InVivoMAb anti-mouse CD8α
Product Details
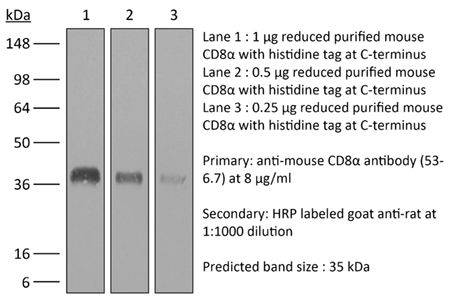
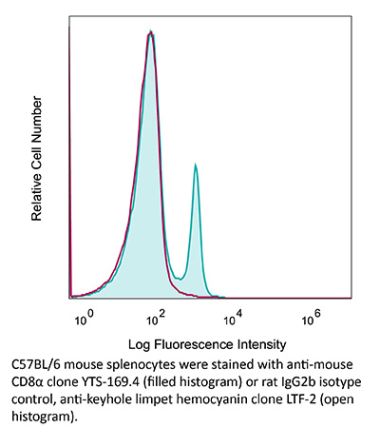
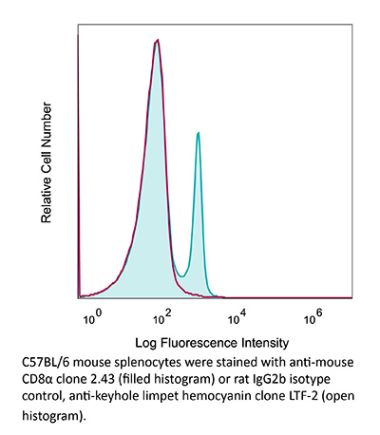
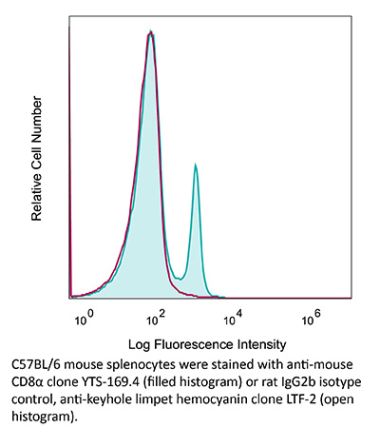
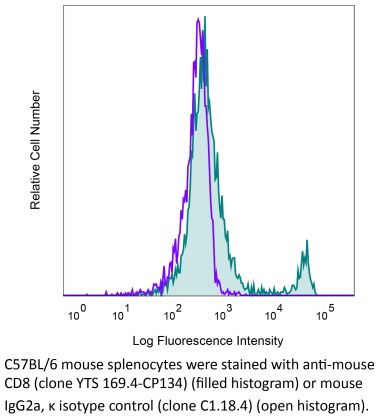
The 53-6.7 monoclonal antibody reacts with mouse CD8α. The CD8 antigen is a transmembrane glycoprotein that acts as a co-receptor for the T cell receptor (TCR). Like the TCR, CD8 binds to class I MHC molecules displayed by antigen presenting cells (APC). CD8 is primarily expressed on the surface of cytotoxic T cells, but can also be found on thymocytes, natural killer cells, and some dendritic cell subsets. CD8 most commonly exists as a heterodimer composed of one CD8α and one CD8β chain however, it can also exist as a homodimer composed of two CD8α chains. Both the CD8α and CD8β chains share significant homology to immunoglobulin variable light chains. The molecular weight of each CD8 chain is approximately 34 kDa. The 53-6.7 antibody exhibits depleting activity when used in vivo.Specifications
| Isotype | Rat IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 6.5 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Mouse Spleen Cells or Thymocyte Membranes |
| Reported Applications |
in vivo CD8+ T cell depletion Immunofluorescence Flow cytometry Western blot |
| Formulation |
PBS, pH 6.5 Contains no stabilizers or preservatives |
| Endotoxin |
<2EU/mg (<0.002EU/μg) Determined by LAL gel clotting assay |
| Purity |
>95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107671 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Additional Formats
Recommended Products
in vivo CD8+ T cell depletion, Flow Cytometry
Wang, W., et al. (2018). "RIP1 Kinase Drives Macrophage-Mediated Adaptive Immune Tolerance in Pancreatic Cancer" Cancer Cell 34(5): 757-774 e757. PubMed
Pancreatic ductal adenocarcinoma (PDA) is characterized by immune tolerance and immunotherapeutic resistance. We discovered upregulation of receptor-interacting serine/threonine protein kinase 1 (RIP1) in tumor-associated macrophages (TAMs) in PDA. To study its role in oncogenic progression, we developed a selective small-molecule RIP1 inhibitor with high in vivo exposure. Targeting RIP1 reprogrammed TAMs toward an MHCII(hi)TNFalpha(+)IFNgamma(+) immunogenic phenotype in a STAT1-dependent manner. RIP1 inhibition in TAMs resulted in cytotoxic T cell activation and T helper cell differentiation toward a mixed Th1/Th17 phenotype, leading to tumor immunity in mice and in organotypic models of human PDA. Targeting RIP1 synergized with PD1-and inducible co-stimulator-based immunotherapies. Tumor-promoting effects of RIP1 were independent of its co-association with RIP3. Collectively, our work describes RIP1 as a checkpoint kinase governing tumor immunity.
in vivo CD8+ T cell depletion
Christensen, A. D., et al. (2015). "Depletion of regulatory T cells in a hapten-induced inflammation model results in prolonged and increased inflammation driven by T cells" Clin Exp Immunol 179(3): 485-499. PubMed
Regulatory T cells (Tregs ) are known to play an immunosuppressive role in the response of contact hypersensitivity (CHS), but neither the dynamics of Tregs during the CHS response nor the exaggerated inflammatory response after depletion of Tregs has been characterized in detail. In this study we show that the number of Tregs in the challenged tissue peak at the same time as the ear-swelling reaches its maximum on day 1 after challenge, whereas the number of Tregs in the draining lymph nodes peaks at day 2. As expected, depletion of Tregs by injection of a monoclonal antibody to CD25 prior to sensitization led to a prolonged and sustained inflammatory response which was dependent upon CD8 T cells, and co-stimulatory blockade with cytotoxic T lymphocyte antigen-4-immunoglobulin (CTLA-4-Ig) suppressed the exaggerated inflammation. In contrast, blockade of the interleukin (IL)-10-receptor (IL-10R) did not further increase the exaggerated inflammatory response in the Treg -depleted mice. In the absence of Tregs , the response changed from a mainly acute reaction with heavy infiltration of neutrophils to a sustained response with more chronic characteristics (fewer neutrophils and dominated by macrophages). Furthermore, depletion of Tregs enhanced the release of cytokines and chemokines locally in the inflamed ear and augmented serum levels of the systemic inflammatory mediators serum amyloid (SAP) and haptoglobin early in the response.
