InVivoMAb anti-mouse 4-1BB (CD137)
Product Details
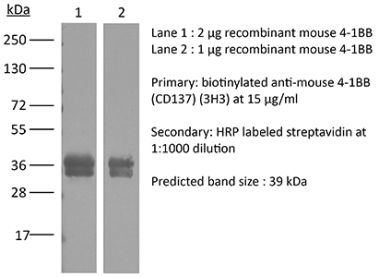
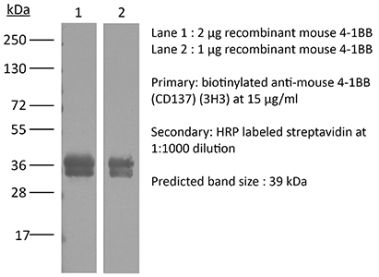
The LOB12.3 monoclonal antibody reacts with mouse 4-1BB, a TNF receptor superfamily member also known as CD137. 4-1BB is a 39 kDa transmembrane protein expressed by T lymphocytes, NK cells, dendritic cells, granulocytes, and mast cells. Upon binding its ligand 4-1BBL, 4-1BB provides costimulatory signals to both CD4 and CD8 T cells through the activation of NF- κB, c-Jun and p38 downstream pathways. The importance of the 4-1BB pathway has been underscored in a number of diseases, including cancer. Agonistic anti-4-1BB antibodies have been reported to induce T cell mediated antitumor immunity. The LOB12.3 antibody is an agonistic antibody that has been shown to stimulate 4-1BB signaling and delay tumor growth in vivo when administered in combination with immune checkpoint inhibitors.Specifications
| Isotype | Rat IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG1 isotype control, anti-horseradish peroxidase |
| Recommended Dilution Buffer | InVivoPure pH 8.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse CD137 human Fc fusion protein |
| Reported Applications | in vivo activation of 4-1BB |
| Formulation |
PBS, pH 8.0 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_10949016 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Additional Formats
Recommended Products
in vivo activation of 4-1BB
Qi, X., et al. (2019). "Optimization of 4-1BB antibody for cancer immunotherapy by balancing agonistic strength with FcgammaR affinity" Nat Commun 10(1): 2141. PubMed
Costimulation of T cell responses with monoclonal antibody agonists (mAb-AG) targeting 4-1BB showed robust anti-tumor activity in preclinical models, but their clinical development was hampered by low efficacy (Utomilumab) or severe liver toxicity (Urelumab). Here we show that isotype and intrinsic agonistic strength co-determine the efficacy and toxicity of anti-4-1BB mAb-AG. While intrinsically strong agonistic anti-4-1BB can activate 4-1BB in the absence of FcgammaRs, weak agonistic antibodies rely on FcgammaRs to activate 4-1BB. All FcgammaRs can crosslink anti-41BB antibodies to strengthen co-stimulation, but activating FcgammaR-induced antibody-dependent cell-mediated cytotoxicity compromises anti-tumor immunity by deleting 4-1BB(+) cells. This suggests balancing agonistic activity with the strength of FcgammaR interaction as a strategy to engineer 4-1BB mAb-AG with optimal therapeutic performance. As a proof of this concept, we have developed LVGN6051, a humanized 4-1BB mAb-AG that shows high anti-tumor efficacy in the absence of liver toxicity in a mouse model of cancer immunotherapy.
in vivo activation of 4-1BB
Dai, M., et al. (2015). "Curing mice with large tumors by locally delivering combinations of immunomodulatory antibodies" Clin Cancer Res 21(5): 1127-1138. PubMed
PURPOSE: Immunomodulatory mAbs can treat cancer, but cures are rare except for small tumors. Our objective was to explore whether the therapeutic window increases by combining mAbs with different modes of action and injecting them into tumors. EXPERIMENTAL DESIGN: Combinations of mAbs to CD137/PD-1/CTLA-4 or CD137/PD-1/CTLA-4/CD19 were administrated intratumorally to mice with syngeneic tumors (B16 and SW1 melanoma, TC1 lung carcinoma), including tumors with a mean surface of approximately 80 mm(2). Survival and tumor growth were assessed. Immunologic responses were evaluated using flow cytometry and qRT-PCR. RESULTS: More than 50% of tumor-bearing mice had complete regression and long-term survival after tumor injection with mAbs recognizing CD137/PD-1/CTLA-4/CD19 with similar responses in three models. Intratumoral injection was more efficacious than intraperitoneal injection in causing rejection also of untreated tumors in the same mice. The three-mAb combination could also induce regression, but was less efficacious. There were few side effects, and therapy-resistant tumors were not observed. Transplanted tumor cells rapidly caused a Th2 response with increased CD19 cells. Successful therapy shifted this response to the Th1 phenotype with decreased CD19 cells and increased numbers of long-term memory CD8 effector cells and T cells making IFNgamma and TNFalpha. CONCLUSIONS: Intratumoral injection of mAbs recognizing CD137/PD-1/CTLA-4/CD19 can eradicate established tumors and reverse a Th2 response with tumor-associated CD19 cells to Th1 immunity, whereas a combination lacking anti-CD19 is less effective. There are several human cancers for which a similar approach may provide clinical benefit.
in vivo activation of 4-1BB
Bartkowiak, T., et al. (2015). "Unique potential of 4-1BB agonist antibody to promote durable regression of HPV+ tumors when combined with an E6/E7 peptide vaccine" Proc Natl Acad Sci U S A 112(38): E5290-5299. PubMed
Antibody modulation of T-cell coinhibitory (e.g., CTLA-4) or costimulatory (e.g., 4-1BB) receptors promotes clinical responses to a variety of cancers. Therapeutic cancer vaccination, in contrast, has produced limited clinical benefit and no curative therapies. The E6 and E7 oncoproteins of human papilloma virus (HPV) drive the majority of genital cancers, and many oropharyngeal tumors. We discovered 15-19 amino acid peptides from HPV-16 E6/E7 for which induction of T-cell immunity correlates with disease-free survival in patients treated for high-grade cervical neoplasia. We report here that intranasal vaccination with these peptides and the adjuvant alpha-galactosylceramide elicits systemic and mucosal T-cell responses leading to reduced HPV(+) TC-1 tumor growth and prolonged survival in mice. We hypothesized that the inability of these T cells to fully reject established tumors resulted from suppression in the tumor microenvironment which could be ameliorated through checkpoint modulation. Combining this E6/E7 peptide vaccine with checkpoint blockade produced only modest benefit; however, coadministration with a 4-1BB agonist antibody promoted durable regression of established genital TC-1 tumors. Relative to other therapies tested, this combination of vaccine and alpha4-1BB promoted the highest CD8(+) versus regulatory FoxP3(+) T-cell ratios, elicited 2- to 5-fold higher infiltration by E7-specific CTL, and evoked higher densities of highly cytotoxic TcEO (T cytotoxic Eomesodermin) CD8 (>70-fold) and ThEO (T helper Eomesodermin) CD4 (>17-fold) T cells. These findings have immediate clinical relevance both in terms of the direct clinical utility of the vaccine studied and in illustrating the potential of 4-1BB antibody to convert therapeutic E6/E7 vaccines already in clinical trials into curative therapies.
