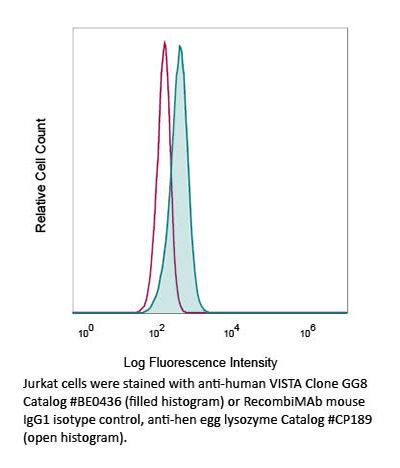
InVivoMAb anti-human VISTA
Product Description
Specifications
| Isotype | Mouse IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG1 isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Immunogen | Not available or unknown |
| Reported Applications |
Immunohistochemistry-paraffin Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Lines JL, Sempere LF, Broughton T, Wang L, Noelle R (2014). "VISTA is a novel broad-spectrum negative checkpoint regulator for cancer immunotherapy" Cancer Immunol Res 10.1158/2326-6066.CIR-14-0072.
PubMed
In the past few years, the field of cancer immunotherapy has made great progress and is finally starting to change the way cancer is treated. We are now learning that multiple negative checkpoint regulators (NCR) restrict the ability of T-cell responses to effectively attack tumors. Releasing these brakes through antibody blockade, first with anti-CTLA4 and now followed by anti-PD1 and anti-PDL1, has emerged as an exciting strategy for cancer treatment. More recently, a new NCR has surfaced called V-domain immunoglobulin (Ig)-containing suppressor of T-cell activation (VISTA). This NCR is predominantly expressed on hematopoietic cells, and in multiple murine cancer models is found at particularly high levels on myeloid cells that infiltrated the tumors. Preclinical studies with VISTA blockade have shown promising improvement in antitumor T-cell responses, leading to impeded tumor growth and improved survival. Clinical trials support combined anti-PD1 and anti-CTLA4 as safe and effective against late-stage melanoma. In the future, treatment may involve combination therapy to target the multiple cell types and stages at which NCRs, including VISTA, act during adaptive immune responses.
-
Kuklinski LF, Yan S, Li Z, Fisher JL, Cheng C, Noelle RJ, Angeles CV, Turk MJ, Ernstoff MS (2018). "VISTA expression on tumor-infiltrating inflammatory cells in primary cutaneous melanoma correlates with poor disease-specific survival" Cancer Immunol
PubMed
Adaptive immune responses contribute to the pathogenesis of melanoma by facilitating immune evasion. V-domain Ig suppressor of T-cell activation (VISTA) is a potent negative regulator of T-cell function and is expressed at high levels on monocytes, granulocytes, and macrophages, and at lower densities on T-cell populations within the tumor microenvironment. In this study, 85 primary melanoma specimens were selected from pathology tissue archives and immunohistochemically stained for CD3, PD-1, PD-L1, and VISTA. Pearson's correlation coefficients identified associations in expression between VISTA and myeloid infiltrate (r = 0.28, p = 0.009) and the density of PD-1+ inflammatory cells (r = 0.31, p = 0.005). The presence of VISTA was associated with a significantly worse disease-specific survival in univariate analysis (hazard ratio = 3.57, p = 0.005) and multivariate analysis (hazard ratio = 3.02, p = 0.02). Our findings show that VISTA expression is an independent negative prognostic factor in primary cutaneous melanoma and suggests its potential as an adjuvant immunotherapeutic intervention in the future.
-
Lines JL, Pantazi E, Mak J, Sempere LF, Wang L, O', Connell S, Ceeraz S, Suriawinata AA, Yan S, Ernstoff MS, Noelle R (2014). "VISTA is an immune checkpoint molecule for human T cells" Cancer Res 10.1158/0008-5472.CAN-13-1504.
PubMed
V-domain Ig suppressor of T cell activation (VISTA) is a potent negative regulator of T-cell function that is expressed on hematopoietic cells. VISTA levels are heightened within the tumor microenvironment, in which its blockade can enhance antitumor immune responses in mice. In humans, blockade of the related programmed cell death 1 (PD-1) pathway has shown great potential in clinical immunotherapy trials. Here, we report the structure of human VISTA and examine its function in lymphocyte negative regulation in cancer. VISTA is expressed predominantly within the hematopoietic compartment with highest expression within the myeloid lineage. VISTA-Ig suppressed proliferation of T cells but not B cells and blunted the production of T-cell cytokines and activation markers. Our results establish VISTA as a negative checkpoint regulator that suppresses T-cell activation, induces Foxp3 expression, and is highly expressed within the tumor microenvironment. By analogy to PD-1 and PD-L1 blockade, VISTA blockade may offer an immunotherapeutic strategy for human cancer.
-
Tzeng A, Diaz-Montero CM, Rayman PA, Kim JS, Pavicic PG, Finke JH, Barata PC, Lamenza M, Devonshire S, Schach K, Emamekhoo H, Ernstoff MS, Hoimes CJ, Rini BI, Garcia JA, Gilligan TD, Ornstein MC, Grivas P (2018). "Immunological Correlates of Response
PubMed
Background: The identification of prognostic and/or predictive biomarkers for response to immune checkpoint inhibitors (ICI) could help guide treatment decisions. Objective: We assessed changes in programmed cell death-1 (PD1)/PD1 ligand (PDL1) expression in key immunomodulatory cell subsets (myeloid-derived suppressor cells [MDSC]; cytotoxic T lymphocytes [CTL]) following ICI therapy and investigated whether these changes correlated with outcomes in patients with metastatic urothelial carcinoma (mUC). Patients and methods: Serial peripheral blood samples were collected from ICI-treated mUC patients. Flow cytometry was used to quantify PD1/PDL1 expression on MDSC (CD33+HLADR-) and CTL (CD8+CD4-) from peripheral blood mononuclear cells. MDSC were grouped into monocytic (M)-MDSC (CD14+CD15-), polymorphonuclear (PMN)-MDSC (CD14-CD15+), and immature (I)-MDSC (CD14-CD15-). Mixed-model regression and Wilcoxon signed-rank or rank-sum tests were performed to assess post-ICI changes in immune biomarker expression and identify correlations between PD1/PDL1 expression and objective response to ICI. Results: Of 41 ICI-treated patients, 26 received anti-PDL1 (23 atezolizumab/3 avelumab) and 15 received anti-PD1 (pembrolizumab) therapy. Based on available data, 27.5% had prior intravesical Bacillus Calmette-Guérin therapy, 42% had prior neoadjuvant chemotherapy, and 70% had prior cystectomy or nephroureterectomy. Successive doses of anti-PDL1 correlated with decreased percentage of PDL1+ (%PDL1+) M-MDSC, while doses of anti-PD1 correlated with decreased %PD1+ M- and I-MDSC. Although pre-treatment %PD1+ CTL did not predict response, a greater %PD1+ CTL within 9 weeks after ICI initiation correlated with objective response. Conclusions: Treatment with ICI correlated with distinct changes in PD1/PDL1-expressing peripheral immune cell subsets, which may predict objective response to ICI. Further studies are required to validate immune molecular expression as a prognostic and/or predictive biomarker for long-term outcomes in mUC.