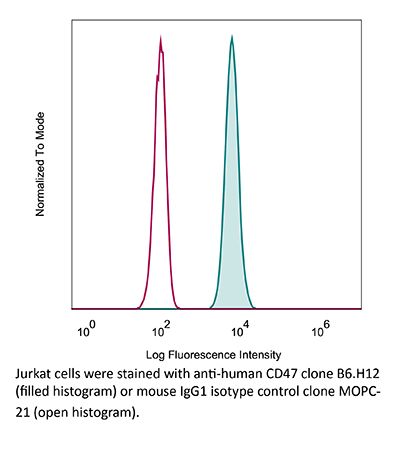
InVivoMAb anti-human CD47
Product Description
Specifications
| Isotype | Mouse IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG1 isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Intact CD47 purified from placenta |
| Reported Applications |
in vitro CD47 neutralization in vivo CD47 neutralization in human tumor xenograft models or humanized mice Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107655 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Gordon, S. R., et al (2017). "PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity" Nature 545(7655): 495-499.
PubMed
Programmed cell death protein 1 (PD-1) is an immune checkpoint receptor that is upregulated on activated T cells for the induction of immune tolerance. Tumour cells frequently overexpress the ligand for PD-1, programmed cell death ligand 1 (PD-L1), facilitating their escape from the immune system. Monoclonal antibodies that block the interaction between PD-1 and PD-L1, by binding to either the ligand or receptor, have shown notable clinical efficacy in patients with a variety of cancers, including melanoma, colorectal cancer, non-small-cell lung cancer and Hodgkin’s lymphoma. Although it is well established that PD-1-PD-L1 blockade activates T cells, little is known about the role that this pathway may have in tumour-associated macrophages (TAMs). Here we show that both mouse and human TAMs express PD-1. TAM PD-1 expression increases over time in mouse models of cancer and with increasing disease stage in primary human cancers. TAM PD-1 expression correlates negatively with phagocytic potency against tumour cells, and blockade of PD-1-PD-L1 in vivo increases macrophage phagocytosis, reduces tumour growth and lengthens the survival of mice in mouse models of cancer in a macrophage-dependent fashion. This suggests that PD-1-PD-L1 therapies may also function through a direct effect on macrophages, with substantial implications for the treatment of cancer with these agents.
-
Lo, J., et al (2015). "Nuclear factor kappa B-mediated CD47 up-regulation promotes sorafenib resistance and its blockade synergizes the effect of sorafenib in hepatocellular carcinoma in mice" Hepatology 62(2): 534-545.
PubMed
Sorafenib is a new standard treatment for patients with advanced hepatocellular carcinoma (HCC). However, the survival benefit of this treatment is modest, partly owing to drug resistance. Recent evidence has demonstrated the existence of tumor-initiating cells (T-ICs) as the culprit for treatment resistance. To examine whether sorafenib resistance was a result of the presence of liver T-ICs, we developed sorafenib-resistant HCC cells both in vitro and in vivo through continuous exposure to sorafenib. Using these models, we found that sorafenib-resistant clones demonstrated enhanced T-IC properties, including tumorigenicity, self-renewal, and invasiveness. In addition, several T-IC markers were found to be up-regulated, among which CD47 was found to be most significant. Using chromatin immunoprecipitation assays and expression analyses, CD47 expression was found to be regulated by nuclear factor kappa B (NF-kappaB) through a specific response element in the promoter of CD47, and the site occupancy and expression were increased and decreased upon stimulation and inhibition of NF-kappaB, respectively. Consistently, NF-kappaB was activated in sorafenib-resistant HCC cells, and this finding was confirmed in clinical HCC samples, which showed a positive correlation between NF-kappaB and CD47 expression. Functional characterization of CD47 in sorafenib-resistant HCC cells was evaluated using a lentivirus-based knockdown approach and showed increased sensitization to sorafenib upon CD47 knockdown. Furthermore, blockade of CD47 using anti-CD47 antibody (Ab) showed a similar effect. Using a patient-derived HCC xenograft mouse model, we found that anti-CD47 Ab (500 mug/mouse) in combination with sorafenib (100 mg/kg, orally) exerted synergistic effects on tumor suppression, as compared with sorafenib and anti-CD47 Ab alone. CONCLUSIONS: NF-kappaB-mediated CD47 up-regulation promotes sorafenib resistance, and targeting CD47 in combination with sorafenib is an attractive therapeutic regimen for the treatment of HCC patients.
-
Lo, J., et al (2015). "Anti-CD47 antibody suppresses tumour growth and augments the effect of chemotherapy treatment in hepatocellular carcinoma" Liver Int. doi : 10.1111/liv.12963.
PubMed
BACKGROUND & AIMS: Hepatocellular carcinoma (HCC) is often associated with metastasis and recurrence leading to a poor prognosis. Therefore, development of novel treatment regimens is urgently needed to improve the survival of HCC patients. In this study, we aimed to investigate the in vitro and in vivo effects of anti-CD47 antibody alone and in combination with chemotherapy in HCC. METHODS: In this study, we examined the functional effects of anti-CD47 antibody (B6H12) on cell proliferation, sphere formation, migration and invasion, chemosensitivity, macrophage-mediated phagocytosis and tumourigenicity both in vitro and in vivo. The therapeutic efficacy of anti-CD47 antibody alone or in combination with doxorubicin was examined in patient-derived HCC xenograft. RESULTS: Blocking CD47 with anti-CD47 monoclonal antibody (B6H12) at 10 mug/ml could suppress self-renewal, tumourigenicity and migration and invasion abilities of MHCC-97L and Huh-7 cells. Interestingly, anti-CD47 antibody synergized the effect of HCC cells to chemotherapeutic drugs including doxorubicin and cisplatin. Blockade of CD47 by anti-CD47 antibody induced macrophage-mediated phagocytosis. Using a patient-derived HCC xenograft mouse model, we found that anti-CD47 antibody (400 mug/mouse) in combination with doxorubicin (2 mg/kg) exerted maximal effects on tumour suppression, as compared with doxorubicin and anti-CD47 antibody alone. CONCLUSIONS: Anti-CD47 antibody treatment could complement chemotherapy which may be a promising therapeutic strategy for the treatment of HCC patients.
