InVivoMAb anti-mouse 4-1BB (CD137)
Product Description
Specifications
| Isotype | Rat IgG2a |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
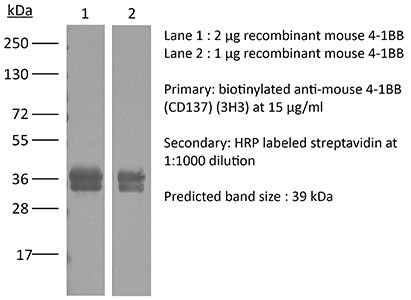
| Immunogen | Mouse CD137 human Fc fusion protein |
| Reported Applications |
in vivo 4-1BB stimulation in vitro 4-1BB stimulation in vitro Organoids/Organ-on-Chip |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_2687721 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Qi, X., et al (2019). "Optimization of 4-1BB antibody for cancer immunotherapy by balancing agonistic strength with FcgammaR affinity" Nat Commun 10(1): 2141.
PubMed
Costimulation of T cell responses with monoclonal antibody agonists (mAb-AG) targeting 4-1BB showed robust anti-tumor activity in preclinical models, but their clinical development was hampered by low efficacy (Utomilumab) or severe liver toxicity (Urelumab). Here we show that isotype and intrinsic agonistic strength co-determine the efficacy and toxicity of anti-4-1BB mAb-AG. While intrinsically strong agonistic anti-4-1BB can activate 4-1BB in the absence of FcgammaRs, weak agonistic antibodies rely on FcgammaRs to activate 4-1BB. All FcgammaRs can crosslink anti-41BB antibodies to strengthen co-stimulation, but activating FcgammaR-induced antibody-dependent cell-mediated cytotoxicity compromises anti-tumor immunity by deleting 4-1BB(+) cells. This suggests balancing agonistic activity with the strength of FcgammaR interaction as a strategy to engineer 4-1BB mAb-AG with optimal therapeutic performance. As a proof of this concept, we have developed LVGN6051, a humanized 4-1BB mAb-AG that shows high anti-tumor efficacy in the absence of liver toxicity in a mouse model of cancer immunotherapy.
-
Giardino Torchia, M. L., et al (2015). "c-IAP ubiquitin protein ligase activity is required for 4-1BB signaling and CD8(+) memory T-cell survival" Eur J Immunol 45(9): 2672-2682.
PubMed
Cellular inhibitor of apoptosis proteins (c-IAP) 1 and 2 are widely expressed ubiquitin protein ligases that regulate a variety of cellular functions, including the sensitivity of T cells to costimulation. 4-1BB is a TNF receptor family member that signals via a complex that includes TRAF family members and the c-IAPs to upregulate NF-kappaB and ERK, and has been implicated in memory T-cell survival. Here, we show that effector and memory T cells from mice expressing a dominant negative E3-inactive c-IAP2 (c-IAP2(H570A) ) have impaired signaling downstream of 4-1BB. When infected with lymphocytic choriomeningitis virus, unlike mice in which c-IAPs were acutely downregulated by c-IAP antagonists, the primary response of c-IAP2(H570A) mice was normal. However, the number of antigen-specific CD8(+) but not CD4(+) T cells declined more rapidly and to a greater extent in c-IAP2(H570A) mice than in WT controls. Studies with T-cell adoptive transfer demonstrated that the enhanced decay of memory cells was T-cell intrinsic. Thus, c-IAP E3 activity is required for 4-1BB coreceptor signaling and maintenance of CD8(+) T-cell memory.
-
Guillerey, C., et al (2015). "Immunosurveillance and therapy of multiple myeloma are CD226 dependent" J Clin Invest 125(5): 2077-2089.
PubMed
Multiple myeloma (MM) is an age-dependent hematological malignancy. Evaluation of immune interactions that drive MM relies on in vitro experiments that do not reflect the complex cellular stroma involved in MM pathogenesis. Here we used Vk*MYC transgenic mice, which spontaneously develop MM, and demonstrated that the immune system plays a critical role in the control of MM progression and the response to treatment. We monitored Vk*MYC mice that had been crossed with Cd226 mutant mice over a period of 3 years and found that CD226 limits spontaneous MM development. The CD226-dependent anti-myeloma immune response against transplanted Vk*MYC MM cells was mediated both by NK and CD8+ T cells through perforin and IFN-gamma pathways. Moreover, CD226 expression was required for optimal antimyeloma efficacy of cyclophosphamide (CTX) and bortezomib (Btz), which are both standardly used to manage MM in patients. Activation of costimulatory receptor CD137 with mAb (4-1BB) exerted strong antimyeloma activity, while inhibition of coinhibitory receptors PD-1 and CTLA-4 had no effect. Taken together, the results of this study provide in vivo evidence that CD226 is important for MM immunosurveillance and indicate that specific immune components should be targeted for optimal MM treatment efficacy. As progressive immunosuppression associates with MM development, strategies aimed to increase immune functions may have important therapeutic implications in MM.
