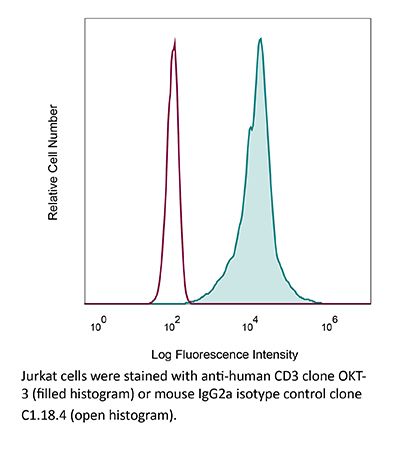
InVivoMAb anti-human CD3
Product Description
Specifications
| Isotype | Mouse IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG2a isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Not available or unknown |
| Reported Applications |
in vitro T cell stimulation/activation in vivo T cell depletion in humanized mice ex vivo T cell inhibition for xenografts Flow cytometry in vitro Organoids/Organ-on-Chip |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107632 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Liu, H., et al (2015). "The Immune Adaptor SLP-76 Binds to SUMO-RANGAP1 at Nuclear Pore Complex Filaments to Regulate Nuclear Import of Transcription Factors in T Cells" Mol Cell 59(5): 840-849.
PubMed
While immune cell adaptors regulate proximal T cell signaling, direct regulation of the nuclear pore complex (NPC) has not been reported. NPC has cytoplasmic filaments composed of RanGAP1 and RanBP2 with the potential to interact with cytoplasmic mediators. Here, we show that the immune cell adaptor SLP-76 binds directly to SUMO-RanGAP1 of cytoplasmic fibrils of the NPC, and that this interaction is needed for optimal NFATc1 and NF-kappaB p65 nuclear entry in T cells. Transmission electron microscopy showed anti-SLP-76 cytoplasmic labeling of the majority of NPCs in anti-CD3 activated T cells. Further, SUMO-RanGAP1 bound to the N-terminal lysine 56 of SLP-76 where the interaction was needed for optimal RanGAP1-NPC localization and GAP exchange activity. While the SLP-76-RanGAP1 (K56E) mutant had no effect on proximal signaling, it impaired NF-ATc1 and p65/RelA nuclear entry and in vivo responses to OVA peptide. Overall, we have identified SLP-76 as a direct regulator of nuclear pore function in T cells.
-
Hill, E. V., et al (2015). "Glycogen synthase kinase-3 controls IL-10 expression in CD4(+) effector T-cell subsets through epigenetic modification of the IL-10 promoter" Eur J Immunol 45(4): 1103-1115.
PubMed
The serine/threonine kinase glycogen synthase kinase-3 (GSK3) plays an important role in balancing pro- and anti-inflammatory cytokines. We have examined the role of GSK3 in production of IL-10 by subsets of CD4(+) T helper cells. Treatment of naive murine CD4(+) T cells with GSK3 inhibitors did not affect their production of IL-10. However, treatment of Th1 and Th2 cells with GSK3 inhibitors dramatically increased production of IL-10. GSK3 inhibition also led to upregulation of IL-10 among Th1, Th2, and Th17 subsets isolated from human blood. The encephalitogenic potential of GSK3 inhibitor treated murine Th1 cells was significantly reduced in adoptive transfer experiments by an IL-10-dependent mechanism. Analysis of the murine IL-10 promoter in response to inhibition of GSK3 in Th1 cells showed modification to a transcriptionally active state indicated by changes in histone H3 acetylation and methylation. Additionally, GSK3 inhibition increased expression of the transcription factors c-Maf, Nfil3, and GATA3, correlating with the increase in IL-10. These findings are important in the context of autoimmune disease since they show that it is possible to reprogram disease-causing cells through GSK3 inhibition.
-
Wunderlich, M., et al (2014). "OKT3 prevents xenogeneic GVHD and allows reliable xenograft initiation from unfractionated human hematopoietic tissues" Blood 123(24): e134-144.
