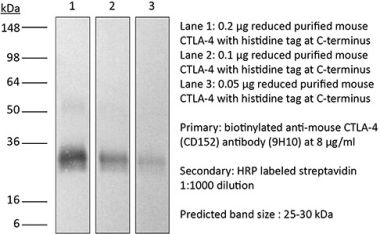
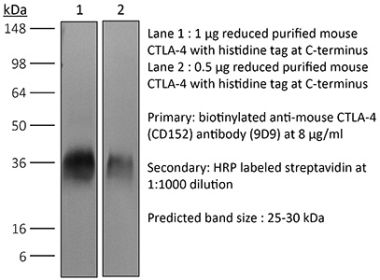
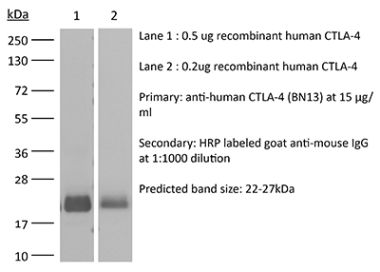
InVivoMAb anti-mouse CTLA-4 (CD152)
Product Details
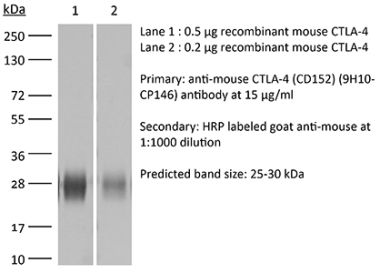
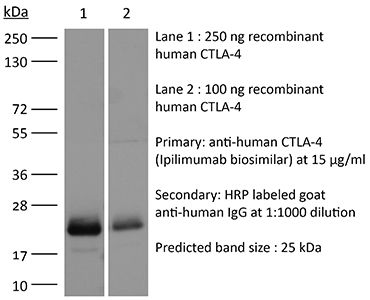
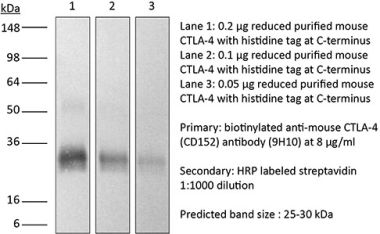
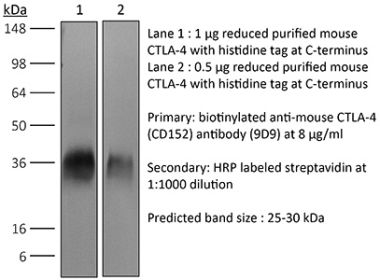
The UC10-4F10-11 monoclonal antibody reacts with mouse CTLA-4 (cytotoxic T lymphocyte antigen-4) also known as CD152. CTLA-4 is a 33 kDa cell surface receptor encoded by the Ctla4 gene that belongs to the CD28 family of the Ig superfamily. CTLA-4 is expressed on activated T and B lymphocytes. CTLA-4 is structurally similar to the T-cell co-stimulatory protein, CD28, and both molecules bind to the B7 family members B7-1 (CD80) and B7-2 (CD86). Upon ligand binding, CTLA-4 negatively regulates cell-mediated immune responses. CTLA-4 plays roles in induction and/or maintenance of immunological tolerance, thymocyte development, and regulation of protective immunity. The critical role of CTLA-4 in immune down-regulation has been demonstrated in CTLA-4 deficient mice, which succumb at 3-5 weeks of age due to the development of a lymphoproliferative disease. CTLA-4 is among a group of inhibitory receptors being explored as cancer treatment targets through immune checkpoint blockade. The UC10-4F10-11 antibody has been shown to promote T cell co-stimulation by blocking CTLA-4 binding to the B7 co-receptors, allowing for CD28 binding.Specifications
| Isotype | Armenian hamster IgG |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb polyclonal Armenian hamster IgG |
| Recommended Dilution Buffer | InVivoPure pH 6.5 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse CTLA-4 IgG2a fusion protein |
| Reported Applications |
in vivo CTLA-4 neutralization in vitro CTLA-4 neutralization Flow cytometry Western blot |
| Formulation |
PBS, pH 6.5 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107598 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Additional Formats
Recommended Products
in vivo CTLA-4 neutralization
Triplett, T. A., et al. (2018). "Reversal of indoleamine 2,3-dioxygenase-mediated cancer immune suppression by systemic kynurenine depletion with a therapeutic enzyme" Nat Biotechnol 36(8): 758-764. PubMed
Increased tryptophan (Trp) catabolism in the tumor microenvironment (TME) can mediate immune suppression by upregulation of interferon (IFN)-gamma-inducible indoleamine 2,3-dioxygenase (IDO1) and/or ectopic expression of the predominantly liver-restricted enzyme tryptophan 2,3-dioxygenase (TDO). Whether these effects are due to Trp depletion in the TME or mediated by the accumulation of the IDO1 and/or TDO (hereafter referred to as IDO1/TDO) product kynurenine (Kyn) remains controversial. Here we show that administration of a pharmacologically optimized enzyme (PEGylated kynureninase; hereafter referred to as PEG-KYNase) that degrades Kyn into immunologically inert, nontoxic and readily cleared metabolites inhibits tumor growth. Enzyme treatment was associated with a marked increase in the tumor infiltration and proliferation of polyfunctional CD8(+) lymphocytes. We show that PEG-KYNase administration had substantial therapeutic effects when combined with approved checkpoint inhibitors or with a cancer vaccine for the treatment of large B16-F10 melanoma, 4T1 breast carcinoma or CT26 colon carcinoma tumors. PEG-KYNase mediated prolonged depletion of Kyn in the TME and reversed the modulatory effects of IDO1/TDO upregulation in the TME.
in vivo CTLA-4 neutralization, Flow Cytometry
Pletinckx, K., et al. (2015). "Immature dendritic cells convert anergic nonregulatory T cells into Foxp3- IL-10+ regulatory T cells by engaging CD28 and CTLA-4" Eur J Immunol 45(2): 480-491. PubMed
Anergic T cells can survive for long time periods passively in a hyporesponsive state without obvious active functions. Thus, the immunological reason for their maintenance is unclear. Here, we induced peptide-specific anergy in T cells from mice by coculturing these cells with immature murine dendritic cells (DCs). We found that these anergic, nonsuppressive IL-10(-) Foxp3(-) CTLA-4(+) CD25(low) Egr2(+) T cells could be converted into suppressive IL-10(+) Foxp3(-) CTLA-4(+) CD25(high) Egr2(+) cells resembling type-1 Treg cells (Tr1) when stimulated a second time by immature DCs in vitro. Addition of TGF-beta during anergy induction favored Foxp3(+) Treg-cell induction, while TGF-beta had little effect when added to the second stimulation. Expression of both CD28 and CTLA-4 molecules on anergic T cells was required to allow their conversion into Tr1-like cells. Suppressor activity was enabled via CD28-mediated CD25 upregulation, acting as an IL-2 sink, together with a CTLA-4-mediated inhibition of NFATc1/alpha activation to shut down IL-2-mediated proliferation. Together, these data provide evidence and mechanistical insights into how persistent anergic T cells may serve as a resting memory pool for Tr1-like cells.