-
Xiao, Z., et al (2015). "Antibody mediated therapy targeting CD47 inhibits tumor progression of hepatocellular carcinoma" Cancer Lett 360(2): 302-309.
PubMed
Human hepatocellular carcinoma (HCC) has a high rate of tumor recurrence and metastasis, resulting in shortened survival times. The efficacy of current systemic therapies for HCC is limited. In this study, we used xenograft tumor models to investigate the use of antibodies that block CD47 and inhibit HCC tumor growth. Immunostaining of tumor tissue and HCC cell lines demonstrated CD47 over-expression in HCC as compared to normal hepatocytes. Macrophage phagocytosis of HCC cells was increased after treatment with CD47 antibodies (CD47mAbs) that block CD47 binding to SIRPalpha. Further, CD47 blockade inhibited tumor growth in both heterotopic and orthotopic models of HCC, and promoted the migration of macrophages into the tumor mass. Our results demonstrate that targeting CD47 by specific antibodies has potential immunotherapeutic efficacy in human HCC.
Product Citations
-
Photothermal therapy synergizes with CD47 blockade by inducing calreticulin exposure and remodeling the tumor extracellular matrix in oral squamous cell carcinoma.
In Cancer Immunol Immunother on 28 January 2026 by Tang, Q., Hu, L., et al.
PubMed
Oral squamous cell carcinoma (OSCC) overexpresses CD47, enabling immune evasion via a "don't eat me" signal to macrophages. Although CD47 blockade shows promise, its efficacy is limited due to a lack of "eat me" signal and contact between macrophages and tumor cells.
-
Macrophages rescue cells from ferroptotic death.
In Cell Death Dis on 1 December 2025 by Hefetz, R., Lianski, S., et al.
PubMed
Ferroptosis, a non-apoptotic form of cell death marked by iron-dependent lipid peroxidation, has a key role in organ injury, degenerative disease, and vulnerability of therapy-resistant cancers. Although substantial progress has been made in understanding the molecular processes relevant to ferroptosis, additional cell-extrinsic processes that determine cell sensitivity toward ferroptosis remain unknown. Here we demonstrate that macrophages co-cultured with ferroptotic cancer cells from various types effectively mitigate cell death induced by GPX4 inhibitors (RSL3 and ML162), GPX4 silencing via shRNA, or the Xc- system inhibitor IKE. Furthermore, macrophages effectively reduced lipid peroxidation in ferroptotic cells. Importantly, macrophage function relies on direct cell-to-cell contact and is affected by their differentiation. Specifically, polarization into M1 macrophages, but not M2, greatly hinders their protective capabilities. Interestingly, unlike apoptotic cells, ferroptotic cells retain elevated levels of the 'don't eat me' signal, CD47, and conversely, fail to present the "eat me" signal phosphatidylserine (PS) on the outer layer of the plasma membrane, providing an opportunity for their rescue. Furthermore, in placental villi explants, macrophages protect trophoblasts from ferroptotic death. These results underscore the intricate interplay between ferroptotic cells and their microenvironment and provide compelling evidence of a yet-unrecognized anti-ferroptotic activity of macrophages as a cell-extrinsic mechanism that could be exploited by cancer cells to escape ferroptosis.
-
Repurposing Cardiac Glycosides to Potentiate CD47 Blockade through Calreticulin-mediated Phagocytic Effects for Lung Cancer Treatment.
In Adv Sci (Weinh) on 1 December 2025 by Ye, Z. H., Yu, W. B., et al.
PubMed
The abundance of macrophages within the tumor microenvironment (TME) of lung cancer represents a noteworthy therapeutic target. Exploiting the phagocytic function of macrophages by blocking the "don't eat me" signal, CD47, has shown significant therapeutic potential. However, novel CD47-targeted combination strategies warrant further investigation. Through an analysis of data obtained from a screening model focused on the macrophage-mediated killing effect, two cardiac glycosides (CGs), ouabain and digoxin, are shown to increase the capacity of macrophages to kill cancer cells after combination with CD47 antibody. Compared with the control, the combination strategy reduced the tumor volume in different lung cancer models and increased the macrophage phagocytosis rate ≈5-fold. Mechanistically, in addition to Fc-FcγR interaction, CGs enhanced the expression of a pro-phagocytotic signal, calreticulin (CRT). Moreover, PERK inhibitor, ER-Golgi protein trafficking inhibitor, and siRNA-mediated knockdown of exocytosis protein exo70, abrogated both CGs-induced CRT upregulation and the ensuing enhancement of phagocytosis. These findings indicate that CGs drive CRT translocation originates from ER to Golgi apparatus, where it subsequently anchors to the cell surface via exo70-mediated exocytosis. Overall, this study offers compelling evidence that supports the clinical translation of an innovative combination regimen for the treatment of patients with lung cancer.
-
The C-type lectin receptor DCIR senses galactose-terminated N-glycans on LRP1 to regulate myeloid cell function
In Research Square on 11 November 2025 by Rombouts, Y., Raymond, B., et al.