in vivo activation of 4-1BB
Murphy, J. T., et al. (2014). "Anaphylaxis caused by repetitive doses of a GITR agonist monoclonal antibody in mice" Blood 123(14): 2172-2180. PubMed
Immunotherapy for cancer using antibodies to enhance T-cell function has been successful in recent clinical trials. Many molecules that improve activation and effector function of T cells have been investigated as potential new targets for immunomodulatory antibodies, including the tumor necrosis factor receptor superfamily members GITR and OX40. Antibodies engaging GITR or OX40 result in significant tumor protection in preclinical models. In this study, we observed that the GITR agonist antibody DTA-1 causes anaphylaxis in mice upon repeated intraperitoneal dosing. DTA-1-induced anaphylaxis requires GITR, CD4(+) T cells, B cells, and interleukin-4. Transfer of serum antibodies from DTA-1-treated mice, which contain high levels of DTA-1-specific immunoglobulin G1 (IgG1), can induce anaphylaxis in naive mice upon administration of an additional dose of DTA-1, suggesting that anaphylaxis results from anti-DTA-1 antibodies. Depletion of basophils and blockade of platelet-activating factor, the key components of the IgG1 pathway of anaphylaxis, rescues the mice from DTA-1-induced anaphylaxis. These results demonstrate a previously undescribed lethal side effect of repetitive doses of an agonist immunomodulatory antibody as well as insight into the mechanism of toxicity, which may offer a means of preventing adverse effects in future clinical trials using anti-GITR or other agonist antibodies as immunotherapies.
in vivo activation of 4-1BB
Manzke, N., et al. (2013). "CD4+ T cells develop antiretroviral cytotoxic activity in the absence of regulatory T cells and CD8+ T cells" J Virol 87(11): 6306-6313. PubMed
Conventional CD4(+) T cells play an important role in viral immunity. In most virus infections, they provide essential help for antiviral B and T cell responses. In chronic infections, including HIV infection, an expansion of regulatory T cells (Tregs) has been demonstrated, which can suppress virus-specific CD4(+) T cell responses in vitro. However, the suppressive activity of Tregs on effector CD4(+) T cells in retroviral infection is less well documented in vivo. We took advantage of a transgenic mouse in which Tregs can be selectively depleted to determine the influence of such cells on retrovirus-specific CD4(+) T cell responses during an ongoing infection. Mice were infected with Friend retrovirus (FV), and Tregs were depleted during the acute phase of the infection. In nondepleted mice, activated CD4(+) T cells produced Th1-type cytokines but did not exhibit any antiviral cytotoxicity as determined in a major histocompatibility complex (MHC) class II-restricted in vivo cytotoxic T lymphocyte (CTL) assay. Depletion of Tregs significantly increased the numbers of virus-specific CD4(+) T cells and improved their cytokine production, whereas it induced only very little CD4(+) T cell cytotoxicity. However, after dual depletion of Tregs and CD8(+) T cells, conventional CD4(+) T cells developed significant cytotoxic activity against FV epitope-labeled target cells in vivo and contributed to the control of virus replication. Thus, both Tregs and CD8(+) T cells influence the cytotoxic activity of conventional CD4(+) T cells during an acute retroviral infection.
in vivo activation of 4-1BB
Wei, H., et al. (2013). "Combinatorial PD-1 blockade and CD137 activation has therapeutic efficacy in murine cancer models and synergizes with cisplatin" PLoS One 8(12): e84927. PubMed
90 days (and was probably curative) by a mechanism which included a systemic CD8(+) T cell response with tumor specificity and immunological memory. Strikingly, combined treatment of cisplatin and CD137/PD-1 mAb also gave rise to the long-term survival of mice with established TC1 lung tumors. A similar combination of the 2 mAbs and cisplatin should be considered for clinical ‘translation’.”}” data-sheets-userformat=”{“2″:14851,”3”:{“1″:0},”4”:{“1″:2,”2″:16777215},”12″:0,”14”:{“1″:2,”2″:1521491},”15″:”Roboto, sans-serif”,”16″:12}”>There is an urgent need for improved therapy for advanced ovarian carcinoma, which may be met by administering immune-modulatory monoclonal antibodies (mAbs) to generate a tumor-destructive immune response. Using the ID8 mouse ovarian cancer model, we investigated the therapeutic efficacy of various mAb combinations in mice with intraperitoneal (i.p.) tumor established by transplanting 3 x 10(6) ID8 cells 10 days previously. While most of the tested mAbs were ineffective when given individually or together, the data confirm our previous finding that 2 i.p. injections of a combination of anti-CD137 with anti-PD-1 mAbs doubles overall survival. Mice treated with this mAb combination have a significantly increased frequency and total number of CD8(+) T cells both in the peritoneal lavage and spleens, and these cells are functional as demonstrated by antigen-specific cytolytic activity and IFN-gamma production. While administration of anti-CD137 mAb as a single agent similarly increases CD8(+) T cells, these have no functional activity, which may be attributed to up-regulation of co-inhibitory PD-1 and TIM-3 molecules induced by CD137. Addition of the anti-cancer drug cisplatin to the 2 mAb combination increased overall survival >90 days (and was probably curative) by a mechanism which included a systemic CD8(+) T cell response with tumor specificity and immunological memory. Strikingly, combined treatment of cisplatin and CD137/PD-1 mAb also gave rise to the long-term survival of mice with established TC1 lung tumors. A similar combination of the 2 mAbs and cisplatin should be considered for clinical ‘translation’.
in vivo activation of 4-1BB
Guo, Z., et al. (2013). "Combined TIM-3 blockade and CD137 activation affords the long-term protection in a murine model of ovarian cancer" J Transl Med 11: 215. PubMed
BACKGROUND: T-cell immunoglobulin and mucin domain 3 (TIM-3) is known as a negative immune regulator and emerging data have implicated TIM-3 a pivotal role in suppressing antitumor immunity. The co-stimulatory receptor CD137 is transiently upregulated on T-cells following activation and increases their proliferation and survival when engaged. Although antagonistic anti-TIM-3 or agonistic anti-CD137 antibodies can promote the rejection of several murine tumors, some poorly immunogenic tumors were refractory to this treatment. In this study, we sought to evaluate whether combined TIM-3 blockade and CD137 activation would significantly improve the immunotherapy in the murine ID8 ovarian cancer model. METHODS: Mice with established ID8 tumor were intraperitoneally injected with single or combined anti-TIM-3/CD137 monoclonal antibody (mAb); mice survival was recorded, the composition and gene expression of tumor-infiltrating immune cells in these mice was analyzed by flow cytometry and quantitative RT-PCR respectively, and the function of CD8(+) cells was evaluated by ELISA and cytotoxicity assay. RESULTS: Either anti-TIM-3 or CD137 mAb alone, although effective in 3 days established tumor, was unable to prevent tumor progression in mice bearing 10 days established tumor, however, combined anti-TIM-3/CD137 mAb significantly inhibited the growth of these tumors with 60% of mice tumor free 90 days after tumor inoculation. Therapeutic efficacy was associated with a systemic immune response with memory and antigen specificity, required CD4(+) cells and CD8(+) cells. The 2 mAb combination increased CD4(+) and CD8(+) cells and decreased immunosuppressive CD4(+)FoxP3(+) regulatory T (Treg) cells and CD11b(+)Gr-1(+) myeloid suppressor cells (MDSC) at tumor sites, giving rise to significantly elevated ratios of CD4(+) and CD8(+) cells to Treg and MDSC; This is consistent with biasing local immune response towards an immunostimulatory Th1 type and is further supported by quantitative RT-PCR data showing the increased Th1-associated genes by anti-TIM-3/CD137 treatment. The increased CD8(+) T cells produced high level of IFN-gamma upon tumor antigen stimulation and displayed antigen-specific cytotoxic activity. CONCLUSIONS: To our knowledge, this is the first report investigating the effects of anti-TIM-3/CD137 combined mAb in a murine ovarian cancer model, and our results may aid the design of future trials for ovarian cancer immunotherapy.