Immunofluorescence
Finisguerra, V., et al. (2015). "MET is required for the recruitment of anti-tumoural neutrophils" Nature 522(7556): 349-353. PubMed
Mutations or amplification of the MET proto-oncogene are involved in the pathogenesis of several tumours, which rely on the constitutive engagement of this pathway for their growth and survival. However, MET is expressed not only by cancer cells but also by tumour-associated stromal cells, although its precise role in this compartment is not well characterized. Here we show that MET is required for neutrophil chemoattraction and cytotoxicity in response to its ligand hepatocyte growth factor (HGF). Met deletion in mouse neutrophils enhances tumour growth and metastasis. This phenotype correlates with reduced neutrophil infiltration to both the primary tumour and metastatic sites. Similarly, Met is necessary for neutrophil transudation during colitis, skin rash or peritonitis. Mechanistically, Met is induced by tumour-derived tumour necrosis factor (TNF)-alpha or other inflammatory stimuli in both mouse and human neutrophils. This induction is instrumental for neutrophil transmigration across an activated endothelium and for inducible nitric oxide synthase production upon HGF stimulation. Consequently, HGF/MET-dependent nitric oxide release by neutrophils promotes cancer cell killing, which abates tumour growth and metastasis. After systemic administration of a MET kinase inhibitor, we prove that the therapeutic benefit of MET targeting in cancer cells is partly countered by the pro-tumoural effect arising from MET blockade in neutrophils. Our work identifies an unprecedented role of MET in neutrophils, suggests a potential ‘Achilles’ heel’ of MET-targeted therapies in cancer, and supports the rationale for evaluating anti-MET drugs in certain inflammatory diseases.
in vivo CD8+ T cell depletion
Yamada, D. H., et al. (2015). "Suppression of Fcgamma-receptor-mediated antibody effector function during persistent viral infection" Immunity 42(2): 379-390. PubMed
Understanding how viruses subvert host immunity and persist is essential for developing strategies to eliminate infection. T cell exhaustion during chronic viral infection is well described, but effects on antibody-mediated effector activity are unclear. Herein, we show that increased amounts of immune complexes generated in mice persistently infected with lymphocytic choriomeningitis virus (LCMV) suppressed multiple Fcgamma-receptor (FcgammaR) functions. The high amounts of immune complexes suppressed antibody-mediated cell depletion, therapeutic antibody-killing of LCMV infected cells and human CD20-expressing tumors, as well as reduced immune complex-mediated cross-presentation to T cells. Suppression of FcgammaR activity was not due to inhibitory FcgammaRs or high concentrations of free antibody, and proper FcgammaR functions were restored when persistently infected mice specifically lacked immune complexes. Thus, we identify a mechanism of immunosuppression during viral persistence with implications for understanding effective antibody activity aimed at pathogen control.
in vivo CD8+ T cell depletion, Flow Cytometry
Walsh, K. B., et al. (2014). "Animal model of respiratory syncytial virus: CD8+ T cells cause a cytokine storm that is chemically tractable by sphingosine-1-phosphate 1 receptor agonist therapy" J Virol 88(11): 6281-6293. PubMed
The cytokine storm is an intensified, dysregulated, tissue-injurious inflammatory response driven by cytokine and immune cell components. The cytokine storm during influenza virus infection, whereby the amplified innate immune response is primarily responsible for pulmonary damage, has been well characterized. Now we describe a novel event where virus-specific T cells induce a cytokine storm. The paramyxovirus pneumonia virus of mice (PVM) is a model of human respiratory syncytial virus (hRSV). Unexpectedly, when C57BL/6 mice were infected with PVM, the innate inflammatory response was undetectable until day 5 postinfection, at which time CD8(+) T cells infiltrated into the lung, initiating a cytokine storm by their production of gamma interferon (IFN-gamma) and tumor necrosis factor alpha (TNF-alpha). Administration of an immunomodulatory sphingosine-1-phosphate (S1P) receptor 1 (S1P1R) agonist significantly inhibited PVM-elicited cytokine storm by blunting the PVM-specific CD8(+) T cell response, resulting in diminished pulmonary disease and enhanced survival. IMPORTANCE: A dysregulated overly exuberant immune response, termed a “cytokine storm,” accompanies virus-induced acute respiratory diseases (VARV), is primarily responsible for the accompanying high morbidity and mortality, and can be controlled therapeutically in influenza virus infection of mice and ferrets by administration of sphingosine-1-phosphate 1 receptor (S1P1R) agonists. Here, two novel findings are recorded. First, in contrast to influenza infection, where the cytokine storm is initiated early by the innate immune system, for pneumonia virus of mice (PVM), a model of RSV, the cytokine storm is initiated late in infection by the adaptive immune response: specifically, by virus-specific CD8 T cells via their release of IFN-gamma and TNF-alpha. Blockading these cytokines with neutralizing antibodies blunts the cytokine storm and protects the host. Second, PVM infection is controlled by administration of an S1P1R agonist.