PubMed
Immunodeficient mice are now readily engrafted with human hematopoietic cells. However, these mice are susceptible to graft-versus-host disease (GVHD) induced by the engraftment and rapid expansion of coinjected human T cells. Therefore, highly purified sample populations must be used, adding significant time, expense, and effort. Here, we have explored in vivo and in vitro methods utilizing anti-T-cell antibodies to circumvent this problem. Intraperitoneal injection of the antibody within 48 hours prevented GVHD. Alternatively, short-term in vitro incubation of cells with antibody immediately before transplant was equally effective. Although in vitro antithymocyte globulin treatment resulted in a dramatic loss of SCID-repopulating cells (SRCs), treatment with OKT3 or UCHT1 abrogated GVHD risk and preserved engraftment potential. Leukemia samples that presented with substantial human T-cell contamination were effectively rescued from GVHD. In addition, OKT3 treatment of unfractionated cord blood resulted in robust engraftment of primary and secondary mice that was indistinguishable from grafts obtained using purified CD34(+) cells. Limiting dilution analysis of unfractionated blood demonstrated a SRC frequency of 1 in 300 to 500 CD34(+) cells, similar to that of purified hematopoietic stem and progenitor cells. This protocol streamlines xenograft studies while significantly reducing the cost and time of the procedure.
-
Rochman, Y., et al (2015). "Functional characterization of human T cell hyporesponsiveness induced by CTLA4-Ig" PLoS One 10(4): e0122198.
PubMed
During activation, T cells integrate multiple signals from APCs and cytokine milieu. The blockade of these signals can have clinical benefits as exemplified by CTLA4-Ig, which blocks interaction of B7 co-stimulatory molecules on APCs with CD28 on T cells. Variants of CTLA4-Ig, abatacept and belatacept are FDA approved as immunosuppressive agents in arthritis and transplantation, yet murine studies suggested that CTLA4-Ig could be beneficial in a number of other diseases. However, detailed analysis of human CD4 cell hyporesponsivness induced by CTLA4-Ig has not been performed. Herein, we established a model to study the effect of CTLA4-Ig on the activation of human naive T cells in a human mixed lymphocytes system. Comparison of human CD4 cells activated in the presence or absence of CTLA4-Ig showed that co-stimulation blockade during TCR activation does not affect NFAT signaling but results in decreased activation of NF-kappaB and AP-1 transcription factors followed by a profound decrease in proliferation and cytokine production. The resulting T cells become hyporesponsive to secondary activation and, although capable of receiving TCR signals, fail to proliferate or produce cytokines, demonstrating properties of anergic cells. However, unlike some models of T cell anergy, these cells did not possess increased levels of the TCR signaling inhibitor CBLB. Rather, the CTLA4-Ig-induced hyporesponsiveness was associated with an elevated level of p27kip1 cyclin-dependent kinase inhibitor.
Product Citations
-
Targeting KIF20A blocks lactylation modification to suppress immune escape in hepatocellular carcinoma.
In iScience on 17 April 2026 by Chen, S., Zhao, L., et al.
PubMed
Hepatocellular carcinoma (HCC) evades anti-PD-1 immunotherapy via an immunosuppressive microenvironment, where lactate links metabolic reprogramming to epigenetic regulation. We analyzed pan-lysine lactylation and H3K18 lactylation (H3K18la) in 89 HCC patient pairs, and validated functional mechanisms using glycolysis inhibition, HCC-CD8+ T cell co-cultures, and rescue assays. In vivo efficacy was assessed in subcutaneous and orthotopic HCC mouse models. H3K18la levels were elevated in HCC, correlating with advanced staging and poor prognosis. Lactate induced H3K18la to transcriptionally upregulate KIF20A, which stabilized the c-Myc/PD-L1 axis and suppressed cytotoxic T cell function. Combined glycolysis inhibition and anti-PD-1 therapy reversed this immunosuppression and synergistically inhibited tumor growth. This study identifies an H3K18la-KIF20A/PD-L1 axis as a key metabolic-epigenetic checkpoint, highlighting glycolysis targeting as a promising strategy to enhance anti-PD-1 responses in HCC.
-
Inflammatory arthritis irAE may represent a unique autoimmune disease primarily driven by T cells but likely not autoantibodies.
In Sci Adv on 3 April 2026 by Zhu, X., Yu, Y., et al.