in vivo CTLA-4 neutralization
Welten, S. P., et al. (2015). "The viral context instructs the redundancy of costimulatory pathways in driving CD8(+) T cell expansion" Elife 4. doi : 10.7554/eLife.07486. PubMed
Signals delivered by costimulatory molecules are implicated in driving T cell expansion. The requirements for these signals, however, vary from dispensable to essential in different infections. We examined the underlying mechanisms of this differential T cell costimulation dependence and found that the viral context determined the dependence on CD28/B7-mediated costimulation for expansion of naive and memory CD8(+) T cells, indicating that the requirement for costimulatory signals is not imprinted. Notably, related to the high-level costimulatory molecule expression induced by lymphocytic choriomeningitis virus (LCMV), CD28/B7-mediated costimulation was dispensable for accumulation of LCMV-specific CD8(+) T cells because of redundancy with the costimulatory pathways induced by TNF receptor family members (i.e., CD27, OX40, and 4-1BB). Type I IFN signaling in viral-specific CD8(+) T cells is slightly redundant with costimulatory signals. These results highlight that pathogen-specific conditions differentially and uniquely dictate the utilization of costimulatory pathways allowing shaping of effector and memory antigen-specific CD8(+) T cell responses.
in vivo CTLA-4 neutralization
Makkouk, A., et al. (2015). "Three steps to breaking immune tolerance to lymphoma: a microparticle approach" Cancer Immunol Res 3(4): 389-398. PubMed
In situ immunization aims at generating antitumor immune responses through manipulating the tumor microenvironment. On the basis of recent advances in the understanding of antitumor immunity, we designed a three-step approach to in situ immunization to lymphoma: (i) inducing immunogenic tumor cell death with the chemotherapeutic drug doxorubicin. Doxorubicin enhances the expression of “eat-me” signals by dying tumor cells, facilitating their phagocytosis by dendritic cells (DC). Because of the vesicant activity of doxorubicin, microparticles made of biodegradable polymer poly(lactide-co-glycolide) or PLGA can safely deliver doxorubicin intratumorally and are effective vaccine adjuvants, (ii) enhancing T-cell activation using anti-OX40 and (iii) sustaining T-cell responses by checkpoint blockade using anti-CTLA-4. In vitro, doxorubicin microparticles were less cytotoxic to DCs than to B lymphoma cells, did not require internalization by tumor cells, and significantly enhanced phagocytosis of tumor cells by DCs as compared with soluble doxorubicin. In mice, this three-step therapy induced CD4- and CD8-dependent systemic immune responses that enhanced T-cell infiltration into distant tumors, leading to their eradication and significantly improving survival. Our findings demonstrate that systemic antitumor immune responses can be generated locally by three-step therapy and merit further investigation as an immunotherapy for patients with lymphoma.
in vivo CTLA-4 neutralization
Mittal, D., et al. (2014). "Antimetastatic effects of blocking PD-1 and the adenosine A2A receptor" Cancer Res 74(14): 3652-3658. PubMed
Adenosine targeting is an attractive new approach to cancer treatment, but no clinical study has yet examined adenosine inhibition in oncology despite the safe clinical profile of adenosine A2A receptor inhibitors (A2ARi) in Parkinson disease. Metastasis is the main cause of cancer-related deaths worldwide, and therefore we have studied experimental and spontaneous mouse models of melanoma and breast cancer metastasis to demonstrate the efficacy and mechanism of a combination of A2ARi in combination with anti-PD-1 monoclonal antibody (mAb). This combination significantly reduces metastatic burden and prolongs the life of mice compared with either monotherapy alone. Importantly, the combination was only effective when the tumor expressed high levels of CD73, suggesting a tumor biomarker that at a minimum could be used to stratify patients that might receive this combination. The mechanism of the combination therapy was critically dependent on NK cells and IFNgamma, and to a lesser extent, CD8(+) T cells and the effector molecule, perforin. Overall, these results provide a strong rationale to use A2ARi with anti-PD-1 mAb for the treatment of minimal residual and metastatic disease.
in vivo CTLA-4 neutralization
Zhu, Y., et al. (2014). "CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models" Cancer Res 74(18): 5057-5069. PubMed
Cancer immunotherapy generally offers limited clinical benefit without coordinated strategies to mitigate the immunosuppressive nature of the tumor microenvironment. Critical drivers of immune escape in the tumor microenvironment include tumor-associated macrophages and myeloid-derived suppressor cells, which not only mediate immune suppression, but also promote metastatic dissemination and impart resistance to cytotoxic therapies. Thus, strategies to ablate the effects of these myeloid cell populations may offer great therapeutic potential. In this report, we demonstrate in a mouse model of pancreatic ductal adenocarcinoma (PDAC) that inhibiting signaling by the myeloid growth factor receptor CSF1R can functionally reprogram macrophage responses that enhance antigen presentation and productive antitumor T-cell responses. Investigations of this response revealed that CSF1R blockade also upregulated T-cell checkpoint molecules, including PDL1 and CTLA4, thereby restraining beneficial therapeutic effects. We found that PD1 and CTLA4 antagonists showed limited efficacy as single agents to restrain PDAC growth, but that combining these agents with CSF1R blockade potently elicited tumor regressions, even in larger established tumors. Taken together, our findings provide a rationale to reprogram immunosuppressive myeloid cell populations in the tumor microenvironment under conditions that can significantly empower the therapeutic effects of checkpoint-based immunotherapeutics.
in vivo CTLA-4 neutralization
Rabenstein, H., et al. (2014). "Differential kinetics of antigen dependency of CD4+ and CD8+ T cells" J Immunol 192(8): 3507-3517. PubMed
Ag recognition via the TCR is necessary for the expansion of specific T cells that then contribute to adaptive immunity as effector and memory cells. Because CD4+ and CD8+ T cells differ in terms of their priming APCs and MHC ligands we compared their requirements of Ag persistence during their expansion phase side by side. Proliferation and effector differentiation of TCR transgenic and polyclonal mouse T cells were thus analyzed after transient and continuous TCR signals. Following equally strong stimulation, CD4+ T cell proliferation depended on prolonged Ag presence, whereas CD8+ T cells were able to divide and differentiate into effector cells despite discontinued Ag presentation. CD4+ T cell proliferation was neither affected by Th lineage or memory differentiation nor blocked by coinhibitory signals or missing inflammatory stimuli. Continued CD8+ T cell proliferation was truly independent of self-peptide/MHC-derived signals. The subset divergence was also illustrated by surprisingly broad transcriptional differences supporting a stronger propensity of CD8+ T cells to programmed expansion. These T cell data indicate an intrinsic difference between CD4+ and CD8+ T cells regarding the processing of TCR signals for proliferation. We also found that the presentation of a MHC class II-restricted peptide is more efficiently prolonged by dendritic cell activation in vivo than a class I bound one. In summary, our data demonstrate that CD4+ T cells require continuous stimulation for clonal expansion, whereas CD8+ T cells can divide following a much shorter TCR signal.