-
Kobayashi, T., et al (2015). "NKT cell-targeted vaccination plus anti-4-1BB antibody generates persistent CD8 T cell immunity against B cell lymphoma" Oncoimmunology 4(3): e990793.
PubMed
Harnessing the immune adjuvant properties of natural killer T (NKT) cells is an effective strategy to generate anticancer immunity. The objective of this study was to increase the potency and durability of vaccine-induced immunity against B cell lymphoma by combining alpha-galactosylceramide (alpha-GalCer)-loaded tumor cell vaccination with an agonistic antibody targeting the immune checkpoint molecule 4-1BB (CD137). We observed potent synergy when combining vaccination and anti-4-1BB antibody treatment resulting in significantly enhanced survival of mice harboring Emu-myc tumors, including complete eradication of lymphoma in over 50% of mice. Tumor-free survival required interferon gamma (IFNgamma)-dependent expansion of CD8+ T cells and was associated with 4-1BB-mediated differentiation of KLRG1+ effector CD8+ T cells. ‘Cured’ mice were also resistant to lymphoma re-challenge 80 days later indicating successful generation of immunological memory. Overall, our results demonstrate that therapeutic anticancer vaccination against B cell lymphoma using an NKT cell ligand can be boosted by subsequent co-stimulation through 4-1BB leading to a sustainable immune response that may enhance outcomes to conventional treatment.
Product Citations
-
Chronic inflammation-responsive hydrogel restores myeloid-T cell crosstalk to reinvigorate antitumor immunity against metastatic colorectal cancer.
In Bioact Mater on 1 August 2026 by Li, X., Fu, W., et al.
PubMed
Chronic inflammation in intermediate/advanced tumors drives burdensome protumor immune cell communication, thereby weakening immunotherapy. Conventional anti-inflammatory therapies focus on alleviating chronic inflammation whereas ignore dysfunction and scarcity of myeloid and T cells, which hinder their intercellular communication restoration. To address the dilemma, an inflammatory condition-triggered protumor inflammation-immunosurveillance shift hydrogel (TRANS) is developed to initiate adaptive immune responses mediated by intercellular communication. Triggered by inflammatory conditions, TRANS releases celecoxib (CXB) to inhibit the COX-2/PGE2 pathway, thereby reprogramming tumor-associated macrophages and mitigating protumor inflammation. Furthermore, TRANS incorporates FMS-like tyrosine kinase 3 ligands (Flt-3L) and 4-1BB agonists (α-CD137) to respectively recruit type 1 conventional DC (cDC1) and revitalize tumor-infiltrating T cells, to rejuvenate immunosurveillance. TRANS inhibits 87.50% and 88.74% of primary and secondary colorectal tumors, generates antitumor immune memory to resist tumor rechallenge, and significantly reduces lung and liver metastases. Rechallenge model shows TRANS leads to antitumor immune memory formation. Single-cell RNA sequencing is preform to elucidate the mechanism of TRANS, which exhibits that TRANS exerts antitumor effects by optimizing the crosstalk between myeloid cells and T cells via CXCL9/10-CXCR3/DPP4. TRANS further gains better control of colorectal cancer when combined with immune checkpoints inhibitors. This study offers a novel perspective on immunotherapy by rebalancing inflammation-immunity dynamics.
-
Elucidating the Mechanisms of SA-4-1BBL-Mediated Cancer Immunoprevention Through Advanced Informatics Approaches.
In Biomolecules on 4 February 2026 by Verma, M., Arguc, F. N., et al.