PubMed
The underlying immunopathogenesis of inflammatory arthritis (IA) immune-related adverse event (irAE) remains obscure. Unlike rheumatoid arthritis (RA), where autoantibodies and B cell dysfunction are central features, the contribution of humoral immunity to IA-irAE is unclear. Here, we performed immunophenotyping of peripheral blood from patients with IA-irAE and compared them with patients with seronegative RA, immune checkpoint inhibition-treated patients without irAE, and healthy controls. IA-irAE was marked with increased cytotoxic gene expression and metabolic activation in T cells and reduced CXCR3 and CCR6 expression in CD4+ T cells. Contrary to seronegative RA, patients with IA-irAE displayed no substantial elevation in autoantibody levels or atypical CD11c+CD21- B cells. IA-irAE was further characterized by elevated levels of interleukin-6 (IL-6), IL-12, and type I interferon, which correlated with the T cell activation phenotypes. Together, our findings define IA-irAE as a disease with certain immunological features distinctive from RA, representing a potentially T cell-driven, autoantibody-independent autoimmunity. These results offer insights into immune tolerance breakdown and therapeutic targeting in irAEs.
-
Pharmacological blockade of rho kinase enhances venetoclax responses in translational models of acute myeloid leukemia.
In Haematologica on 2 April 2026 by Golla, U., Bhalodia, R., et al.
PubMed
Acute Myeloid Leukemia (AML) is an aggressive hematologic malignancy requiring concomitant targeting of critical cellular survival pathways due to resistance and frequent relapse with monotherapies. Venetoclax (VEN), a BCL-2 inhibitor, is one such promising clinical agent best utilized in combination therapies due to transient responses and acquired resistance. Given the involvement of the Rho/ROCK pathway in VEN activity, we combined Rho-associated coiled-coil-containing protein kinase inhibitors (ROCKi))with VEN to achieve superior antileukemic activity. The ROCKi (Fasudil, DJ4, GSK269962A) synergized with VEN to enhance cytotoxicity in both VEN-sensitive and VEN-resistant cell lines in vitro. Among the three ROCKi, GSK269962A (GSK) was best-tolerated in combination with VEN and effectively inhibited leukemia growth across multiple AML cell line-derived xenograft models in vivo. The GSK+VEN combination exhibited additive to synergistic cytotoxicity in primary AML patient cells ex vivo and enhanced antileukemic activity in a patientderived xenograft model. Additionally, the GSK+VEN combination significantly decreased the clonogenicity of primary AML cells, relatively sparing normal cells. Functional assays demonstrated enhanced apoptosis (Annexin V, caspase-3/7), elevated reactive oxygen species, and mitochondrial depolarization in both VENsensitive and VEN-resistant AML cells following combination treatment. Mechanistically, GSK augmented venetoclax responses by downregulating anti-apoptotic proteins (BCL2, MCL1) and inducing pro-apoptotic mediators (NOXA, MCL1 short isoforms), including in VEN-resistant AML cells. Together, these findings across multiple preclinical AML models demonstrate synergistic antileukemic activity and support combining VEN with ROCKi as a promising therapeutic strategy for AML.
-
Facile induction of immune tolerance by an interleukin-2-TGFβ surrogate agonist.
In Nature on 11 March 2026 by Sun, Q., Barrett, A. K., et al.
PubMed
CD4+ regulatory T cells (Treg cells) are essential for immune tolerance1. Peripherally induced Treg cells (pTreg cells) complement thymic Treg cells by broadening Treg cell reactivity in response to a changing antigenic landscape2. Although both TGFβ and IL-2 synergistically promote functional pTreg cell development in vitro3-6, their combined roles in inducing pTreg cell generation in vivo have not been exploited for tolerizing immunotherapy. Here we designed an IL-2-TGFβ 'surrogate' co-agonist by creating a single-chain fusion protein between IL-2 and a low-affinity TGFβ mimic agonist derived from a helminth parasite7. This IL-2-TGFβ surrogate functions as an AND-gated co-agonist and enabled simultaneous cis-activation of IL-2-STAT5 and TGFβ-SMAD2/3 signalling specifically in T cells that express IL-2 receptors. The IL-2-TGFβ surrogate agonist robustly induced antigen-specific, functional and stable pTreg cells in vivo within peripheral lymphoid organs in mice immunized with ovalbumin (OVA) and myelin oligodendrocyte glycoprotein (MOG)35-55. The induced pTreg cells display an effector-like, actively expanding state with high RORγt expression, enabling efficient migration and suppression of intestinal inflammation. Treatment with this agonist effectively quelled immune activation in mouse models of allergen-induced allergic inflammation and self-antigen-driven autoimmune neuroinflammation, suggesting a strategy for the induction of antigen-specific pTreg cells in vivo to establish immune tolerance in inflammatory, allergic and autoimmune diseases.