in vivo activation of 4-1BB
Kwong, B., et al. (2013). "Localized immunotherapy via liposome-anchored Anti-CD137 + IL-2 prevents lethal toxicity and elicits local and systemic antitumor immunity" Cancer Res 73(5): 1547-1558. PubMed
Immunostimulatory agonists such as anti-CD137 and interleukin (IL)-2 have elicited potent antitumor immune responses in preclinical studies, but their clinical use is limited by inflammatory toxicities that result upon systemic administration. We hypothesized that by rigorously restricting the biodistribution of immunotherapeutic agents to a locally accessible lesion and draining lymph node(s), effective local and systemic antitumor immunity could be achieved in the absence of systemic toxicity. We anchored anti-CD137 and an engineered IL-2Fc fusion protein to the surfaces of PEGylated liposomes, whose physical size permitted dissemination in the tumor parenchyma and tumor-draining lymph nodes but blocked entry into the systemic circulation following intratumoral injection. In the B16F10 melanoma model, intratumoral liposome-coupled anti-CD137 + IL-2Fc therapy cured a majority of established primary tumors while avoiding the lethal inflammatory toxicities caused by equivalent intratumoral doses of soluble immunotherapy. Immunoliposome therapy induced protective antitumor memory and elicited systemic antitumor immunity that significantly inhibited the growth of simultaneously established distal tumors. Tumor inhibition was CD8(+) T-cell-dependent and was associated with increased CD8(+) T-cell infiltration in both treated and distal tumors, enhanced activation of tumor antigen-specific T cells in draining lymph nodes, and a reduction in regulatory T cells in treated tumors. These data suggest that local nanoparticle-anchored delivery of immuno-agonists represents a promising strategy to improve the therapeutic window and clinical applicability of highly potent but otherwise intolerable regimens of cancer immunotherapy. Cancer Res; 73(5); 1547-58. (c)2012 AACR.
in vivo activation of 4-1BB
Dai, M., et al. (2013). "Long-lasting complete regression of established mouse tumors by counteracting Th2 inflammation" J Immunother 36(4): 248-257. PubMed
40% of mice with SW1 tumors remained healthy >150 days after last treatment and are probably cured. Therapeutic efficacy was associated with a systemic immune response with memory and antigen specificity, required CD4 cells and involved CD8 cells and NK cells to a less extent. The 3 mAb combination significantly decreased CD19 cells at tumor sites, increased IFN-gamma and TNF-alpha producing CD4 and CD8 T cells and mature CD86 dendritic cells (DC), and it increased the ratios of effector CD4 and CD8 T cells to CD4Foxp3 regulatory T (Treg) cells and to CD11bGr-1 myeloid suppressor cells (MDSC). This is consistent with shifting the tumor microenvironment from an immunosuppressive Th2 to an immunostimulatory Th1 type and is further supported by PCR data. Adding an anti-CD19 mAb to the 3 mAb combination in the SW1 model further increased therapeutic efficacy. Data from ongoing experiments show that intratumoral injection of a combination of mAbs to CD137PD-1CTLA4CD19 can induce complete regression and dramatically prolong survival also in the TC1 carcinoma and B16 melanoma models, suggesting that the approach has general validity.”}” data-sheets-userformat=”{“2″:14851,”3”:{“1″:0},”4”:{“1″:2,”2″:16777215},”12″:0,”14”:{“1″:2,”2″:1521491},”15″:”Roboto, sans-serif”,”16″:12}”>Mice with intraperitoneal ID8 ovarian carcinoma or subcutaneous SW1 melanoma were injected with monoclonal antibodies (mAbs) to CD137PD-1CTLA4 7-15 days after tumor initiation. Survival of mice with ID8 tumors tripled and >40% of mice with SW1 tumors remained healthy >150 days after last treatment and are probably cured. Therapeutic efficacy was associated with a systemic immune response with memory and antigen specificity, required CD4 cells and involved CD8 cells and NK cells to a less extent. The 3 mAb combination significantly decreased CD19 cells at tumor sites, increased IFN-gamma and TNF-alpha producing CD4 and CD8 T cells and mature CD86 dendritic cells (DC), and it increased the ratios of effector CD4 and CD8 T cells to CD4Foxp3 regulatory T (Treg) cells and to CD11bGr-1 myeloid suppressor cells (MDSC). This is consistent with shifting the tumor microenvironment from an immunosuppressive Th2 to an immunostimulatory Th1 type and is further supported by PCR data. Adding an anti-CD19 mAb to the 3 mAb combination in the SW1 model further increased therapeutic efficacy. Data from ongoing experiments show that intratumoral injection of a combination of mAbs to CD137PD-1CTLA4CD19 can induce complete regression and dramatically prolong survival also in the TC1 carcinoma and B16 melanoma models, suggesting that the approach has general validity.
in vivo activation of 4-1BB
Curran, M. A., et al. (2011). "Combination CTLA-4 blockade and 4-1BB activation enhances tumor rejection by increasing T-cell infiltration, proliferation, and cytokine production" PLoS One 6(4): e19499. PubMed
BACKGROUND: The co-inhibitory receptor Cytotoxic T-Lymphocyte Antigen 4 (CTLA-4) attenuates immune responses and prevent autoimmunity, however, tumors exploit this pathway to evade the host T-cell response. The T-cell co-stimulatory receptor 4-1BB is transiently upregulated on T-cells following activation and increases their proliferation and inflammatory cytokine production when engaged. Antibodies which block CTLA-4 or which activate 4-1BB can promote the rejection of some murine tumors, but fail to cure poorly immunogenic tumors like B16 melanoma as single agents. METHODOLOGY/PRINCIPAL FINDINGS: We find that combining alphaCTLA-4 and alpha4-1BB antibodies in the context of a Flt3-ligand, but not a GM-CSF, based B16 melanoma vaccine promoted synergistic levels of tumor rejection. 4-1BB activation elicited strong infiltration of CD8+ T-cells into the tumor and drove the proliferation of these cells, while CTLA-4 blockade did the same for CD4+ effector T-cells. Anti-4-1BB also depressed regulatory T-cell infiltration of tumors. 4-1BB activation strongly stimulated inflammatory cytokine production in the vaccine and tumor draining lymph nodes and in the tumor itself. The addition of CTLA-4 blockade further increased IFN-gamma production from CD4+ effector T-cells in the vaccine draining node and the tumor. Anti 4-1BB treatment, with or without CTLA-4 blockade, induced approximately 75% of CD8+ and 45% of CD4+ effector T-cells in the tumor to express the killer cell lectin-like receptor G1 (KLRG1). Tumors treated with combination antibody therapy showed 1.7-fold greater infiltration by these KLRG1+CD4+ effector T-cells than did those treated with alpha4-1BB alone. CONCLUSIONS/SIGNIFICANCE: This study shows that combining T-cell co-inhibitory blockade with alphaCTLA-4 and active co-stimulation with alpha4-1BB promotes rejection of B16 melanoma in the context of a suitable vaccine. In addition, we identify KLRG1 as a useful marker for monitoring the anti-tumor immune response elicited by this therapy. These findings should aid in the design of future trials for the immunotherapy of melanoma.