in vivo CD8+ T cell depletion, Flow Cytometry
Uddin, M. N., et al. (2014). "TNF-alpha-dependent hematopoiesis following Bcl11b deletion in T cells restricts metastatic melanoma" J Immunol 192(4): 1946-1953. PubMed
Using several tumor models, we demonstrate that mice deficient in Bcl11b in T cells, although having reduced numbers of T cells in the peripheral lymphoid organs, developed significantly less tumors compared with wild-type mice. Bcl11b(-/-) CD4(+) T cells, with elevated TNF-alpha levels, but not the Bcl11b(-/-) CD8(+) T cells, were required for the reduced tumor burden, as were NK1.1(+) cells, found in increased numbers in Bcl11b(F/F)/CD4-Cre mice. Among NK1.1(+) cells, the NK cell population was predominant in number and was the only population displaying elevated granzyme B levels and increased degranulation, although not increased proliferation. Although the number of myeloid-derived suppressor cells was increased in the lungs with metastatic tumors of Bcl11b(F/F)/CD4-Cre mice, their arginase-1 levels were severely reduced. The increase in NK cell and myeloid-derived suppressor cell numbers was associated with increased bone marrow and splenic hematopoiesis. Finally, the reduced tumor burden, increased numbers of NK cells in the lung, and increased hematopoiesis in Bcl11b(F/F)/CD4-Cre mice were all dependent on TNF-alpha. Moreover, TNF-alpha treatment of wild-type mice also reduced the tumor burden and increased hematopoiesis and the numbers and activity of NK cells in the lung. In vitro treatment with TNF-alpha of lineage-negative hematopoietic progenitors increased NK and myeloid differentiation, further supporting a role of TNF-alpha in promoting hematopoiesis. These studies reveal a novel role for TNF-alpha in the antitumor immune response, specifically in stimulating hematopoiesis and increasing the numbers and activity of NK cells.
in vivo CD8+ T cell depletion, Flow Cytometry
Cyktor, J. C., et al. (2013). "Clonal expansions of CD8+ T cells with IL-10 secreting capacity occur during chronic Mycobacterium tuberculosis infection" PLoS One 8(3): e58612. PubMed
The exact role of CD8(+) T cells during Mycobacterium tuberculosis (Mtb) infection has been heavily debated, yet it is generally accepted that CD8(+) T cells contribute to protection against Mtb. In this study, however, we show that the Mtb-susceptible CBA/J mouse strain accumulates large numbers of CD8(+) T cells in the lung as infection progresses, and that these cells display a dysfunctional and immunosuppressive phenotype (PD-1(+), Tim-3(+), CD122(+)). CD8(+) T cell expansions from the lungs of Mtb-infected CBA/J mice were also capable of secreting the immunosuppressive cytokine interleukin-10 (IL-10), although in vivo CD8(+) T cell depletion did not significantly alter Mtb burden. Further analysis revealed that pulmonary CD8(+) T cells from Mtb-infected CBA/J mice were clonally expanded, preferentially expressing T cell receptor (TcR) Vbeta chain 8 (8.2, 8.3) or Vbeta 14. Although Vbeta8(+) CD8(+) T cells were responsible for the majority of IL-10 production, in vivo depletion of Vbeta8(+) did not significantly change the outcome of Mtb infection, which we hypothesize was a consequence of their dual IL-10/IFN-gamma secreting profiles. Our data demonstrate that IL-10-secreting CD8(+) T cells can arise during chronic Mtb infection, although the significance of this T cell population in tuberculosis pathogenesis remains unclear.
in vivo CD8+ T cell depletion
Hervieu, A., et al. (2013). "Dacarbazine-mediated upregulation of NKG2D ligands on tumor cells activates NK and CD8 T cells and restrains melanoma growth" J Invest Dermatol 133(2): 499-508. PubMed
Dacarbazine (DTIC) is a cytotoxic drug widely used for melanoma treatment. However, the putative contribution of anticancer immune responses in the efficacy of DTIC has not been evaluated. By testing how DTIC affects host immune responses to cancer in a mouse model of melanoma, we unexpectedly found that both natural killer (NK) and CD8(+) T cells were indispensable for DTIC therapeutic effect. Although DTIC did not directly affect immune cells, it triggered the upregulation of NKG2D ligands on tumor cells, leading to NK cell activation and IFNgamma secretion in mice and humans. NK cell-derived IFNgamma subsequently favored upregulation of major histocompatibility complex class I molecules on tumor cells, rendering them sensitive to cytotoxic CD8(+) T cells. Accordingly, DTIC markedly enhanced cytotoxic T lymphocyte antigen 4 inhibition efficacy in vivo in an NK-dependent manner. These results underscore the immunogenic properties of DTIC and provide a rationale to combine DTIC with immunotherapeutic agents that relieve immunosuppression in vivo.
Immunofluorescence
Schwager, K., et al. (2013). "The immunocytokine L19-IL2 eradicates cancer when used in combination with CTLA-4 blockade or with L19-TNF" J Invest Dermatol 133(3): 751-758. PubMed
Systemic high-dose IL2 promotes long-term survival in a subset of metastatic melanoma patients, but this treatment is accompanied by severe toxicities. The immunocytokine L19-IL2, in which IL2 is fused to the human L19 antibody capable of selective accumulation on tumor neovasculature, has recently shown encouraging clinical activity in patients with metastatic melanoma. In this study, we have investigated the therapeutic performance of L19-IL2, administered systemically in combination with a murine anti-CTLA-4 antibody or with a second clinical-stage immunocytokine (L19-TNF) in two syngeneic immunocompetent mouse models of cancer. We observed complete tumor eradications when L19-IL2 was used in combination with CTLA-4 blockade. Interestingly, mice cured from F9 tumors developed new lesions when rechallenged with tumor cells after therapy, whereas mice cured from CT26 tumors were resistant to tumor rechallenge. Similarly, L19-IL2 induced complete remissions when administered in a single intratumoral injection in combination with L19-TNF, whereas the two components did not lead to cures when administered as single agents. These findings provide a rationale for combination trials in melanoma, as the individual therapeutic agents have been extensively studied in clinical trials, and the antigen recognized by the L19 antibody has an identical sequence in mouse and man.