in vivo CTLA-4 neutralization, Flow Cytometry
Honda, T., et al. (2014). "Tuning of antigen sensitivity by T cell receptor-dependent negative feedback controls T cell effector function in inflamed tissues" Immunity 40(2): 235-247. PubMed
Activated T cells must mediate effector responses sufficiently to clear pathogens while avoiding excessive tissue damage. Here we have combined dynamic intravital microscopy with ex vivo assessments of T cell cytokine responses to generate a detailed spatiotemporal picture of CD4(+) T cell effector regulation in the skin. In response to antigen, effector T cells arrested transiently on antigen-presenting cells, briefly producing cytokine and then resuming migration. Antigen recognition led to upregulation of the programmed death-1 (PD-1) glycoprotein by T cells and blocking its canonical ligand, programmed death-ligand 1 (PD-L1), lengthened the duration of migration arrest and cytokine production, showing that PD-1 interaction with PD-L1 is a major negative feedback regulator of antigen responsiveness. We speculate that the immune system employs T cell recruitment, transient activation, and rapid desensitization to allow the T cell response to rapidly adjust to changes in antigen presentation and minimize collateral injury to the host.
in vivo CTLA-4 neutralization
Allard, B., et al. (2013). "Targeting CD73 enhances the antitumor activity of anti-PD-1 and anti-CTLA-4 mAbs" Clin Cancer Res 19(20): 5626-5635. PubMed
PURPOSE: Monoclonal antibodies (mAb) that block programmed death (PD)-1 or cytotoxic T lymphocyte antigen (CTLA-4) receptors have been associated with durable clinical responses against a variety of cancer types and hold great potential as novel cancer therapeutics. Recent evidence suggest that targeted blockade of multiple immunosuppressive pathways can induce synergistic antitumor responses. EXPERIMENTAL DESIGN: In this study, we investigated whether targeted blockade of CD73, an ectonucleotidase that catabolizes the hydrolysis of extracellular adenosine monophosphate (AMP) to adenosine, can enhance the antitumor activity of anti-CTLA-4 and anti-PD-1 mAbs against transplanted and chemically induced mouse tumors. RESULTS: Anti-CD73 mAb significantly enhanced the activity of both anti-CTLA-4 and anti-PD-1 mAbs against MC38-OVA (colon) and RM-1 (prostate) subcutaneous tumors, and established metastatic 4T1.2 breast cancer. Anti-CD73 mAb also significantly enhanced the activity of anti-PD-1 mAb against 3-methylcholanthrene (MCA)-induced fibrosarcomas. Gene-targeted mice revealed that single-agent therapies and combinatorial treatments were dependent on host IFN-gamma and CD8(+) T cells, but independent of perforin. Interestingly, anti-CD73 mAb preferentially synergized with anti-PD-1 mAb. We investigated the effect of extracellular adenosine on tumor-infiltrating T cells and showed that activation of A2A adenosine receptor enhances PD-1 expression, but not CTLA-4 expression, on tumor-specific CD8+ T cells and CD4+ Foxp3+ T regulatory cells. CONCLUSIONS: Taken together, our study revealed that targeted blockade of CD73 can enhance the therapeutic activity of anti-PD-1 and anti-CTLA-4 mAbs and may thus potentiate therapeutic strategies targeting immune checkpoint inhibitors in general.
in vivo CTLA-4 neutralization
Hafalla, J. C., et al. (2012). "The CTLA-4 and PD-1/PD-L1 inhibitory pathways independently regulate host resistance to Plasmodium-induced acute immune pathology" PLoS Pathog 8(2): e1002504. PubMed
The balance between pro-inflammatory and regulatory immune responses in determining optimal T cell activation is vital for the successful resolution of microbial infections. This balance is maintained in part by the negative regulators of T cell activation, CTLA-4 and PD-1/PD-L, which dampen effector responses during chronic infections. However, their role in acute infections, such as malaria, remains less clear. In this study, we determined the contribution of CTLA-4 and PD-1/PD-L to the regulation of T cell responses during Plasmodium berghei ANKA (PbA)-induced experimental cerebral malaria (ECM) in susceptible (C57BL/6) and resistant (BALB/c) mice. We found that the expression of CTLA-4 and PD-1 on T cells correlates with the extent of pro-inflammatory responses induced during PbA infection, being higher in C57BL/6 than in BALB/c mice. Thus, ECM develops despite high levels of expression of these inhibitory receptors. However, antibody-mediated blockade of either the CTLA-4 or PD-1/PD-L1, but not the PD-1/PD-L2, pathways during PbA-infection in ECM-resistant BALB/c mice resulted in higher levels of T cell activation, enhanced IFN-gamma production, increased intravascular arrest of both parasitised erythrocytes and CD8(+) T cells to the brain, and augmented incidence of ECM. Thus, in ECM-resistant BALB/c mice, CTLA-4 and PD-1/PD-L1 represent essential, independent and non-redundant pathways for maintaining T cell homeostasis during a virulent malaria infection. Moreover, neutralisation of IFN-gamma or depletion of CD8(+) T cells during PbA infection was shown to reverse the pathologic effects of regulatory pathway blockade, highlighting that the aetiology of ECM in the BALB/c mice is similar to that in C57BL/6 mice. In summary, our results underscore the differential and complex regulation that governs immune responses to malaria parasites.
in vivo CTLA-4 neutralization
Haque, A., et al. (2010). "CD4+ natural regulatory T cells prevent experimental cerebral malaria via CTLA-4 when expanded in vivo" PLoS Pathog 6(12): e1001221. PubMed
Studies in malaria patients indicate that higher frequencies of peripheral blood CD4(+) Foxp3(+) CD25(+) regulatory T (Treg) cells correlate with increased blood parasitemia. This observation implies that Treg cells impair pathogen clearance and thus may be detrimental to the host during infection. In C57BL/6 mice infected with Plasmodium berghei ANKA, depletion of Foxp3(+) cells did not improve parasite control or disease outcome. In contrast, elevating frequencies of natural Treg cells in vivo using IL-2/anti-IL-2 complexes resulted in complete protection against severe disease. This protection was entirely dependent upon Foxp3(+) cells and resulted in lower parasite biomass, impaired antigen-specific CD4(+) T and CD8(+) T cell responses that would normally promote parasite tissue sequestration in this model, and reduced recruitment of conventional T cells to the brain. Furthermore, Foxp3(+) cell-mediated protection was dependent upon CTLA-4 but not IL-10. These data show that T cell-mediated parasite tissue sequestration can be reduced by regulatory T cells in a mouse model of malaria, thereby limiting malaria-induced immune pathology.