in vivo activation of 4-1BB
Taraban, V. Y., et al. (2002). "Expression and costimulatory effects of the TNF receptor superfamily members CD134 (OX40) and CD137 (4-1BB), and their role in the generation of anti-tumor immune responses" Eur J Immunol 32(12): 3617-3627. PubMed
This study addresses the relative importance of CD134 (OX40) and CD137 (4-1BB) in the costimulation of CD4+ and CD8+ T cells under comparable conditions of antigenic stimulation. We demonstrate that CD134 is capable of directly costimulating CD8+ T cells. However, costimulation of CD8+ T cells by CD134 is less potent than that triggered by CD137. The higher costimulatory activity of CD137, when compared with CD134, correlates well with its faster expression kinetics and higher levels on CD8+ T cells. Furthermore, induction of CD137 expression on CD8+ T cells is highly sensitive to low levels of TCR stimulation, which is in contrast with CD134. Conversely, CD134 is more effective than CD137 in costimulating CD4+ T cells. This, however, could not be attributed to differential expression. We also demonstrate that the transient nature of CD134 and CD137 expression on activated CD4+ T cells is the resultof proteolytic shedding. Consistent with the greater ability of CD137 to costimulate CD8+ T cells, stimulation of CD137 in vivo is considerably more effective than CD134 in augmenting anti-tumor immune responses. Therefore, agents that stimulate signaling via CD137 are likely to be more useful in clinical conditions where highly effective CD8+ CTL responses are required.
- Cancer Research,
- Immunology and Microbiology
A Novel MHC-Independent Mechanism of Tumor Cell Killing by CD8+T Cells
Preprint on BioRxiv : the Preprint Server for Biology on 3 February 2023 by Lerner, E., Woroniecka, K., et al.
PubMed
The accepted paradigm for both cellular and antitumor immunity relies upon tumor cell kill by CD8 + T cells recognizing cognate antigens presented in the context of target cell major histocompatibility complex class I (MHC I) molecules. Likewise, a classically described mechanism of tumor immune escape is tumor MHC-I downregulation. Here, we report that CD8 + T cells maintain the capacity to kill tumor cells that are entirely devoid of MHC-I expression. This capacity proves to be dependent on interactions between T cell NKG2D and tumor NKG2D ligands (NKG2DL). Necessarily, tumor cell kill in these instances is antigen-independent, although prior T cell antigen-specific activation is required and can be furnished by myeloid cells or even neighboring MHC-replete tumors cells. These mechanisms are active in vivo in mice, as well as in vitro in human tumor systems, and are obviated by NKG2D knockout or blockade. Tumor cell killing following T cell NKG2D engagement is Fas-independent and appears to involve granzyme. These studies potentially obviate the long-advanced notion that downregulation of MHC-I is a viable means of tumor immune escape, and instead identify the NKG2D/NKG2DL axis as a novel therapeutic target for enhancing T cell-dependent anti-tumor immunity against MHC loss variants.
- Cancer Research,
- Immunology and Microbiology,
- Mus musculus (House mouse)
Targeting T cell checkpoints 41BB and LAG3 and myeloid cell CXCR1/CXCR2 results in antitumor immunity and durable response in pancreatic cancer.
In Nature Cancer on 1 January 2023 by Gulhati, P., Schalck, A., et al.
PubMed
Pancreatic ductal adenocarcinoma (PDAC) is considered non-immunogenic, with trials showing its recalcitrance to PD1 and CTLA4 immune checkpoint therapies (ICTs). Here, we sought to systematically characterize the mechanisms underlying de novo ICT resistance and to identify effective therapeutic options for PDAC. We report that agonist 41BB and antagonist LAG3 ICT alone and in combination, increased survival and antitumor immunity, characterized by modulating T cell subsets with antitumor activity, increased T cell clonality and diversification, decreased immunosuppressive myeloid cells and increased antigen presentation/decreased immunosuppressive capability of myeloid cells. Translational analyses confirmed the expression of 41BB and LAG3 in human PDAC. Since single and dual ICTs were not curative, T cell-activating ICTs were combined with a CXCR1/2 inhibitor targeting immunosuppressive myeloid cells. Triple therapy resulted in durable complete responses. Given similar profiles in human PDAC and the availability of these agents for clinical testing, our findings provide a testable hypothesis for this lethal disease. © 2022. The Author(s), under exclusive licence to Springer Nature America, Inc.
- Cancer Research,
- Immunology and Microbiology
The loss of epithelial Smad4 drives immune evasion via CXCL1 while displaying vulnerability to combinatorial immunotherapy in gastric cancer.
In Cell Reports on 27 December 2022 by An, H. W., Seok, S. H., et al.
PubMed
SMAD4 is frequently mutated and inactivated in human gastric cancer (GC). Although the epithelial cell-autonomous functions of Smad4 have been extensively studied, its contribution to tumor immunity is largely undetermined. Here, we report that the loss of Smad4 expression in GC cells endows them with the ability to evade tumor immunity. Unlike their Smad4-proficient counterparts, Smad4-deficient stomach organoids can evade host immunity to form tumors in immunocompetent mice. Smad4-deficient GC cells show expanded CD133+ cancer stem-like cells while suppressing dendritic cell (DC) differentiation and cytotoxic T cells with granulocytic myeloid-derived suppressor cell (G-MDSC) accumulation through a secretome containing CXCL1. Moreover, Smad4 deficiency increases programmed cell death ligand-1 (PD-L1) and decreases 4-1BBL expressions, indicating a change in immunogenicity. Combinatorial immune checkpoint blockade (ICB) of anti-PD-L1 and anti-CTLA-4 or agonistic anti-4-1BB antibodies effectively treats ICB monotherapy-resistant Smad4-deficient allografts, exposing a specific vulnerability. Collectively, these data provide a rational basis for ICB strategies in treating advanced GC with Smad4 deficiency. Copyright © 2022 The Authors. Published by Elsevier Inc. All rights reserved.
- In Vivo,
- Mus musculus (House mouse),
- Cancer Research
Elimination of acquired resistance to PD-1 blockade via the concurrent depletion of tumour cells and immunosuppressive cells.
In Nature Biomedical Engineering on 1 November 2021 by Xue, G., Wang, Z., et al.
PubMed
Antigen release resulting from the death of tumour cells induced by chemotherapies and targeted therapies can augment the antitumour responses induced by immune checkpoint blockade (ICB). However, tumours responding to ICB therapies often become resistant to them. Here we show that the specific targeting of tumour cells promotes the growth of tumour-cell variants that are resistant to ICB, and that the acquired resistance can be overcome via the concurrent depletion of tumour cells and of major types of immunosuppressive cell via a monoclonal antibody binding the enzyme CD73, which we identified as highly expressed on tumour cells and on regulatory T cells, myeloid-derived suppressor cells and tumour-associated macrophages, but not on cytolytic T lymphocytes, natural killer cells and dendritic cells. In mice with murine tumours, the systemic administration of anti-PD1 antibodies and anti-CD73 antibodies conjugated to a near-infrared dye prevented near-infrared-irradiated tumours from acquiring resistance to ICB and resulted in the eradication of advanced tumours. The elimination of immunosuppressive cells may overcome acquired resistance to ICB across a range of tumour types and combination therapies. © 2021. The Author(s), under exclusive licence to Springer Nature Limited.