in vivo CD8+ T cell depletion, Flow Cytometry
Hafalla, J. C., et al. (2012). "The CTLA-4 and PD-1/PD-L1 inhibitory pathways independently regulate host resistance to Plasmodium-induced acute immune pathology" PLoS Pathog 8(2): e1002504. PubMed
The balance between pro-inflammatory and regulatory immune responses in determining optimal T cell activation is vital for the successful resolution of microbial infections. This balance is maintained in part by the negative regulators of T cell activation, CTLA-4 and PD-1/PD-L, which dampen effector responses during chronic infections. However, their role in acute infections, such as malaria, remains less clear. In this study, we determined the contribution of CTLA-4 and PD-1/PD-L to the regulation of T cell responses during Plasmodium berghei ANKA (PbA)-induced experimental cerebral malaria (ECM) in susceptible (C57BL/6) and resistant (BALB/c) mice. We found that the expression of CTLA-4 and PD-1 on T cells correlates with the extent of pro-inflammatory responses induced during PbA infection, being higher in C57BL/6 than in BALB/c mice. Thus, ECM develops despite high levels of expression of these inhibitory receptors. However, antibody-mediated blockade of either the CTLA-4 or PD-1/PD-L1, but not the PD-1/PD-L2, pathways during PbA-infection in ECM-resistant BALB/c mice resulted in higher levels of T cell activation, enhanced IFN-gamma production, increased intravascular arrest of both parasitised erythrocytes and CD8(+) T cells to the brain, and augmented incidence of ECM. Thus, in ECM-resistant BALB/c mice, CTLA-4 and PD-1/PD-L1 represent essential, independent and non-redundant pathways for maintaining T cell homeostasis during a virulent malaria infection. Moreover, neutralisation of IFN-gamma or depletion of CD8(+) T cells during PbA infection was shown to reverse the pathologic effects of regulatory pathway blockade, highlighting that the aetiology of ECM in the BALB/c mice is similar to that in C57BL/6 mice. In summary, our results underscore the differential and complex regulation that governs immune responses to malaria parasites.
in vivo CD8+ T cell depletion
Chyou, S., et al. (2011). "Coordinated regulation of lymph node vascular-stromal growth first by CD11c+ cells and then by T and B cells" J Immunol 187(11): 5558-5567. PubMed
Lymph node blood vessels play important roles in the support and trafficking of immune cells. The blood vasculature is a component of the vascular-stromal compartment that also includes the lymphatic vasculature and fibroblastic reticular cells (FRCs). During immune responses as lymph nodes swell, the blood vasculature undergoes a rapid proliferative growth that is initially dependent on CD11c(+) cells and vascular endothelial growth factor (VEGF) but is independent of lymphocytes. The lymphatic vasculature grows with similar kinetics and VEGF dependence, suggesting coregulation of blood and lymphatic vascular growth, but lymphatic growth has been shown to be B cell dependent. In this article, we show that blood vascular, lymphatic, and FRC growth are coordinately regulated and identify two distinct phases of vascular-stromal growth–an initiation phase, characterized by upregulated vascular-stromal proliferation, and a subsequent expansion phase. The initiation phase is CD11c(+) cell dependent and T/B cell independent, whereas the expansion phase is dependent on B and T cells together. Using CCR7(-/-) mice and selective depletion of migratory skin dendritic cells, we show that endogenous skin-derived dendritic cells are not important during the initiation phase and uncover a modest regulatory role for CCR7. Finally, we show that FRC VEGF expression is upregulated during initiation and that dendritic cells can stimulate increased fibroblastic VEGF, suggesting the scenario that lymph node-resident CD11c(+) cells orchestrate the initiation of blood and lymphatic vascular growth in part by stimulating FRCs to upregulate VEGF. These results illustrate how the lymph node microenvironment is shaped by the cells it supports.
in vivo CD8+ T cell depletion
Kumar, D., et al. (2011). "Intranasal administration of an inactivated Yersinia pestis vaccine with interleukin-12 generates protective immunity against pneumonic plague" Clin Vaccine Immunol 18(11): 1925-1935. PubMed
Inhalation of Yersinia pestis causes pneumonic plague, which rapidly progresses to death. A previously licensed killed whole-cell vaccine is presently unavailable due to its reactogenicity and inconclusive evidence of efficacy. The present study now shows that vaccination intranasally (i.n.) with inactivated Y. pestis CO92 (iYp) adjuvanted with interleukin-12 (IL-12) followed by an i.n. challenge with a lethal dose of Y. pestis CO92 prevented bacterial colonization and protected 100% of mice from pneumonic plague. Survival of the vaccinated mice correlated with levels of systemic and lung antibodies, reduced pulmonary pathology and proinflammatory cytokines, and the presence of lung lymphoid cell aggregates. Protection against pneumonic plague was partially dependent upon Fc receptors and could be transferred to naive mice with immune mouse serum. On the other hand, protection was not dependent upon complement, and following vaccination, depletion of CD4 and/or CD8 T cells before challenge did not affect survival. In summary, the results demonstrate the safety, immunogenicity, and protective efficacy of i.n. administered iYp plus IL-12 in a mouse model of pneumonic plague.
in vivo CD8+ T cell depletion
Simma, O., et al. (2009). "Identification of an indispensable role for tyrosine kinase 2 in CTL-mediated tumor surveillance" Cancer Res 69(1): 203-211. PubMed
We showed previously that Tyk2(-/-) natural killer cells lack the ability to lyse leukemic cells. As a consequence, the animals are leukemia prone. Here, we show that the impaired tumor surveillance extends to T cells. Challenging Tyk2(-/-) mice with EL4 thymoma significantly decreased disease latency. The crucial role of Tyk2 for CTL function was further characterized using the ovalbumin-expressing EG7 cells. Tyk2(-/-) OT-1 mice developed EG7-induced tumors significantly faster compared with wild-type (wt) controls. In vivo assays confirmed the defect in CD8(+) cytotoxicity on Tyk2 deficiency and clearly linked it to type I IFN signaling. An impaired CTL activity was only observed in IFNAR1(-/-) animals but not on IFNgamma or IL12p35 deficiency. Accordingly, EG7-induced tumors grew faster in IFNAR1(-/-) and Tyk2(-/-) but not in IFNgamma(-/-) or IL12p35(-/-) mice. Adoptive transfer experiments defined a key role of Tyk2 in CTL-mediated tumor surveillance. In contrast to wt OT-1 cells, Tyk2(-/-) OT-1 T cells were incapable of controlling EG7-induced tumor growth.