PubMed
Cancer immunoprevention leverages the immune system's surveillance mechanisms to mitigate tumor development. Vaccines that constitute a tumor antigen and an immune adjuvant are perceived as immunoprevention modalities. However, relevant tumor antigens are unknown for non-viral cancers, which constitute most human cancers. Our group has recently shown that SA-4-1BBL, a novel agonist of CD137 receptor, but not antibodies, shows immunoprevention efficacy against various tumors. Advanced bioinformatics analyses of bulk RNA-seq data were conducted to elucidate mechanisms underlying cancer immunoprevention. Mice received subcutaneous injections of SA-4-1BBL or agonistic 3H3 antibody, and the injection-site tissue (IS) and draining lymph nodes (LN) were analyzed for differential gene expression. SA-4-1BBL induced a compartmentalized and temporally dynamic immune program characterized by early effector activation at IS and sustained immune regulation in draining LN. K-means clustering of 4564 DEGs identified eight functionally distinct clusters. IS-enriched clusters contained activation genes for CD4+ T and NK cells, including Cd28, Klra1, Cd4, Cd40, and Cd40l, while LN clusters were enriched for regulatory genes (Tnfaip3, Irf5, Col1a2) that ensure immune priming and homeostatic restraint for a balanced response. SA-4-1BBL generated a more selective and durable activation of adaptive immunity, TCR signaling, Th1/Th2 differentiation, and NK cytotoxicity. 3H3 activated broader innate inflammatory programs, including Toll-like receptor and neurodegeneration-linked pathways. IMPRes analysis showed that SA-4-1BBL activates sequential immune-regulatory circuits centered on Stat1, Cd247, and Ifng and modulates the CD151-TGF-β axis. These findings demonstrate that SA-4-1BBL elicits a balanced immune response, ensuring both safety and efficacy in preventing cancer development.
-
Enrichment of decidual CD11c + CD8 + T cells with altered immune function in early pregnancy loss.
In Nat Commun on 21 July 2025 by Guo, L., Guo, A., et al.
PubMed
Early pregnancy loss (EPL) is closely associated with imbalances in the maternal-foetal immune microenvironment. Here we identify CD11c + CD8 + T cells, an unconventional cytotoxic T cell subset, as significantly enriched and activated in EPL cases. These cells contribute to immune dysregulation and inhibit trophoblast invasion through secreting granzyme B, perforin, CD107a, TNF-α, and IFN-γ. Furthermore, we present an effective early prediction model for EPL, based on cytokine and cytotoxic molecule profiles of CD11c + CD8 + T cells in maternal serum, collected 12-16 days post-embryo transfer. Functional assays reveal that IFN-γ triggers trophoblast pyroptosis via the NLRP3/Caspase-1/GSDMD pathway, impairing trophoblast invasion. In vivo validation using abortion-prone mice and an anti-4-1BB antibody-induced model of CD11c + CD8 + T cell activation confirms increased embryo resorption and reduced trophoblast infiltration. These findings highlight the role of dysregulated CD11c + CD8 + T cells at the maternal-foetal interface in EPL, and suggest their potential as biomarkers and therapeutic targets for EPL-management.
-
An amphiregulin reporter mouse enables transcriptional and clonal expansion analysis of reparative lung Tregs.
In JCI Insight on 8 July 2025 by Loffredo, L. F., Kaiser, K. A., et al.
PubMed
Regulatory T cells (Tregs) are known to play critical roles in tissue repair via provision of growth factors, such as amphiregulin (Areg). Areg-producing Tregs have previously been difficult to study because of an inability to isolate live Areg-producing cells. In this report, we created a reporter mouse to detect Areg expression in live cells (AregThy1.1). We employed influenza A and bleomycin models of lung damage to sort Areg-producing and non-Areg-producing Tregs for transcriptomic analyses. Single-cell RNA-Seq revealed distinct subpopulations of Tregs and allowed transcriptomic comparisons of damage-induced populations. Single-cell TCR sequencing showed that Treg clonal expansion was biased toward Areg-producing Tregs and largely occurred within damage-induced subgroups. Gene module analysis revealed functional divergence of Tregs into immunosuppression-oriented and tissue repair-oriented groups, leading to identification of candidate receptors for induction of repair activity in Tregs. We tested these using an ex vivo assay for Treg-mediated tissue repair, identifying 4-1BB agonism as a mechanism for reparative activity induction. Overall, we demonstrate that the AregThy1.1 mouse is a promising tool for investigating tissue repair activity in leukocytes.