- Cancer Research,
- Immunology and Microbiology
Tumors establish resistance to immunotherapy by regulating Treg recruitment via CCR4.
In Journal for Immunotherapy of Cancer on 1 November 2020 by Marshall, L. A., Marubayashi, S., et al.
PubMed
Checkpoint inhibitors (CPIs) such as anti-PD(L)-1 and anti-CTLA-4 antibodies have resulted in unprecedented rates of antitumor responses and extension of survival of patients with a variety of cancers. But some patients fail to respond or initially respond but later relapse as they develop resistance to immune therapy. One of the tumor-extrinsic mechanisms for resistance to immune therapy is the accumulation of regulatory T cells (Treg) in tumors. In preclinical and clinical studies, it has been suggested that tumor trafficking of Treg is mediated by CC chemokine receptor 4 (CCR4). Over 90% of human Treg express CCR4 and migrate toward CCL17 and CCL22, two major CCR4 ligands that are either high at baseline or upregulated in tumors on CPI treatment. Hence, CCR4 antagonism has the potential to be an effective antitumor treatment by reducing the accumulation of Treg into the tumor microenvironment (TME). We developed in vitro and in vivo models to assess Treg migration and antitumor efficacy using a potent and selective CCR4 antagonist, CCR4-351. We used two separate tumor models, Pan02 and CT26 mouse tumors, that have high and low CCR4 ligand expression, respectively. Tumor growth inhibition as well as the frequency of tumor-infiltrating Treg and effector T cells was assessed following the treatment with CCR4 antagonist alone or in combination with CPI. Using a selective and highly potent, novel small molecule inhibitor of CCR4, we demonstrate that migration of CCR4+ Treg into the tumor drives tumor progression and resistance to CPI treatment. In tumor models with high baseline levels of CCR4 ligands, blockade of CCR4 reduced the number of Treg and enhanced antitumor immune activity. Notably, in tumor models with low baseline level of CCR4 ligands, treatment with immune CPIs resulted in significant increases of CCR4 ligands and Treg numbers. Inhibition of CCR4 reduced Treg frequency and potentiated the antitumor effects of CPIs. Taken together, we demonstrate that CCR4-dependent Treg recruitment into the tumor is an important tumor-extrinsic mechanism for immune resistance. Blockade of CCR4 led to reduced frequency of Treg and resulted in increased antitumor activity, supporting the clinical development of CCR4 inhibitors in combination with CPI for the treatment of cancer. CPI upregulates CCL17 and CCL22 expression in tumors and increases Treg migration into the TME. Pharmacological antagonism of the CCR4 receptor effectively inhibits Treg recruitment and results in enhanced antitumor efficacy either as single agent in CCR4 ligandhigh tumors or in combination with CPIs in CCR4 ligandlow tumors. © Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
- In Vivo,
- Mus musculus (House mouse)
Trispecific natural killer cell nanoengagers for targeted chemoimmunotherapy.
In Science Advances on 1 July 2020 by Au, K. M., Park, S. I., et al.
PubMed
Activation of the innate immune system and natural killer (NK) cells has been a key effort in cancer immunotherapy research. Here, we report a nanoparticle-based trispecific NK cell engager (nano-TriNKE) platform that can target epidermal growth factor receptor (EGFR)-overexpressing tumors and promote the recruitment and activation of NK cells to eradicate these cancer cells. Moreover, the nanoengagers can deliver cytotoxic chemotherapeutics to further improve their therapeutic efficacy. We have demonstrated that effective NK cell activation can be achieved by the spatiotemporal coactivation of CD16 and 4-1BB stimulatory molecules on NK cells with nanoengagers, and the nanoengagers are more effective than free antibodies. We also show that biological targeting, either through radiotherapy or EGFR, is critical to the therapeutic effects of nanoengagers. Last, EGFR-targeted nanoengagers can augment both NK-activating agents and chemotherapy (epirubicin) as highly effective anticancer agents, providing robust chemoimmunotherapy. Copyright © 2020 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works. Distributed under a Creative Commons Attribution NonCommercial License 4.0 (CC BY-NC).
A PSMA-Targeting CD3 Bispecific Antibody Induces Antitumor Responses that Are Enhanced by 4-1BB Costimulation.
In Cancer Immunology Research on 1 May 2020 by Chiu, D., Tavaré, R., et al.
PubMed
Patients with hematologic cancers have improved outcomes after treatment with bispecific antibodies that bind to CD3 on T cells and that redirect T cells toward cancer cells. However, clinical benefit against solid tumors remains to be shown. We made a bispecific antibody that targets both the common prostate tumor-specific antigen PSMA and CD3 (PMSAxCD3) and provide evidence for tumor inhibition in several preclinical solid tumor models. Mice expressing the human extracellular regions of CD3 and PSMA were generated to examine antitumor efficacy in the presence of an intact immune system and PSMA expression in normal tissues. PSMAxCD3 accumulated in PSMA-expressing tissues and tumors as detected by immuno-PET imaging. Although PSMAxCD3 induced T-cell activation and showed antitumor efficacy in mice with low tumor burden, PSMAxCD3 lost efficacy against larger solid tumors, mirroring the difficulty of treating solid tumors in the clinic. Costimulatory receptors can enhance T-cell responses. We show here that costimulation can enhance the antitumor efficacy of PSMAxCD3. In particular, 4-1BB stimulation in combination with PSMAxCD3 enhanced T-cell activation and proliferation, boosted efficacy against larger tumors, and induced T-cell memory, leading to durable antitumor responses. The combination of CD3 bispecific antibodies and anti-4-1BB costimulation represents a therapeutic approach for the treatment of solid tumors. ©2020 American Association for Cancer Research.
- Cancer Research
4-1BB Agonism Averts TIL Exhaustion and Licenses PD-1 Blockade in Glioblastoma and Other Intracranial Cancers.
In Clinical Cancer Research on 15 March 2020 by Woroniecka, K. I., Rhodin, K. E., et al.
PubMed
The success of checkpoint blockade against glioblastoma (GBM) has been disappointing. Anti-PD-1 strategies may be hampered by severe T-cell exhaustion. We sought to develop a strategy that might license new efficacy for checkpoint blockade in GBM. We characterized 4-1BB expression in tumor-infiltrating lymphocytes (TIL) from human GBM. We implanted murine tumor models including glioma (CT2A), melanoma (B16), breast (E0771), and lung carcinomas intracranially and subcutaneously, characterized 4-1BB expression, and tested checkpoint blockade strategies in vivo. Our data reveal that 4-1BB is frequently present on nonexhausted CD8+ TILs in human and murine GBM. In murine gliomas, 4-1BB agonism and PD-1 blockade demonstrate a synergistic survival benefit in a CD8+ T-cell-dependent manner. The combination decreases TIL exhaustion and improves TIL functionality. This strategy proves most successful against intracranial CT2A gliomas. Efficacy in all instances correlates with the levels of 4-1BB expression on CD8+ TILs, rather than with histology or with intracranial versus subcutaneous tumor location. Proffering 4-1BB expression to T cells licenses combination 4-1BB agonism and PD-1 blockade in models where TIL 4-1BB levels had previously been low and the treatment ineffective. Although poor T-cell activation and severe T-cell exhaustion appear to be limiting factors for checkpoint blockade in GBM, 4-1BB agonism obviates these limitations and produces long-term survival when combined with anti-PD-1 therapy. Furthermore, this combination therapy is limited by TIL 4-1BB expression, but not by the intracranial compartment, and therefore may be particularly well-suited to GBM. ©2019 American Association for Cancer Research.