- Mus musculus (House mouse),
- Cancer Research
CCR2 and CCR5 co-inhibition modulates immunosuppressive myeloid milieu in glioma and synergizes with anti-PD-1 therapy.
In Oncoimmunology on 9 April 2024 by Pant, A., Hwa-Lin Bergsneider, B., et al.
PubMed
Immunotherapy has revolutionized the treatment of cancers. Reinvigorating lymphocytes with checkpoint blockade has become a cornerstone of immunotherapy for multiple tumor types, but the treatment of glioblastoma has not yet shown clinical efficacy. A major hurdle to treat GBM with checkpoint blockade is the high degree of myeloid-mediated immunosuppression in brain tumors that limits CD8 T-cell activity. A potential strategy to improve anti-tumor efficacy against glioma is to use myeloid-modulating agents to target immunosuppressive cells, such as myeloid-derived suppressor cells (MDSCs) in the tumor microenvironment. We found that the co-inhibition of the chemokine receptors CCR2 and CCR5 in murine model of glioma improves the survival and synergizes robustly with anti-PD-1 therapy. Moreover, the treatment specifically reduced the infiltration of monocytic-MDSCs (M-MDSCs) into brain tumors and increased lymphocyte abundance and cytokine secretion by tumor-infiltrating CD8 T cells. The depletion of T-cell subsets and myeloid cells abrogated the effects of CCR2 and CCR5 blockade, indicating that while broad depletion of myeloid cells does not improve survival, specific reduction in the infiltration of immunosuppressive myeloid cells, such as M-MDSCs, can boost the anti-tumor immune response of lymphocytes. Our study highlights the potential of CCR2/CCR5 co-inhibition in reducing myeloid-mediated immunosuppression in GBM patients. © 2024 The Author(s). Published with license by Taylor & Francis Group, LLC.
- Mus musculus (House mouse)
Antitumor Effect of Platinum-Modified STING Agonist MSA-2.
In ACS Omega on 16 January 2024 by Wang, M., Cai, Y., et al.
PubMed
The stimulator of interferon genes (STING)-activated innate immune pathway is strong and durable for tumor immunotherapy. MSA-2 is an available non-nucleotide human STING agonist that promotes the tumor immunotherapy of STING activation. However, strategies for remolding and improving the immunotherapy effects of MSA-2 are of value for clinical applications. Here, we synthesized the platinum salt-modified MSA-2 (MSA-2-Pt) due to platinum salt being a classic chemotherapeutic drug. We found that MSA-2-Pt could achieve double-effect antitumor immunotherapy, including inducing cell death by platinum and activating the STING pathway by MSA-2. In the colon carcinoma MC38 model (sensitive to immune checkpoint immunotherapy tumor) and melanoma B16F10 model (poorly immunogenic and highly aggressive tumor), the MSA-2-Pt had a good antitumor effect, which was a little better than MSA-2 with intratumor injections. The results present a promising strategy for STING activation in tumor immunotherapy and broadening platinum-based drugs. © 2024 The Authors. Published by American Chemical Society.
- Mus musculus (House mouse),
- Cancer Research
Targeting stroma and tumor, silencing galectin 1 treats orthotopic mouse hepatocellular carcinoma.
In Acta Pharmaceutica Sinica. B on 1 January 2024 by Setayesh, T., Hu, Y., et al.
PubMed
This study examines inhibiting galectin 1 (Gal1) as a treatment option for hepatocellular carcinoma (HCC). Gal1 has immunosuppressive and cancer-promoting roles. Our data showed that Gal1 was highly expressed in human and mouse HCC. The levels of Gal1 positively correlated with the stages of human HCC and negatively with survival. The roles of Gal1 in HCC were studied using overexpression (OE) or silencing using Igals1 siRNA delivered by AAV9. Prior to HCC initiation induced by RAS and AKT mutations, lgals1-OE and silencing had opposite impacts on tumor load. The treatment effect of lgals1 siRNA was further demonstrated by intersecting HCC at different time points when the tumor load had already reached 9% or even 42% of the body weight. Comparing spatial transcriptomic profiles of Gal1 silenced and OE HCC, inhibiting matrix formation and recognition of foreign antigen in CD45+ cell-enriched areas located at tumor-margin likely contributed to the anti-HCC effects of Gal1 silencing. Within the tumors, silencing Gal1 inhibited translational initiation, elongation, and termination. Furthermore, Gal1 silencing increased immune cells as well as expanded cytotoxic T cells within the tumor, and the anti-HCC effect of lgals1 siRNA was CD8-dependent. Overall, Gal1 silencing has a promising potential for HCC treatment. © 2024 The Authors.
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Enniatin A inhibits the chaperone Hsp90 and unleashes the immune system against triple-negative breast cancer.
In IScience on 15 December 2023 by Eisa, N. H., Crowley, V. M., et al.
PubMed
Low response rates and immune-related adverse events limit the remarkable impact of cancer immunotherapy. To improve clinical outcomes, preclinical studies have shown that combining immunotherapies with N-terminal Hsp90 inhibitors resulted in improved efficacy, even though induction of an extensive heat shock response (HSR) and less than optimal dosing of these inhibitors limited their clinical efficacy as monotherapies. We discovered that the natural product Enniatin A (EnnA) targets Hsp90 and destabilizes its client oncoproteins without inducing an HSR. EnnA triggers immunogenic cell death in triple-negative breast cancer (TNBC) syngeneic mouse models and exhibits superior antitumor activity compared to Hsp90 N-terminal inhibitors. EnnA reprograms the tumor microenvironment (TME) to promote CD8+ T cell-dependent antitumor immunity by reducing PD-L1 levels and activating the chemokine receptor CX3CR1 pathway. These findings provide strong evidence for transforming the immunosuppressive TME into a more tumor-hostile milieu by engaging Hsp90 with therapeutic agents involving novel mechanisms of action. © 2023 The Authors.
- Immunology and Microbiology
CD8 T cell response and its released cytokine IFN-γ are necessary for lung alveolar epithelial repair during bacterial pneumonia.
