligands we compared their requirements of Ag persistence during their expansion phase side by side. Proliferation and effector differentiation of TCR transgenic and polyclonal mouse T cells were thus analyzed after transient and continuous TCR signals. Following equally strong stimulation, CD4+ T cell proliferation depended on prolonged Ag presence, whereas CD8+ T cells were able to divide and differentiate into effector cells despite discontinued Ag presentation. CD4+ T cell proliferation was neither affected by Th lineage or memory differentiation nor blocked by coinhibitory signals or missing inflammatory stimuli. Continued CD8+ T cell proliferation was truly independent of self-peptide/MHC-derived signals. The subset divergence was also illustrated by surprisingly broad transcriptional differences supporting a stronger propensity of CD8+ T cells to programmed expansion. These T cell data indicate an intrinsic difference between CD4+ and CD8+ T cells regarding the processing of TCR signals for proliferation. We also found that the presentation of a MHC class II-restricted peptide is more efficiently prolonged by dendritic cell activation in vivo than a class I bound one. In summary, our data demonstrate that CD4+ T cells require continuous stimulation for clonal expansion, whereas CD8+ T cells can divide following a much shorter TCR signal.
-
Gu, A. D., et al (2015). "A critical role for transcription factor Smad4 in T cell function that is independent of transforming growth factor beta receptor signaling" Immunity 42(1): 68-79.
PubMed
Transforming growth factor-beta (TGF-beta) suppresses T cell function to maintain self-tolerance and to promote tumor immune evasion. Yet how Smad4, a transcription factor component of TGF-beta signaling, regulates T cell function remains unclear. Here we have demonstrated an essential role for Smad4 in promoting T cell function during autoimmunity and anti-tumor immunity. Smad4 deletion rescued the lethal autoimmunity resulting from transforming growth factor-beta receptor (TGF-betaR) deletion and compromised T-cell-mediated tumor rejection. Although Smad4 was dispensable for T cell generation, homeostasis, and effector function, it was essential for T cell proliferation after activation in vitro and in vivo. The transcription factor Myc was identified to mediate Smad4-controlled T cell proliferation. This study thus reveals a requirement of Smad4 for T-cell-mediated autoimmunity and tumor rejection, which is beyond the current paradigm. It highlights a TGF-betaR-independent role for Smad4 in promoting T cell function, autoimmunity, and anti-tumor immunity.
Product Citations
-
The effector differentiation of TCRαβ+CD8αα+ intraepithelial lymphocytes is reciprocally regulated by BCL6 and BLIMP1.
In Cell Rep on 20 March 2026 by Xing, Q., Xie, S., et al.
PubMed
CD8αα+TCRαβ+ intraepithelial lymphocytes (IELs) play crucial roles in maintaining intestinal homeostasis and host protection. However, the functional regulation of these cells remains unclear. Here, we have discovered and characterized two distinct developmental stages within intestinal CD8αα+ αβ IELs: a stem-like, defined by BCL6, TCF1, and CD160 expression, and an effector-like, with granzyme B expression and Prdm1 transcription. The differentiation from stem-like to effector-like CD8αα+ αβ IELs is promoted by T cell receptor (TCR) and IL-12 signaling and is controlled by the opposing actions of BCL6 and BLIMP1. Loss of BCL6 promotes the development of effector-like CD8αα+ αβ IELs, leading to increased effector molecule expression and heightened inflammation under dextran sodium sulfate (DSS)-induced colitis. In contrast, BLIMP1 deficiency perturbs the effector differentiation and reduces the susceptibility to gut inflammation. Our study thus reveals a critical antagonistic function between BCL6 and BLIMP1 in governing the fate decisions of CD8αα+ αβ IEL subsets.
-
SLC7A5 regulates B cell metabolism and plasma cell differentiation independent of leucine transport.
In J Immunol on 17 March 2026 by Tao, A. Y., Hu, K., et al.
PubMed
B cells play critical roles in humoral immunity to infection, vaccination, and autoimmunity. The differentiation of B cells into antibody-producing plasma cells (PCs) has been extensively studied, but the role of metabolic transporters that mediate nutrient uptake during PC differentiation is not well-understood. Here, we characterized the dependence of B cells and PC differentiation on the neutral amino acid transporter SLC7A5. We demonstrate that SLC7A5 promotes B cell functions including proliferation and PC differentiation in vitro and in vivo after immunization with T dependent and independent antigens. Deletion of SLC7A5 in B cells suppressed the function of mTORC1 and enforced mTORC1 activity rescued PC differentiation. The role of SLC7A5 in B cells appears to be unrelated to leucine uptake because B cells were insensitive to extracellular leucine depletion. Defects in SLC7A5-deficient B cells could, however, be rescued by extracellular methionine supplementation, suggesting a role for methionine in SLC7A5-dependent B cell function and PC differentiation. Our study provides evidence for a leucine-independent role of SLC7A5 in B cell function and PC differentiation.
-
Spatial tumor evolution panorama of ovarian cancer.
In Cell Rep Med on 17 March 2026 by Feng, C., Yang, Y., et al.
PubMed
Ovarian cancer is an aggressive disease characterized by intraperitoneal dissemination and a distinctive microenvironment. By generating metastatic cohorts encompassing approximately 60 pairs of whole-genome and RNA sequencing, 100 single-cell samples, and 2.5 million spatial transcriptomics (ST) spots, we delineate site-specific tumor-host colocalization patterns. Utilizing our STARLETS framework, we elucidate a Darwinian evolutionary trajectory in which hypoxia and immune pressures select for clones that eventually metastasize. High-resolution ST and ultimate dimensional imaging of solvent-cleared organs (uDISCO) imaging further identify a tripartite ensemble comprising MMP11+ myCAFs, epithelial cells, and SPP1+ macrophages in ascites and metastases, which can be modulated via SPP1-CD44 inhibition. SPP1+ macrophages predict therapeutic responses in clinical trials, including oncolytic virus and poly(ADP-ribose) polymerase inhibitor treatments. Collectively, our study advances insights into spatial dynamics that hold promise for therapeutic approaches in ovarian cancer.
-
Uridine depletion impairs CD8⁺ T cell antitumor activity through N-glycosylation.
In Cell Metab on 3 March 2026 by Xiao, J., Li, Z., et al.
PubMed
Immune checkpoint blockade (ICB) faces limitations owing to high cost and restricted efficacy. This study identifies SNX17 as a key mediator of ICB resistance. Elevated SNX17 correlates with poor anti-PD-1 response in humans and mice. SNX17 deletion in tumor cells inhibits tumor growth via CD8+ T cell-dependent mechanisms. SNX17 reduces uridine in the tumor microenvironment (TME), suppressing IFN-γ and upregulating PD1 in CD8+ T cells. Exogenous uridine shows antitumor efficacy comparable to anti-PD-1/PD-L1 in low-SNX17 tumors and overcomes resistance in high-SNX17 models. Uridine enhances CD8+ T cell function by promoting CD45 N-glycosylation and LCK phosphorylation. Mechanistically, SNX17 stabilizes RUNX2, promoting UPP1 transcription and uridine degradation in the TME. These findings position SNX17 as an ICB response biomarker and nominate uridine as a cost-effective immunotherapeutic strategy.