- Cancer Research
VEGF-C-driven lymphatic drainage enables immunosurveillance of brain tumours.
In Nature on 1 January 2020 by Song, E., Mao, T., et al.
PubMed
Immune surveillance against pathogens and tumours in the central nervous system is thought to be limited owing to the lack of lymphatic drainage. However, the characterization of the meningeal lymphatic network has shed light on previously unappreciated ways that an immune response can be elicited to antigens that are expressed in the brain1-3. Despite progress in our understanding of the development and structure of the meningeal lymphatic system, the contribution of this network in evoking a protective antigen-specific immune response in the brain remains unclear. Here, using a mouse model of glioblastoma, we show that the meningeal lymphatic vasculature can be manipulated to mount better immune responses against brain tumours. The immunity that is mediated by CD8 T cells to the glioblastoma antigen is very limited when the tumour is confined to the central nervous system, resulting in uncontrolled tumour growth. However, ectopic expression of vascular endothelial growth factor C (VEGF-C) promotes enhanced priming of CD8 T cells in the draining deep cervical lymph nodes, migration of CD8 T cells into the tumour, rapid clearance of the glioblastoma and a long-lasting antitumour memory response. Furthermore, transfection of an mRNA construct that expresses VEGF-C works synergistically with checkpoint blockade therapy to eradicate existing glioblastoma. These results reveal the capacity of VEGF-C to promote immune surveillance of tumours, and suggest a new therapeutic approach to treat brain tumours.
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Domatinostat favors the immunotherapy response by modulating the tumor immune microenvironment (TIME).
In Journal for Immunotherapy of Cancer on 8 November 2019 by Bretz, A. C., Parnitzke, U., et al.
PubMed
The efficacy of PD-(L)1 blockade depends on the composition of the tumor immune microenvironment (TIME) and is generally higher in tumors with pre-existing cytotoxic T cells (CTL) than in those with low CTL numbers. Nonetheless, a significant proportion of patients with pre-existing immunity fail to respond, indicating a therapeutic potential for combining PD-(L)1 blockade with additional immunomodulatory agents in both CTL-high and -low immune phenotypes. Here, we evaluated domatinostat (4SC-202), a class I-selective histone deacetylase (HDAC) inhibitor, for its effect on the TIME and its antitumoral efficacy using syngeneic mouse models with CTL-high or CTL-low tumors. Domatinostat was evaluated in PD-1 blockade-insensitive CTL-low (CT26) and CTL-high (C38) syngeneic models alone and in combination with different immune-inhibitory and -stimulatory approaches. Effects on the immunophenotype were assessed via flow cytometry and RNA-seq analyses. The changes in RNA-seq-based immune signatures determined in a murine setting were investigated in patient samples from the first-dose cohort of the SENSITIZE trial (NCT03278665) evaluating domatinostat combined with pembrolizumab in advanced-stage melanoma patients refractory/nonresponding to PD-1 blockade. Domatinostat increased the expression of antigen-presenting machinery (APM) genes and MHC class I and II molecules, along with CTL infiltration, in tumors of both immune phenotypes. In combination with PD-(L)1 blockade, domatinostat augmented antitumor effects substantially above the effects of single-agent therapies, displaying greater benefit in tumors with pre-existing CTLs. In this setting, the combination of domatinostat with agonistic anti-4-1BB or both PD-1 and LAG3 blockade further increased the antitumor efficacy. In CTL-low tumors, domatinostat enhanced the expression of genes known to reinforce immune responses against tumors. Specifically, domatinostat increased the expression of Ifng and genes associated with responses to pembrolizumab and nivolumab. Clinically, these findings were confirmed in patients with advanced melanoma treated with domatinostat for 14 days, who demonstrated elevated expression of APM and MHC genes, the IFNG gene, and the IFN-γ and pembrolizumab response signatures in individual tumor samples. In summary, these data suggest a promising potential of domatinostat in combination with immunotherapy to improve the outcome of refractory cancer patients.
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Immunotherapy with Monoclonal Antibodies in Lung Cancer of Mice: Oxidative Stress and Other Biological Events.
In Cancers on 4 September 2019 by Tang, J., Ramis-Cabrer, D., et al.
PubMed
Background: Lung cancer (LC) is a major leading cause of death worldwide. Immunomodulators that target several immune mechanisms have proven to reduce tumor burden in experimental models through induction of the immune microenvironment. We hypothesized that other biological mechanisms may also favor tumor burden reduction in lung cancer-bearing mice treated with immunomodulators. Methods: Tumor weight, area, T cells and tumor growth (immunohistochemistry), oxidative stress, apoptosis, autophagy, and signaling (NF-κB and sirtuin-1) markers were analyzed (immunoblotting) in subcutaneous tumor of BALB/c mice injected with LP07 adenocarcinoma cells treated with monoclonal antibodies (CD-137, CTLA-4, PD-1, and CD-19, N = 9/group) and non-treated control animals. Results: Compared to non-treated cancer mice, in tumors of monoclonal-treated animals, tumor area and weight and ki-67 were significantly reduced, while T cell counts, oxidative stress, apoptosis, autophagy, activated p65, and sirtuin-1 markers were increased. Conclusions: Immunomodulators elicited a reduction in tumor burden (reduced tumor size and weight) through decreased tumor proliferation and increased oxidative stress, apoptosis, autophagy, and signaling markers, which may have interfered with the immune profile of the tumor microenvironment. Future research should be devoted to the elucidation of the specific contribution of each biological mechanism to the reduced tumor burden.
- Cancer Research,
- Endocrinology and Physiology
Reduced lung cancer burden by selective immunomodulators elicits improvements in muscle proteolysis and strength in cachectic mice.
In Journal of Cellular Physiology on 1 August 2019 by Salazar-Degracia, A., Granado-Martínez, P., et al.
PubMed
Identification of to what extent tumor burden influences muscle mass independently of specific treatments for cancer-cachexia remains to be elucidated. We hypothesized that reduced tumor burden by selective treatment of tumor with immunomodulators may exert beneficial effects on muscle wasting and function in mice. Body and muscle weight, grip strength, physical activity, muscle morphometry, apoptotic nuclei, troponin-I systemic levels, interleukin-6, proteolytic markers, and tyrosine release, and apoptosis markers were determined in diaphragm and gastrocnemius muscles of lung cancer (LP07 adenocarcinoma cells) mice (BALB/c) treated with monoclonal antibodies (mAbs), against immune check-points and pathways (CD-137, cytotoxic T-lymphocyte associated protein-4, programed cell death-1, and CD-19; N = 10/group). Nontreated lung cancer cachectic mice were the controls. T and B cell numbers and macrophages were counted in tumors of both mouse groups. Compared to nontreated cachectic mice, in the mAbs-treated animals, T cells increased, no differences in B cells or macrophages, the variables final body weight, body weight and grip strength gains significantly improved. In diaphragm and gastrocnemius of mAbs-treated cachectic mice, number of apoptotic nuclei, tyrosine release, proteolysis, and apoptosis markers significantly decreased compared to nontreated cachectic mice. Systemic levels of troponin-I significantly decreased in treated cachectic mice compared to nontreated animals. We conclude that reduced tumor burden as a result of selective treatment of the lung cancer cells with immunomodulators elicits per se beneficial effects on muscle mass loss through attenuation of several biological mechanisms that lead to increased protein breakdown and apoptosis, which translated into significant improvements in limb muscle strength but not in physical activity parameters. © 2019 Wiley Periodicals, Inc.