in vivo CTLA-4 neutralization
Rowe, J. H., et al. (2009). "Cytotoxic T-lymphocyte antigen 4 blockade augments the T-cell response primed by attenuated Listeria monocytogenes resulting in more rapid clearance of virulent bacterial challenge" Immunology 128(1 Suppl): e471-478. PubMed
Cytotoxic T-lymphocyte antigen 4 (CTLA-4) uniformly suppresses antigen-specific T cells during chronic infection with bacterial, parasitic or viral pathogens. However, the importance of CTLA-4 in controlling the T-cell response during acute infection or after priming with live attenuated vaccine vectors has not been well characterized. Since strategies aimed at blocking CTLA-4 are being actively developed to therapeutically augment T-cell-mediated immunity, the effects of CTLA-4 blockade on T-cell activation during these conditions need to be more clearly defined. We have examined the role of CTLA-4 in a prime-challenge model of acute bacterial infection using both attenuated and virulent strains of the intracellular bacterium Listeria monocytogenes. Although Foxp3(+) CD4(+) T cells are the predominant CTLA-4-expressing cell type in naive mice, antigen-specific Foxp3(-) CD4(+) cells upregulate CTLA-4 expression after primary L. monocytogenes infection. Blockade of CTLA-4 results in increased numbers of L. monocytogenes-specific CD4 and CD8 T cells after primary infection with attenuated L. monocytogenes, and confers more rapid bacterial clearance after secondary challenge with virulent L. monocytogenes. Accordingly, CTLA-4 plays an important suppressive role in T-cell priming and protective immunity in a prime-challenge model of acute bacterial infection.
in vivo CTLA-4 neutralization, in vitro CTLA-4 neutralization
Noval Rivas, M., et al. (2009). "Reviving function in CD4+ T cells adapted to persistent systemic antigen" J Immunol 183(7): 4284-4291. PubMed
In bone marrow-transplanted patients, chronic graft-versus-host disease is a complication that results from the persistent stimulation of recipient minor histocompatibility Ag (mHA)-specific T cells contained within the graft. In this study, we developed a mouse model where persistent stimulation of donor T cells by recipient’s mHA led to multiorgan T cell infiltration. Exposure to systemic mHA, however, deeply modified T cell function and chronically stimulated T cells developed a long-lasting state of unresponsiveness, or immune adaptation, characterized by their inability to mediate organ immune damages in vivo. However, analysis of the gene expression profile of adapted CD4+ T cells revealed the specific coexpression of genes known to promote differentiation and function of Th1 effector cells as well as genes coding for proteins that control T cell activity, such as cell surface-negative costimulatory molecules and regulatory cytokines. Strikingly, blockade of negative costimulation abolished T cell adaptation and stimulated strong IFN-gamma production and severe multiorgan wasting disease. Negative costimulation was also shown to control lethal LPS-induced toxic shock in mice with adapted T cells, as well as the capacity of adapted T cells to reject skin graft. Our results demonstrate that negative costimulation is the molecular mechanism used by CD4+ T cells to adapt their activity in response to persistent antigenic stimulation. The effector function of CD4+ T cells that have adapted to chronic Ag presentation can be activated by stimuli strong enough to overcome regulatory signals delivered to the T cells by negative costimulation.
- Mus musculus (House mouse),
- Cancer Research
INHBA/Activin A promotes tumor growth and induces resistance to anti-PD-L1 therapy by suppressing IFN-γ signaling
Preprint on BioRxiv : the Preprint Server for Biology on 8 December 2023 by Li, F., Gu, L., et al.
PubMed
Inhibin beta A (INHBA) and its homodimer activin A have pleiotropic effects on modulation of immune responses and tumor progression, respectively, but it remains uncertain whether tumors may release activin A to regulate anti-tumor immunity. As evidenced by our RNA-Seq and in vitro results, the interferon-γ (IFN-γ) signaling pathway was significantly down-regulated by tumor intrinsic activin A. Tumor INHBA deficiency led to lower expression of PD-L1 induced by IFN-γ, resulting in poor responsiveness to anti-PD-L1 therapy. On the other hand, decreased secretion of IFN-γ-stimulated chemokines, including C-X-C motif chemokine 9 (CXCL9) and 10 (CXCL10), impaired the infiltration of effector T cells into the tumor microenvironment. Furthermore, the activin A-specific antibody garetosmab improved anti-tumor immunity and its combination with the anti-PD-L1 antibody atezolizumab showed a superior therapeutic effect to monotherapy. Our findings reveal that INHBA/activin A is involved in anti-tumor immunity by inhibiting the IFN-γ signaling pathway and considered to be a potential target to overcome anti-PD-L1 resistance in clinical cancer treatment.
- Cancer Research,
- Immunology and Microbiology
Interactions of Indoleamine 2,3-dioxygenase-expressing LAMP3+ dendritic cells with CD4+ regulatory T cells and CD8+ exhausted T cells: synergistically remodeling of the immunosuppressive microenvironment in cervical cancer and therapeutic implication
In Cancer Communications (London, England) on 1 November 2023 by Qu, X., Wang, Y., et al.