In Frontiers in Immunology on 13 November 2023 by Zhang, X., Ali, M., et al.
PubMed
Alveolar epithelial regeneration depends on the activity of resident quiescent progenitor cells. Alveolar epithelial type II (AT2) cells are known as the alveolar epithelial progenitor cells. They exit quiescent state, proliferate rapidly in response to injury and differentiate into alveolar epithelial type I (AT1) cells to regenerate the damaged alveolar epithelium. Although AT2 cell plasticity has been a very intense field of research, the role of CD8 T cell response and their released cytokine IFN-γ, in regulating AT2 cell plasticity and alveolar epithelial repair and regeneration after injury remains largely unknown. We used flow cytometry to quantify the amount of CD8 T cells in mouse lungs after bacterial pneumonia caused by Streptococcus pneumoniae. To determine whether CD8 T cells and their released cytokine IFN-γ are necessary for AT2 cell activity during alveolar epithelial regeneration, we performed loss of function studies using anti-CD8 or anti-IFN-γ monoclonal antibody (mAb) treatment in vivo. We assessed the effects of CD8 T cells and cytokine IFN-γ on AT2 cell differentiation capacity using the AT2- CD8 T cell co-culture system in vitro. We detected a transient wave of accumulation of CD8 T cells in mouse lungs, which coincided with the burst of AT2 cell proliferation during alveolar epithelial repair and regeneration in mice following bacterial pneumonia caused by Streptococcus pneumoniae. Depletion of CD8 T cells or neutralization of cytokine IFN-γ using anti-CD8 or anti-IFN-γ monoclonal antibody significantly reduced AT2 cell proliferation and differentiation into AT1 cells in mice after bacterial pneumonia. Furthermore, co-culture of CD8 T cells or cytokine IFN-γ with AT2 cells promoted AT2-to-AT1 cell differentiation in both murine and human systems. Conversely, blockade of IFN-γ signaling abrogated the increase in AT2-to-AT1 cell differentiation in the AT2- CD8 T cell co-culture system. Our data demonstrate that CD8 T-cell response and cytokine IFN-γ are necessary for promoting AT2 cell activity during alveolar epithelial repair and regeneration after acute lung injury caused by bacterial pneumonia. Copyright © 2023 Zhang, Ali, Pantuck, Yang, Lin, Bahmed, Kosmider and Tian.
- Cancer Research,
- Genetics,
- Immunology and Microbiology
DNA damage induced by CDK4 and CDK6 blockade triggers anti-tumor immune responses through cGAS-STING pathway.
In Communications Biology on 13 October 2023 by Fan, H., Liu, W., et al.
PubMed
CDK4/6 are important regulators of cell cycle and their inhibitors have been approved as anti-cancer drugs. Here, we report a STING-dependent anti-tumor immune mechanism responsible for tumor suppression by CDK4/6 blockade. Clinical datasets show that in human tissues, CDK4 and CDK6 are over-expressed and their expressions are negatively correlated with patients' overall survival and T cell infiltration. Deletion of Cdk4 or Cdk6 in tumor cells significantly reduce tumor growth. Mechanistically, we find that Cdk4 or Cdk6 deficiency contributes to an increased level of endogenous DNA damage, which triggers the cGAS-STING signaling pathway to activate type I interferon response. Knockout of Sting is sufficient to reverse and partially reverse the anti-tumor effect of Cdk4 and Cdk6 deficiency respectively. Therefore, our findings suggest that CDK4/6 inhibitors may enhance anti-tumor immunity through the STING-dependent type I interferon response. © 2023. Springer Nature Limited.
- In Vivo,
- Mus musculus (House mouse),
- Immunology and Microbiology,
- Cancer Research
Selective targeting of IL2Rβγ combined with radiotherapy triggers CD8- and NK-mediated immunity, abrogating metastasis in HNSCC.
In Cell Reports Medicine on 15 August 2023 by Gadwa, J., Amann, M., et al.
PubMed
The implementation of cancer immunotherapies has seen limited clinical success in head and neck squamous cell carcinoma (HNSCC). Interleukin-2 (IL-2), which modulates the survival and functionality of lymphocytes, is an attractive target for new immunotherapies but one that is limited by presence of regulatory T cells (Tregs) expressing the high-affinity IL-2Rα. The bispecific immunocytokine PD1-IL2v preferentially delivers IL-2 signaling through IL-2Rβγ on PD-1-expressing cells. Selectively targeting the intermediate-affinity IL-2Rβγ can be leveraged to induce anti-tumor immune responses in effector T cells and natural killer (NK) cells while limiting the negative regulation of IL-2Rα activation on Tregs. Using radiation therapy (RT) in combination with PD1-IL2v improves local tumor control and survival, and controls metastatic spread in orthotopic HNSCC tumor models. PD1-IL2v drives systemic activation and expansion of circulating and tumor-infiltrating cytotoxic T cells and NK cells while limiting Treg-mediated immunosuppression. These data show that PD1-L2v induces durable systemic tumor control in HNSCC. Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
- FC/FACS,
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
The co-delivery of adenovirus-based immune checkpoint vaccine elicits a potent anti-tumor effect in renal carcinoma.
In NPJ Vaccines on 4 August 2023 by Jiang, N., Zheng, Y., et al.
PubMed
Immune-based checkpoint therapy has made significant progress in cancer treatment, but its therapeutic effect is limited. A replication-defective adenovirus (Ad) vaccine encoding tumor antigen carbonic anhydrase IX (CAIX) combined with Ad-encoding immune checkpoint PD-L1 was developed to treat renal carcinoma. Three tumor models, subcutaneous, lung metastasis and orthotopic tumor were established, and Ad vaccines were used to immunize them and evaluate the vaccine's therapeutic effect. Compared to the single Ad vaccine group, the subcutaneous tumor growth was significantly reduced in Ad-CAIX/Ad-PD-L1 combination group. Co-immunization of Ad-CAIX/Ad-PD-L1 enhanced the induction and maturation of CD11c+ or CD8+CD11c+ DCs in the spleen and tumor and promoted the strong tumor-specific CD8+ T cell immune responses. In vivo CD8 T cell deletion assay showed that the anti-tumor effect of the Ad-CAIX/Ad-PD-L1 vaccine was mainly dependent on functional CD8+ T cell immune responses. Furthermore, the Ad-CAIX/Ad-PD-L1 vaccine effectively inhibited tumor growth and lung metastasis in metastatic or orthotopic models. These results indicate that the combination strategy of the immune checkpoint vaccine shows promising potential as an approach for malignant tumor therapy. © 2023. Springer Nature Limited.