- Biochemistry and Molecular biology,
- Cancer Research,
- Cell Biology,
- Immunology and Microbiology
Molecular and metabolic pathways mediating curative treatment of a non-Hodgkin B cell lymphoma by Sindbis viral vectors and anti-4-1BB monoclonal antibody.
In Journal for Immunotherapy of Cancer on 15 July 2019 by Yu, M., Scherwitzl, I., et al.
PubMed
Limitations to current therapies for treating non-Hodgkin B cell lymphoma include relapse, toxicity and high cost. Thus, there remains a need for novel therapies. Oncolytic viral (OV) therapy has become a promising cancer immunotherapy because of its potential effectiveness, specificity and long-lasting immunity. We describe and characterize a novel cancer immunotherapy combining Sindbis virus (SV) vectors and the agonistic monoclonal antibody (mAb) to the T cell costimulatory receptor, 4-1BB (CD137). A20 lymphoma was transfected with luciferase and tumor cells were inoculated to BALB/c mice. Tumor growth was monitored by IVIS imaging. Tumor bearing mice were treated with Sindbis virus, α4-1BB Ab or SV plus α4-1BB Ab. On day 7 after treatment, splenocytes were harvested and surface markers, cytokines, and transcription factors were measured by flow cytometry or Elispot. Splenic T cells were isolated and RNA transcriptome analysis was performed. Tumor cured mice were rechallenged with tumor for testing immunological memory. SV vectors in combination with α4-1BB monoclonal antibody (mAb) completely eradicated a B-cell lymphoma in a preclinical mouse model, a result that could not be achieved with either treatment alone. Tumor elimination involves a synergistic effect of the combination that significantly boosts T cell cytotoxicity, IFNγ production, T cell proliferation, migration, and glycolysis. In addition, all mice that survived after treatment developed long lasting antitumor immunity, as shown by the rejection of A20 tumor rechallenge. We identified the molecular pathways, including upregulated cytokines, chemokines and metabolic pathways in T cells that are triggered by the combined therapy and help to achieve a highly effective anti-tumor response. Our study provides a novel, alternative method for B cell lymphoma treatment and describes a rationale to help translate SV vectors plus agonistic mAb into clinical applications.
- Cancer Research
Oxidative Stress as a Mediator of the Immunomodulation Exerted by Monoclonal Antibodies in the Treatment of Lung Cancer in Mice
Preprint on Preprints.org on 10 June 2019 by Tang, J., Ramis-Cabrer, D., et al.
PubMed
h4>Background: /h4> Lung cancer (LC) is a major leading cause of death worldwide. Immunomodulators that target several immune mechanisms have proven to reduce tumor burden in experimental models through induction of the immune microenvironment. We hypothesized that other biological mechanisms may also favor tumor burden reduction in lung cancer-bearing mice treated with immunomodulators. h4>Methods:/h4> Tumor weight, area, and immune cells (T, B, macrophages, and TNF-alpha levels, immunohistochemistry) and tumor growth, oxidative stress, apoptosis, autophagy, and sirtuin-1 markers were analyzed (immunoblotting) in subcutaneous tumor of BALB/c mice injected with LP07 adenocarcinoma cells treated with monoclonal antibodies (CD-137, CTLA-4, PD-1, and CD-19, N=9/group) and non-treated control animals. h4>Results:/h4> Compared to non-treated cancer mice, in tumors of monoclonal-treated animals, tumor area and weight and ki-67 significantly reduced, while T cell counts, oxidative stress, apoptosis, autophagy, and sirtuin-1 marker increased. h4>Conclusion:/h4> Immunomodulators elicited a reduction in tumor burden (reduced tumor size and weight) through decreased tumor proliferation and increased oxidative stress, apoptosis, autophagy, and sirtuin-1 levels, which may have interfered with the immune profile of the tumor microenvironment. Future research should be devoted to the elucidation of the specific contribution of each biological mechanism to the reduced tumor burden.
- Cancer Research,
- Immunology and Microbiology
Nanoparticle Conjugation of Human Papillomavirus 16 E7-long Peptides Enhances Therapeutic Vaccine Efficacy against Solid Tumors in Mice.
In Cancer Immunology Research on 1 November 2018 by Galliverti, G., Tichet, M., et al.
PubMed
Treatment of patients bearing human papillomavirus (HPV)-related cancers with synthetic long-peptide (SLP) therapeutic vaccines has shown promising results in clinical trials against premalignant lesions, whereas responses against later stage carcinomas have remained elusive. We show that conjugation of a well-documented HPV-E7 SLP to ultra-small polymeric nanoparticles (NP) enhances the antitumor efficacy of therapeutic vaccination in different mouse models of HPV+ cancers. Immunization of TC-1 tumor-bearing mice with a single dose of NP-conjugated E7LP (NP-E7LP) generated a larger pool of E7-specific CD8+ T cells with increased effector functions than unconjugated free E7LP. At the tumor site, NP-E7LP prompted a robust infiltration of CD8+ T cells that was not accompanied by concomitant accumulation of regulatory T cells (Tregs), resulting in a higher CD8+ T-cell to Treg ratio. Consequently, the amplified immune response elicited by the NP-E7LP formulation led to increased regression of large, well-established tumors, resulting in a significant percentage of complete responses that were not achievable by immunizing with the non-NP-conjugated long-peptide. The partial responses were characterized by distinct phases of regression, stable disease, and relapse to progressive growth, establishing a platform to investigate adaptive resistance mechanisms. The efficacy of NP-E7LP could be further improved by therapeutic activation of the costimulatory receptor 4-1BB. This NP-E7LP formulation illustrates a "solid-phase" antigen delivery strategy that is more effective than a conventional free-peptide ("liquid") vaccine, further highlighting the potential of using such formulations for therapeutic vaccination against solid tumors. Cancer Immunol Res; 6(11); 1301-13. ©2018 AACR. ©2018 American Association for Cancer Research.
- Cancer Research
Tumor Regression and Cure Depends on Sustained Th1 Responses.
In Journal of Immunotherapy (Hagerstown, Md. : 1997) on 1 October 2018 by Dai, M., Hellstrom, I., et al.