PubMed
Cervical cancer (CC) is the fourth most common cancer in women worldwide. Although immunotherapy has been applied in clinical practice, its therapeutic efficacy remains far from satisfactory, necessitating further investigation of the mechanism of CC immune remodeling and exploration of novel treatment targets. This study aimed to investigate the mechanism of CC immune remodeling and explore potential therapeutic targets. We conducted single-cell RNA sequencing on a total of 17 clinical specimens, including normal cervical tissues, high-grade squamous intraepithelial lesions, and CC tissues. To validate our findings, we conducted multicolor immunohistochemical staining of CC tissues and constructed a subcutaneous tumorigenesis model in C57BL/6 mice using murine CC cell lines (TC1) to evaluate the effectiveness of combination therapy involving indoleamine 2,3-dioxygenase 1 (IDO1) inhibition and immune checkpoint blockade (ICB). We used the unpaired two-tailed Student's t-test, Mann-Whitney test, or Kruskal-Wallis test to compare continuous data between two groups and one-way ANOVA with Tukey's post hoc test to compare data between multiple groups. Malignant cervical epithelial cells did not manifest noticeable signs of tumor escape, whereas lysosomal-associated membrane protein 3-positive (LAMP3+ ) dendritic cells (DCs) in a mature state with immunoregulatory roles were found to express IDO1 and affect tryptophan metabolism. These cells interacted with both tumor-reactive exhausted CD8+ T cells and CD4+ regulatory T cells, synergistically forming a vicious immunosuppressive cycle and mediating CC immune escape. Further validation through multicolor immunohistochemical staining showed co-localization of neoantigen-reactive T cells (CD3+ , CD4+ /CD8+ , and PD-1+ ) and LAMP3+ DCs (CD80+ and PD-L1+ ). Additionally, a combination of the IDO1 inhibitor with an ICB agent significantly reduced tumor volume in the mouse model of CC compared with an ICB agent alone. Our study suggested that a combination treatment consisting of targeting IDO1 and ICB agent could improve the therapeutic efficacy of current CC immunotherapies. Additionally, our results provided crucial insights for designing drugs and conducting future clinical trials for CC. © 2023 The Authors. Cancer Communications published by John Wiley & Sons Australia, Ltd on behalf of SUN YAT-SEN UNIVERSITY CANCER CENTER.
- Cancer Research,
- Immunology and Microbiology
Combination IFNβ and Membrane-Stable CD40L Maximize Tumor Dendritic Cell Activation and Lymph Node Trafficking to Elicit Systemic T-cell Immunity.
In Cancer Immunology Research on 3 April 2023 by Zheng, H., Yu, X., et al.
PubMed
Oncolytic virus therapies induce the direct killing of tumor cells and activation of conventional dendritic cells (cDC); however, cDC activation has not been optimized with current therapies. We evaluated the adenoviral delivery of engineered membrane-stable CD40L (MEM40) and IFNβ to locally activate cDCs in mouse tumor models. Combined tumor MEM40 and IFNβ expression induced the highest cDC activation coupled with increased lymph node migration, increased systemic antitumor CD8+ T-cell responses, and regression of established tumors in a cDC1-dependent manner. MEM40 + IFNβ combined with checkpoint inhibitors led to effective control of distant tumors and lung metastases. An oncolytic adenovirus (MEM-288) expressing MEM40 + IFNβ in phase I clinical testing induced cancer cell loss concomitant with enhanced T-cell infiltration and increased systemic presence of tumor T-cell clonotypes in non-small cell lung cancer (NSCLC) patients. This approach to simultaneously target two major DC-activating pathways has the potential to significantly affect the solid tumor immunotherapy landscape. ©2023 American Association for Cancer Research.
- Immunology and Microbiology
Phosphoinositide acyl chain saturation drives CD8+ effector T cell signaling and function.
In Nature Immunology on 1 March 2023 by Edwards-Hicks, J., Apostolova, P., et al.
PubMed
How lipidome changes support CD8+ effector T (Teff) cell differentiation is not well understood. Here we show that, although naive T cells are rich in polyunsaturated phosphoinositides (PIPn with 3-4 double bonds), Teff cells have unique PIPn marked by saturated fatty acyl chains (0-2 double bonds). PIPn are precursors for second messengers. Polyunsaturated phosphatidylinositol bisphosphate (PIP2) exclusively supported signaling immediately upon T cell antigen receptor activation. In late Teff cells, activity of phospholipase C-γ1, the enzyme that cleaves PIP2 into downstream mediators, waned, and saturated PIPn became essential for sustained signaling. Saturated PIP was more rapidly converted to PIP2 with subsequent recruitment of phospholipase C-γ1, and loss of saturated PIPn impaired Teff cell fitness and function, even in cells with abundant polyunsaturated PIPn. Glucose was the substrate for de novo PIPn synthesis, and was rapidly utilized for saturated PIP2 generation. Thus, separate PIPn pools with distinct acyl chain compositions and metabolic dependencies drive important signaling events to initiate and then sustain effector function during CD8+ T cell differentiation. © 2023. The Author(s), under exclusive licence to Springer Nature America, Inc.
- Cancer Research,
- Immunology and Microbiology
Rigosertib promotes anti-tumor immunity via autophagic degradation of PD-L1 in colorectal cancer cells
Preprint on BioRxiv : the Preprint Server for Biology on 8 February 2023 by Zhou, X., Fu, D., et al.
PubMed
Rigosertib (RGS) is a benzyl styryl sulfone which exhibits impressive cytotoxicity in cancer cells. However, its modulating effect on tumor immune microenvironment remains elusive. In our experiments, compared with immunodeficient mouse model, increased tumor growth arrest and robust anti-tumor immunity were observed in RGS-treated colorectal cancer (CRC) xenograft tumors in immunocompetent mice. Intriguingly, RGS markedly down-regulated programmed cell death ligand 1 (PD-L1) expression in both vivo and in vitro. Meanwhile, RGS increased autophagic vacuole number in CRC cells as seen by transmission electron microscopy and immunofluorescence. Moreover, increased LC3-II level and tandem- mRFP- GFP- LC3 labeled vacuole accumulation demonstrated RGS-induced autophagic flux. Mechanistically, it is the activation of AMP-activated protein kinase-UNC-51-like kinase 1 (AMPK-ULK1) axis, rather than the canonical mTOR signaling pathway, that plays a pivotal role in RGS-induced autophagy. AMPK-ULK1 dependent autophagy inhibition, by either short interfering RNA or chemical inhibitors, blocked RGS-induced PD-L1 degradation. Finally, RGS exhibited synergistic anti-tumor activity with cytotoxic T-lymphocyte-associated protein 4 monoclonal antibody in the CRC xenograft model. Furthermore, apart from the immunomodulatory effect, we also confirmed the direct cytotoxicity of RGS in inducing mitochondria-related apoptosis. Altogether, considering its PD-L1 inhibitory and cytotoxic effects, RGS could be a promising drug for CRC therapy.
- Cancer Research,
- Mus musculus (House mouse)
Suppression of Tumor or Host Intrinsic CMTM6 Drives Antitumor Cytotoxicity in a PD-L1-Independent Manner.
In Cancer Immunology Research on 3 February 2023 by Long, Y., Chen, R., et al.