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Vinblastine resets tumor-associated macrophages toward M1 phenotype and promotes antitumor immune response.
In Journal for Immunotherapy of Cancer on 1 August 2023 by Wang, Y. N., Wang, Y. Y., et al.
PubMed
Massive tumor-associated macrophage (TAM) infiltration is observed in many tumors, which usually display the immune-suppressive M2-like phenotype but can also be converted to an M1-like antitumor phenotype due to their high degree of plasticity. The macrophage polarization state is associated with changes in cell shape, macrophage morphology is associated with activation status. M1 macrophages appeared large and rounded, while M2 macrophages were stretched and elongated cells. Manipulating cell morphology has been shown to affect the polarization state of macrophages. The shape of the cell is largely dependent on cytoskeletal proteins, especially, microtubules. As a microtubule-targetting drug, vinblastine (VBL) has been used in chemotherapy. However, no study to date has explored the effect of VBL on TAM shape changes and its role in tumor immune response. We used fluorescent staining of the cytoskeleton and quantitative analysis to reveal the morphological differences between M0, M1, M2, TAM and VBL-treated TAM. Flow cytometry was used to confirm the polarization states of these macrophages using a cell surface marker-based classification. In vivo antibody depletion experiments in tumor mouse models were performed to test whether macrophages and CD8+ T cell populations were required for the antitumor effect of VBL. VBL and anti-PD-1 combination therapy was then investigated in comparison with monotherapy. RNA-seq of TAM of treated and untreated with VBL was performed to explore the changes in pathway activities. siRNA mediated knockdown experiments were performed to verify the target pathway that was affected by VBL treatment. Here, we showed that VBL, an antineoplastic agent that destabilizes microtubule, drove macrophage polarization into the M1-like phenotype both in vitro and in tumor models. The antitumor effect of VBL was attenuated in the absence of macrophages or CD8+ T cells. Mechanistically, VBL induces the activation of NF-κB and Cyba-dependent reactive oxygen species generation, thus polarizing TAMs to the M1 phenotype. In parallel, VBL promotes the nuclear translocation of transcription factor EB, inducing lysosome biogenesis and a dramatic increase in phagocytic activity in macrophages. This study explored whether manipulating cellular morphology affects macrophage polarization and consequently induces an antitumor response. Our data reveal a previously unrecognized antitumor mechanism of VBL and suggest a drug repurposing strategy combining VBL with immune checkpoint inhibitors to improve malignant tumor immunotherapy. © Author(s) (or their employer(s)) 2023. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
- Cancer Research
CD200R signaling contributes to unfavorable tumor microenvironment through regulating production of chemokines by tumor-associated myeloid cells.
In IScience on 16 June 2023 by Lin, C. H., Talebian, F., et al.
PubMed
CD200 is overexpressed in many solid tumors and considered as an immune checkpoint molecule dampening cancer immunity. In this study, we found that CD200R-/- mice were significantly more potent in rejecting these CD200+ tumors. scRNA sequencing demonstrated that tumors from CD200R-/- mice had more infiltration of CD4+ and CD8+ T cells, and NK cells but less infiltration of neutrophils. Antibody depletion experiments revealed that immune effector cells are crucial in inhibiting tumor growth in CD200R-/- mice. Mechanistically, we found that CD200R signaling regulates the expression of chemokines in tumor-associated myeloid cells (TAMCs). In the absence of CD200R, TAMCs increased expression of CCL24 and resulted in increased infiltration of eosinophils, which contributes to anti-tumor activity. Overall, we conclude that CD200R signaling contributes to unfavorable TME through chemokine-dependent recruitment of immune suppressive neutrophils and exclusion of anti-cancer immune effectors. Our study has implications in developing CD200-CD200R targeted immunotherapy of solid tumors. © 2023 The Author(s).
- Mus musculus (House mouse)
CCR4 and CCR7 differentially regulate thymocyte localization with distinct outcomes for central tolerance.
In eLife on 2 June 2023 by Li, Y., Guaman Tipan, P., et al.
PubMed
Central tolerance ensures autoreactive T cells are eliminated or diverted to the regulatory T cell lineage, thus preventing autoimmunity. To undergo central tolerance, thymocytes must enter the medulla to test their T-cell receptors (TCRs) for autoreactivity against the diverse self-antigens displayed by antigen-presenting cells (APCs). While CCR7 is known to promote thymocyte medullary entry and negative selection, our previous studies implicate CCR4 in these processes, raising the question of whether CCR4 and CCR7 play distinct or redundant roles in central tolerance. Here, synchronized positive selection assays, two-photon time-lapse microscopy, and quantification of TCR-signaled apoptotic thymocytes, demonstrate that CCR4 and CCR7 promote medullary accumulation and central tolerance of distinct post-positive selection thymocyte subsets in mice. CCR4 is upregulated within hours of positive selection signaling and promotes medullary entry and clonal deletion of immature post-positive selection thymocytes. In contrast, CCR7 is expressed several days later and is required for medullary localization and negative selection of mature thymocytes. In addition, CCR4 and CCR7 differentially enforce self-tolerance, with CCR4 enforcing tolerance to self-antigens presented by activated APCs, which express CCR4 ligands. Our findings show that CCR7 expression is not synonymous with medullary localization and support a revised model of central tolerance in which CCR4 and CCR7 promote early and late stages of negative selection, respectively, via interactions with distinct APC subsets. © 2023, Li, Guaman Tipan et al.
- Cancer Research
Simultaneous targeting of PD-1 and IL-2Rβγ with radiation therapy inhibits pancreatic cancer growth and metastasis.
In Cancer Cell on 8 May 2023 by Piper, M., Hoen, M., et al.