PubMed
While immunomodulatory monoclonal antibodies (mAbs) have therapeutic efficacy against many tumors, few patients are cured. Attempting to improve their therapeutic efficacy we have applied the TC1 mouse lung carcinoma model and injected established subcutaneous tumors intratumorally with 3 weekly doses of various combinations of mAbs. Combinations of mAbs to CTLA4/PD1/CD137 (the 3 mAb combination) and to CTLA4/PD1/CD137/CD19 (the 4 mAb combination) were most efficacious to induce complete regression of both the injected tumor and an untreated tumor in the same mouse. Tumor cure was consistently associated with shifting a Th2 to a Th1 response in tumor-draining lymph nodes and spleen and it involved epitope specific and long-lived memory T cells as well as M1 macrophages. This shift and accompanying tumor rejection was harder to achieve as the treated tumors increased in size. Relapse of tumors which had initially regressed following treatment with immunomodulatory mAbs was associated with return of a Th2 microenvironment in tumors, tumor-draining lymph nodes and spleens rather than the emergence of immune-resistant tumor cells. While mAbs to CTLA4 plus PD-1 were therapeutically ineffective, combining the 2 of them with intraperitoneal cisplatin, 10 mg/kg, induced long-term complete tumor regression in most mice with small TC1 tumors and the therapeutic efficacy against larger tumors improved by administrating cisplatin together with the 3 or 4 mAb combination.
- Cancer Research,
- Immunology and Microbiology
Mucosal HPV E6/E7 Peptide Vaccination in Combination with Immune Checkpoint Modulation Induces Regression of HPV+ Oral Cancers.
In Cancer Research on 15 September 2018 by Dorta-Estremera, S., Chin, R. L., et al.
PubMed
High-risk human papillomavirus (HPV)-associated squamous cell carcinomas of the oropharynx (SCCOP) are among the fastest growing cancers. After standard-of-care treatment, however, patients with HPV+ SCCOP have better overall and disease-specific survival than patients with HPV- SCCOP, suggesting the importance of HPV-specific immunity. We reasoned that therapeutic vaccination targeting the HPV-16 E6 and E7 oncogenes could elicit high-affinity, high-frequency tumor antigen-specific T-cell responses, which could then be augmented and shielded from suppression in the tumor microenvironment by immune checkpoint modulation. In this study, we used a preclinical syngeneic mouse model of oral cancer comprised of mouse tonsil-derived epithelial cells stably expressing HPV-16 E6 and E7 genes along with H-ras oncogene (mEER) to identify combinations of vaccination and checkpoint antibodies capable of promoting tumor regression. Intranasal HPV E6/E7 peptide vaccination and single checkpoint antibodies failed to elicit responses in more than half of animals; however, 4-1BB agonist antibody along with either CD40 agonist antibody or CTLA-4 blockade eliminated the majority of established mEER tumors. The combination of intranasal HPV peptide vaccine and α4-1BB and αCTLA-4 antibodies produced curative efficacy and a better safety profile against orally implanted mEER tumors. Correlates of protective immunity included enhanced intratumoral levels of CD8 T cells relative to immunosuppressive regulatory T cells and myeloid-derived suppressor cells. Overall, our results demonstrate combination vaccine-immunotherapy modalities as novel treatment options for HPV+ SCCOP.Significance: Combinations of vaccine and checkpoint modulation are effective and safe treatment options for HPV+ oral cancers. Cancer Res; 78(18); 5327-39. ©2018 AACR. ©2018 American Association for Cancer Research.
- Cancer Research,
- Immunology and Microbiology
Sequestration of T cells in bone marrow in the setting of glioblastoma and other intracranial tumors.
In Nature Medicine on 1 September 2018 by Chongsathidkiet, P., Jackson, C., et al.
PubMed
T cell dysfunction contributes to tumor immune escape in patients with cancer and is particularly severe amidst glioblastoma (GBM). Among other defects, T cell lymphopenia is characteristic, yet often attributed to treatment. We reveal that even treatment-naïve subjects and mice with GBM can harbor AIDS-level CD4 counts, as well as contracted, T cell-deficient lymphoid organs. Missing naïve T cells are instead found sequestered in large numbers in the bone marrow. This phenomenon characterizes not only GBM but a variety of other cancers, although only when tumors are introduced into the intracranial compartment. T cell sequestration is accompanied by tumor-imposed loss of S1P1 from the T cell surface and is reversible upon precluding S1P1 internalization. In murine models of GBM, hindering S1P1 internalization and reversing sequestration licenses T cell-activating therapies that were previously ineffective. Sequestration of T cells in bone marrow is therefore a tumor-adaptive mode of T cell dysfunction, whose reversal may constitute a promising immunotherapeutic adjunct.
- In Vitro,
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Biopolymers codelivering engineered T cells and STING agonists can eliminate heterogeneous tumors.
In The Journal of Clinical Investigation on 1 June 2017 by Smith, T. T., Moffett, H. F., et al.
PubMed
Therapies using T cells that are programmed to express chimeric antigen receptors (CAR T cells) consistently produce positive results in patients with hematologic malignancies. However, CAR T cell treatments are less effective in solid tumors for several reasons. First, lymphocytes do not efficiently target CAR T cells; second, solid tumors create an immunosuppressive microenvironment that inactivates T cell responses; and third, solid cancers are typified by phenotypic diversity and thus include cells that do not express proteins targeted by the engineered receptors, enabling the formation of escape variants that elude CAR T cell targeting. Here, we have tested implantable biopolymer devices that deliver CAR T cells directly to the surfaces of solid tumors, thereby exposing them to high concentrations of immune cells for a substantial time period. In immunocompetent orthotopic mouse models of pancreatic cancer and melanoma, we found that CAR T cells can migrate from biopolymer scaffolds and eradicate tumors more effectively than does systemic delivery of the same cells. We have also demonstrated that codelivery of stimulator of IFN genes (STING) agonists stimulates immune responses to eliminate tumor cells that are not recognized by the adoptively transferred lymphocytes. Thus, these devices may improve the effectiveness of CAR T cell therapy in solid tumors and help protect against the emergence of escape variants.
- Cancer Research
Curing mice with large tumors by locally delivering combinations of immunomodulatory antibodies.
In Clinical Cancer Research on 1 March 2015 by Dai, M., Yip, Y. Y., et al.
PubMed
Immunomodulatory mAbs can treat cancer, but cures are rare except for small tumors. Our objective was to explore whether the therapeutic window increases by combining mAbs with different modes of action and injecting them into tumors. Combinations of mAbs to CD137/PD-1/CTLA-4 or CD137/PD-1/CTLA-4/CD19 were administrated intratumorally to mice with syngeneic tumors (B16 and SW1 melanoma, TC1 lung carcinoma), including tumors with a mean surface of approximately 80 mm(2). Survival and tumor growth were assessed. Immunologic responses were evaluated using flow cytometry and qRT-PCR. More than 50% of tumor-bearing mice had complete regression and long-term survival after tumor injection with mAbs recognizing CD137/PD-1/CTLA-4/CD19 with similar responses in three models. Intratumoral injection was more efficacious than intraperitoneal injection in causing rejection also of untreated tumors in the same mice. The three-mAb combination could also induce regression, but was less efficacious. There were few side effects, and therapy-resistant tumors were not observed. Transplanted tumor cells rapidly caused a Th2 response with increased CD19 cells. Successful therapy shifted this response to the Th1 phenotype with decreased CD19 cells and increased numbers of long-term memory CD8 effector cells and T cells making IFNγ and TNFα. Intratumoral injection of mAbs recognizing CD137/PD-1/CTLA-4/CD19 can eradicate established tumors and reverse a Th2 response with tumor-associated CD19 cells to Th1 immunity, whereas a combination lacking anti-CD19 is less effective. There are several human cancers for which a similar approach may provide clinical benefit. ©2014 American Association for Cancer Research.