PubMed
CKLF-like MARVEL transmembrane domain-containing protein 6 (CMTM6) is known to be a regulator of membranal programmed death ligand 1 (PD-L1) stability and a factor associated with malignancy progression, but the effects and mechanisms of CMTM6 on tumor growth, as well as its potential as a target for therapy, are still largely unknown. Here, we show that CMTM6 expression increased with tumor progression in both patients and mice. Ablation of CMTM6 significantly reduced human and murine tumor growth in a manner dependent on T-cell immunity. Tumor CMTM6 suppression broke resistance to immune-checkpoint inhibitors and remodeled the tumor immune microenvironment, as specific antitumor cytotoxicity was enhanced and contributed primarily to tumor inhibition. Without the PD-1/PD-L1 axis, CMTM6 suppression still significantly dampened tumor growth dependent on cytotoxic cells. Furthermore, we identified that CMTM6 was widely expressed on immune cells. T-cell CMTM6 levels increased with sustained immune activation and intratumoral immune exhaustion and affected T cell-intrinsic PD-L1 levels. Host CMTM6 knockout significantly restrained tumor growth in a manner dependent on CD8+ T cells and not entirely dependent on PD-L1. Thus, we developed and evaluated the antitumor efficacy of CMTM6-targeting adeno-associated virus (AAV), which effectively mobilized antitumor immunity and could be combined with various antitumor drugs. Our findings reveal that both tumor and host CMTM6 are involved in antitumor immunity with or without the PD-1/PD-L1 axis and that gene therapy targeting CMTM6 is a promising strategy for cancer immunotherapy. ©2022 The Authors; Published by the American Association for Cancer Research.
- Cancer Research,
- Genetics,
- Immunology and Microbiology
DNA barcoding reveals ongoing immunoediting of clonal cancer populations during metastatic progression and immunotherapy response.
In Nature Communications on 7 November 2022 by Baldwin, L. A., Bartonicek, N., et al.
PubMed
Cancers evade the immune system through the process of cancer immunoediting. While immune checkpoint inhibitors are effective for reactivating tumour immunity in some cancer types, many other solid cancers, including breast cancer, remain largely non-responsive. Understanding how non-responsive cancers evade immunity and whether this occurs at the clonal level will improve immunotherapeutic design. Here we use DNA barcoding to track murine mammary cancer cell clones during immunoediting and determine clonal transcriptional profiles that allow immune evasion following anti-PD1 plus anti-CTLA4 immunotherapy. Clonal diversity is significantly restricted by immunotherapy treatment in both primary tumours and metastases, demonstrating selection for pre-existing breast cancer cell populations and ongoing immunoediting during metastasis and treatment. Immunotherapy resistant clones express a common gene signature associated with poor survival of basal-like breast cancer patient cohorts. At least one of these genes has an existing small molecule that can potentially be used to improve immunotherapy response. © 2022. The Author(s).
- Immunology and Microbiology
PD-1 and CTLA-4 exert additive control of effector regulatory T cells
Preprint on BioRxiv : the Preprint Server for Biology on 22 July 2022 by Perry, J. A., Lanzar, Z., et al.
PubMed
At homeostasis, a substantial proportion of Foxp3 + T regulatory cells (T regs ) have an activated phenotype associated with enhanced TCR signals and these effector T reg cells (eT regs ) co-express elevated levels of PD-1 and CTLA-4. Short term in vivo blockade of the PD-1 or CTLA-4 pathways results in increased eT reg populations, while combination blockade of both pathways had an additive effect. Mechanistically, combination blockade resulted in a reduction of suppressive phospho-SHP2 Y580 in T reg cells which was associated with increased eT reg proliferation, enhanced production of IL-10, and altered dendritic cell expression of CD80 and MHC-II. Thus, at homeostasis, PD-1 and CTLA-4 function additively to regulate eT reg function and the ability to target these pathways in T reg cells may be useful to modulate inflammation.
- Cancer Research,
- Immunology and Microbiology,
- Mus musculus (House mouse)
Anti-CTLA-4 and anti-PD-1 immunotherapies repress tumor progression in preclinical breast and colon model with independent regulatory T cells response.
In Translational Oncology on 1 June 2022 by Rupp, T., Genest, L., et al.
PubMed
The recent development of immunotherapy represents a significant breakthrough in cancer therapy. Several immunotherapies provide robust efficacy gains in a wide variety of cancers. However, in some patients the immune checkpoint blockade remains ineffective due to poor therapeutic response and tumor relapse. An improved understanding of the mechanisms underlying tumor-immune system interactions can improve clinical management of cancer. Here, we report preclinical data evaluating two murine antibodies corresponding to recent FDA-approved antibodies for human therapy, e.g. anti-CTLA-4 and anti-PD-1. We demonstrated in two mouse syngeneic grafting models of triple negative breast or colon cancer that the two antibodies displayed an efficient anticancer activity, which is enhanced by combination treatment in the breast cancer model. We also demonstrated that CTLA-4 targeting reduced metastasis formation in the colon cancer metastasis model. In addition, using cytometry-based multiplex analysis, we showed that anti-CTLA-4 and anti-PD-1 affected the tumor immune microenvironment differently and in particular the tumor immune infiltration. This work demonstrated anti-cancer effect of CTLA-4 or PD-1 blockade on mouse colon and triple negative breast and on tumor-infiltrating immune cell subpopulations that could improve our knowledge and benefit the breast and colon cancer tumor research community. Copyright © 2022. Published by Elsevier Inc.
- Cancer Research,
- Immunology and Microbiology
Glucocorticoid receptor regulates PD-L1 and MHC-I in pancreatic cancer cells to promote immune evasion and immunotherapy resistance.
In Nature Communications on 6 December 2021 by Deng, Y., Xia, X., et al.
PubMed
Despite unprecedented responses of some cancers to immune checkpoint blockade (ICB) therapies, the application of checkpoint inhibitors in pancreatic cancer has been unsuccessful. Glucocorticoids and glucocorticoid receptor (GR) signaling are long thought to suppress immunity by acting on immune cells. Here we demonstrate a previously undescribed tumor cell-intrinsic role for GR in activating PD-L1 expression and repressing the major histocompatibility complex class I (MHC-I) expression in pancreatic ductal adenocarcinoma (PDAC) cells through transcriptional regulation. In mouse models of PDAC, either tumor cell-specific depletion or pharmacologic inhibition of GR leads to PD-L1 downregulation and MHC-I upregulation in tumor cells, which in turn promotes the infiltration and activity of cytotoxic T cells, enhances anti-tumor immunity, and overcomes resistance to ICB therapy. In patients with PDAC, GR expression correlates with high PD-L1 expression, low MHC-I expression, and poor survival. Our results reveal GR signaling in cancer cells as a tumor-intrinsic mechanism of immunosuppression and suggest that therapeutic targeting of GR is a promising way to sensitize pancreatic cancer to immunotherapy. © 2021. The Author(s).
- In Vivo,
- Mus musculus (House mouse),
- Cancer Research
Elimination of acquired resistance to PD-1 blockade via the concurrent depletion of tumour cells and immunosuppressive cells.
In Nature Biomedical Engineering on 1 November 2021 by Xue, G., Wang, Z., et al.
PubMed
Antigen release resulting from the death of tumour cells induced by chemotherapies and targeted therapies can augment the antitumour responses induced by immune checkpoint blockade (ICB). However, tumours responding to ICB therapies often become resistant to them. Here we show that the specific targeting of tumour cells promotes the growth of tumour-cell variants that are resistant to ICB, and that the acquired resistance can be overcome via the concurrent depletion of tumour cells and of major types of immunosuppressive cell via a monoclonal antibody binding the enzyme CD73, which we identified as highly expressed on tumour cells and on regulatory T cells, myeloid-derived suppressor cells and tumour-associated macrophages, but not on cytolytic T lymphocytes, natural killer cells and dendritic cells. In mice with murine tumours, the systemic administration of anti-PD1 antibodies and anti-CD73 antibodies conjugated to a near-infrared dye prevented near-infrared-irradiated tumours from acquiring resistance to ICB and resulted in the eradication of advanced tumours. The elimination of immunosuppressive cells may overcome acquired resistance to ICB across a range of tumour types and combination therapies. © 2021. The Author(s), under exclusive licence to Springer Nature Limited.
- Cancer Research,
- Immunology and Microbiology
Systemic delivery of TNF-armed myxoma virus plus immune checkpoint inhibitor eliminates lung metastatic mouse osteosarcoma.
In Molecular Therapy Oncolytics on 24 September 2021 by Christie, J. D., Appel, N., et al.
PubMed
Solid cancers that metastasize to the lungs represent a major therapeutic challenge. Current treatment paradigms for lung metastases consist of radiation therapy, chemotherapies, and surgical resection, but there is no single treatment or combination that is effective for all tumor types. To address this, oncolytic myxoma virus (MYXV) engineered to express human tumor necrosis factor (vMyx-hTNF) was tested after systemic administration in an immunocompetent mouse K7M2-Luc lung metastatic osteosarcoma model. Virus therapy efficacy against pre-seeded lung metastases was assessed after systemic infusion of either naked virus or ex vivo-loaded autologous bone marrow leukocytes or peripheral blood mononuclear cells (PBMCs). Results of this study showed that the PBMC pre-loaded strategy was the most effective at reducing tumor burden and increasing median survival time, but sequential intravenous multi-dosing with naked virus was comparably effective to a single infusion of PBMC-loaded virus. PBMC-loaded vMyx-hTNF also potentially synergized very effectively with immune checkpoint inhibitors anti-PD-1, anti-PD-L1, and anti-cytotoxic T lymphocyte associated protein 4 (CTLA-4). Finally, in addition to the pro-immune stimulation caused by unarmed MYXV, the TNF transgene of vMyx-hTNF further induced the unique expression of numerous additional cytokines associated with the innate and adaptive immune responses in this model. We conclude that systemic ex vivo virotherapy with TNF-α-armed MYXV represents a new potential strategy against lung metastatic cancers like osteosarcoma and can potentially act synergistically with established checkpoint immunotherapies. © 2021 The Author(s).
- In Vivo,
- Mus musculus (House mouse),
- Cancer Research
Targeting Pin1 renders pancreatic cancer eradicable by synergizing with immunochemotherapy.
In Cell on 2 September 2021 by Koikawa, K., Kibe, S., et al.
PubMed
Pancreatic ductal adenocarcinoma (PDAC) is characterized by notorious resistance to current therapies attributed to inherent tumor heterogeneity and highly desmoplastic and immunosuppressive tumor microenvironment (TME). Unique proline isomerase Pin1 regulates multiple cancer pathways, but its role in the TME and cancer immunotherapy is unknown. Here, we find that Pin1 is overexpressed both in cancer cells and cancer-associated fibroblasts (CAFs) and correlates with poor survival in PDAC patients. Targeting Pin1 using clinically available drugs induces complete elimination or sustained remissions of aggressive PDAC by synergizing with anti-PD-1 and gemcitabine in diverse model systems. Mechanistically, Pin1 drives the desmoplastic and immunosuppressive TME by acting on CAFs and induces lysosomal degradation of the PD-1 ligand PD-L1 and the gemcitabine transporter ENT1 in cancer cells, besides activating multiple cancer pathways. Thus, Pin1 inhibition simultaneously blocks multiple cancer pathways, disrupts the desmoplastic and immunosuppressive TME, and upregulates PD-L1 and ENT1, rendering PDAC eradicable by immunochemotherapy. Copyright © 2021 Elsevier Inc. All rights reserved.
- Immunology and Microbiology
Siglecs-7/9 function as inhibitory immune checkpoints in vivo and can be targeted to enhance therapeutic antitumor immunity.
In Proceedings of the National Academy of Sciences of the United States of America on 29 June 2021 by Ibarlucea-Benitez, I., Weitzenfeld, P., et al.
PubMed
Given the role of myeloid cells in T cell activation and in the antitumor response, targeting checkpoint molecules expressed on this population represents a promising strategy to augment antitumor immunity. However, myeloid checkpoints that can be effectively used as immunotherapy targets are still lacking. Here, we demonstrate the therapeutic potential of targeting the myeloid receptors Siglec-7 and Siglec-9 in vivo. By using a humanized immunocompetent murine model, we demonstrate that human Siglec-7 and Siglec-9, in addition to the murine homolog Siglec-E, inhibit the endogenous antitumor immune response, as well as the response to tumor-targeting and immune checkpoint inhibiting antibodies in vivo. The impact of these Siglecs on tumor progression is highly dependent on the anatomical distribution of the tumor and, as a consequence, the local tumor microenvironment, as tumors with a more immune-suppressive tumor microenvironment are less sensitive to Siglec perturbation. Finally, to assess the potential of these two receptors as targets for immunotherapy, we developed Fc engineered blocking antibodies to Siglec-7 and Siglec-9 and demonstrate that Siglec-7 and Siglec-9 blockade can significantly reduce tumor burden in vivo, demonstrating the therapeutic potential of targeting these two receptors.
- Cancer Research,
- Genetics,
- Immunology and Microbiology
DNA barcoding reveals ongoing immunoediting of clonal cancer populations during metastatic progression and in response to immunotherapy
Preprint on BioRxiv : the Preprint Server for Biology on 11 January 2021 by Baldwin, L. A., Bartonicek, N., et al.
PubMed
Cancers evade the immune system in order to grow or metastasise through the process of cancer immunoediting. While immune checkpoint inhibitors have been effective for reactivating tumour immunity in some types of cancer, many other solid cancers, including breast cancer, remain largely non-responsive. Understanding the way non-responsive cancers evolve to evade immunity, what resistance pathways are activated and whether this occurs at the clonal level will improve immunotherapeutic design. We tracked cancer cell clones during the immunoediting process and determined clonal transcriptional profiles that allow immune evasion in murine mammary tumour growth in response to immunotherapy with anti-PD1 and anti-CTLA4. Clonal diversity was significantly restricted by immunotherapy treatment in both primary tumours and metastases. These findings demonstrate that immunoediting selects for pre-existing breast cancer cell populations and that immunoediting is not static, it is ongoing during metastasis and immunotherapy treatment. Isolation of immunotherapy resistant clones revealed unique and overlapping transcriptional signatures. The overlapping gene signature was associated with poor survival of basal-like breast cancer patients in two cohorts. At least one of these overlapping genes has an existing small molecule that can potentially be used to improve immunotherapy response.
- Mus musculus (House mouse)
Nanoscale coordination polymers induce immunogenic cell death by amplifying radiation therapy mediated oxidative stress.
In Nature Communications on 8 January 2021 by Huang, Z., Wang, Y., et al.
PubMed
Radiation therapy can potentially induce immunogenic cell death, thereby priming anti-tumor adaptive immune responses. However, radiation-induced systemic immune responses are very rare and insufficient to meet clinical needs. Here, we demonstrate a synergetic strategy for boosting radiation-induced immunogenic cell death by constructing gadolinium-hemin based nanoscale coordination polymers to simultaneously perform X-ray deposition and glutathione depletion. Subsequently, immunogenic cell death is induced by sensitized radiation to potentiate checkpoint blockade immunotherapies against primary and metastatic tumors. In conclusion, nanoscale coordination polymers-sensitized radiation therapy exhibits biocompatibility and therapeutic efficacy in preclinical cancer models, and has the potential for further application in cancer radio-immunotherapy.
- Cancer Research,
- Immunology and Microbiology
CD155 on Tumor Cells Drives Resistance to Immunotherapy by Inducing the Degradation of the Activating Receptor CD226 in CD8+ T Cells.
In Immunity on 13 October 2020 by Braun, M., Aguilera, A. R., et al.
PubMed
The activating receptor CD226 is expressed on lymphocytes, monocytes, and platelets and promotes anti-tumor immunity in pre-clinical models. Here, we examined the role of CD226 in the function of tumor-infiltrating lymphocytes (TILs) and resistance to immunotherapy. In murine tumors, a large proportion of CD8+ TILs had decreased surface expression of CD226 and exhibited features of dysfunction, whereas CD226hi TILs were highly functional. This correlation was seen also in TILs isolated from HNSCC patients. Mutation of CD226 at tyrosine 319 (Y319) led to increased CD226 surface expression, enhanced anti-tumor immunity and improved efficacy of immune checkpoint blockade (ICB). Mechanistically, tumor-derived CD155, the ligand for CD226, initiated phosphorylation of Y319 by Src kinases, thereby enabling ubiquitination of CD226 by CBL-B, internalization, and proteasomal degradation. In pre-treatment samples from melanoma patients, CD226+CD8+ T cells correlated with improved progression-free survival following ICB. Our findings argue for the development of therapies aimed at maintaining the expression of CD226. Copyright © 2020 Elsevier Inc. All rights reserved.
- Immunology and Microbiology
ACKR4 restrains antitumor immunity by regulating CCL21.
In The Journal of Experimental Medicine on 1 June 2020 by Whyte, C. E., Osman, M., et al.
PubMed
Current immunotherapies involving CD8+ T cell responses show remarkable promise, but their efficacy in many solid tumors is limited, in part due to the low frequency of tumor-specific T cells in the tumor microenvironment (TME). Here, we identified a role for host atypical chemokine receptor 4 (ACKR4) in controlling intratumor T cell accumulation and activation. In the absence of ACKR4, an increase in intratumor CD8+ T cells inhibited tumor growth, and nonhematopoietic ACKR4 expression was critical. We show that ACKR4 inhibited CD103+ dendritic cell retention in tumors through regulation of the intratumor abundance of CCL21. In addition, preclinical studies indicate that ACKR4 and CCL21 are potential therapeutic targets to enhance responsiveness to immune checkpoint blockade or T cell costimulation. © 2020 Crown copyright. The government of Australia, Canada, or the UK ("the Crown") owns the copyright interests of authors who are government employees. The Crown Copyright is not transferable.
- Cancer Research,
- Immunology and Microbiology
Dendritic Cell Paucity Leads to Dysfunctional Immune Surveillance in Pancreatic Cancer.
In Cancer Cell on 16 March 2020 by Hegde, S., Krisnawan, V. E., et al.
PubMed
Here, we utilized spontaneous models of pancreatic and lung cancer to examine how neoantigenicity shapes tumor immunity and progression. As expected, neoantigen expression during lung adenocarcinoma development leads to T cell-mediated immunity and disease restraint. By contrast, neoantigen expression in pancreatic ductal adenocarcinoma (PDAC) results in exacerbation of a fibro-inflammatory microenvironment that drives disease progression and metastasis. Pathogenic TH17 responses are responsible for this neoantigen-induced tumor progression in PDAC. Underlying these divergent T cell responses in pancreas and lung cancer are differences in infiltrating conventional dendritic cells (cDCs). Overcoming cDC deficiency in early-stage PDAC leads to disease restraint, while restoration of cDC function in advanced PDAC restores tumor-restraining immunity and enhances responsiveness to radiation therapy. Copyright © 2020 Elsevier Inc. All rights reserved.
- Mus musculus (House mouse),
- Immunology and Microbiology
Quorum Regulation via Nested Antagonistic Feedback Circuits Mediated by the Receptors CD28 and CTLA-4 Confers Robustness to T Cell Population Dynamics.
In Immunity on 18 February 2020 by Zenke, S., Palm, M. M., et al.
PubMed
T cell responses upon infection display a remarkably reproducible pattern of expansion, contraction, and memory formation. If the robustness of this pattern builds entirely on signals derived from other cell types or if activated T cells themselves contribute to the orchestration of these population dynamics-akin to bacterial quorum regulation-is unclear. Here, we examined this question using time-lapse microscopy, genetic perturbation, bioinformatic predictions, and mathematical modeling. We found that ICAM-1-mediated cell clustering enabled CD8+ T cells to collectively regulate the balance between proliferation and apoptosis. Mechanistically, T cell expressed CD80 and CD86 interacted with the receptors CD28 and CTLA-4 on neighboring T cells; these interactions fed two nested antagonistic feedback circuits that regulated interleukin 2 production in a manner dependent on T cell density as confirmed by in vivo modulation of this network. Thus, CD8+ T cell-population-intrinsic mechanisms regulate cellular behavior, thereby promoting robustness of population dynamics. Copyright © 2020 Elsevier Inc. All rights reserved.