PubMed
In pancreatic ductal adenocarcinoma (PDAC) patients, we show that response to radiation therapy (RT) is characterized by increased IL-2Rβ and IL-2Rγ along with decreased IL-2Rα expression. The bispecific PD1-IL2v is a PD-1-targeted IL-2 variant (IL-2v) immunocytokine with engineered IL-2 cis targeted to PD-1 and abolished IL-2Rα binding, which enhances tumor-antigen-specific T cell activation while reducing regulatory T cell (Treg) suppression. Using PD1-IL2v in orthotopic PDAC KPC-driven tumor models, we show marked improvement in local and metastatic survival, along with a profound increase in tumor-infiltrating CD8+ T cell subsets with a transcriptionally and metabolically active phenotype and preferential activation of antigen-specific CD8+ T cells. In combination with single-dose RT, PD1-IL2v treatment results in a robust, durable expansion of polyfunctional CD8+ T cells, T cell stemness, tumor-specific memory immune response, natural killer (NK) cell activation, and decreased Tregs. These data show that PD1-IL2v leads to profound local and distant response in PDAC. Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
- Cancer Research
Neutrophil Conversion to a Tumor-Killing Phenotype Underpins Effective Microbial Therapy.
In Cancer Research on 14 April 2023 by Yam, A. O., Bailey, J., et al.
PubMed
The inflammatory microenvironment of solid tumors creates a protumorigenic milieu that resembles chronic inflammation akin to a subverted wound healing response. Here, we investigated the effect of converting the tumor microenvironment from a chronically inflamed state to one of acute microbial inflammation by injecting microbial bioparticles directly into tumors. Intratumoral microbial bioparticle injection led to rapid and dramatic changes in the tumor immune composition, the most striking of which was a substantial increase in the presence of activated neutrophils. In situ photoconversion and intravital microscopy indicated that tumor neutrophils transiently switched from sessile producers of VEGF to highly motile neutrophils that clustered to make neutrophil-rich domains in the tumor. The neutrophil clusters remodeled tumor tissue and repressed tumor growth. Single-cell transcriptional analysis of microbe-stimulated neutrophils showed a profound shift in gene expression towards heightened activation and antimicrobial effector function. Microbe-activated neutrophils also upregulated chemokines known to regulate neutrophil and CD8+ T-cell recruitment. Microbial therapy also boosted CD8+ T-cell function and enhanced the therapeutic benefit of checkpoint inhibitor therapy in tumor-bearing mice and provided protection in a model of tumor recurrence. These data indicate that one of the major effector mechanisms of microbial therapy is the conversion of tumor neutrophils from a wound healing to an acutely activated cytotoxic phenotype, highlighting a rationale for broader deployment of microbial therapy in the treatment of solid cancers. Intratumoral injection of microbial bioparticles stimulates neutrophil antitumor functions, suggesting pathways for optimizing efficacy of microbial therapies and paving the way for their broader utilization in the clinic. ©2023 The Authors; Published by the American Association for Cancer Research.
- Cancer Research
STING inhibits the reactivation of dormant metastasis in lung adenocarcinoma.
In Nature on 1 April 2023 by Hu, J., Sanchez-Rivera, F. J., et al.
PubMed
Metastasis frequently develops from disseminated cancer cells that remain dormant after the apparently successful treatment of a primary tumour. These cells fluctuate between an immune-evasive quiescent state and a proliferative state liable to immune-mediated elimination1-6. Little is known about the clearing of reawakened metastatic cells and how this process could be therapeutically activated to eliminate residual disease in patients. Here we use models of indolent lung adenocarcinoma metastasis to identify cancer cell-intrinsic determinants of immune reactivity during exit from dormancy. Genetic screens of tumour-intrinsic immune regulators identified the stimulator of interferon genes (STING) pathway as a suppressor of metastatic outbreak. STING activity increases in metastatic progenitors that re-enter the cell cycle and is dampened by hypermethylation of the STING promoter and enhancer in breakthrough metastases or by chromatin repression in cells re-entering dormancy in response to TGFβ. STING expression in cancer cells derived from spontaneous metastases suppresses their outgrowth. Systemic treatment of mice with STING agonists eliminates dormant metastasis and prevents spontaneous outbreaks in a T cell- and natural killer cell-dependent manner-these effects require cancer cell STING function. Thus, STING provides a checkpoint against the progression of dormant metastasis and a therapeutically actionable strategy for the prevention of disease relapse. © 2023. The Author(s), under exclusive licence to Springer Nature Limited.
- In Vivo,
- Mus musculus (House mouse),
- Cancer Research
Tumor PD-L1 engages myeloid PD-1 to suppress type I interferon to impair cytotoxic T lymphocyte recruitment.
In Cancer Cell on 13 March 2023 by Klement, J. D., Redd, P. S., et al.
PubMed
The cellular and molecular mechanisms underlying tumor cell PD-L1 (tPD-L1) function in tumor immune evasion are incompletely understood. We report here that tPD-L1 does not suppress cytotoxic T lymphocyte (CTL) activity in co-cultures of tumor cells and tumor-specific CTLs and exhibits no effect on primary tumor growth. However, deleting tPD-L1 decreases lung metastasis in a CTL-dependent manner in tumor-bearing mice. Depletion of myeloid cells or knocking out PD-1 in myeloid cells (mPD-1) impairs tPD-L1 promotion of tumor lung metastasis in mice. Single-cell RNA sequencing (scRNA-seq) reveals that tPD-L1 engages mPD-1 to activate SHP2 to antagonize the type I interferon (IFN-I) and STAT1 pathway to repress Cxcl9 and impair CTL recruitment to lung metastases. Human cancer patient response to PD-1 blockade immunotherapy correlates with IFN-I response in myeloid cells. Our findings determine that tPD-L1 engages mPD-1 to activate SHP2 to suppress the IFN-I-STAT1-CXCL9 pathway to impair CTL tumor recruitment in lung metastasis. Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Antigen-presenting type-I conventional dendritic cells facilitate curative checkpoint blockade immunotherapy in pancreatic cancer
Preprint on BioRxiv : the Preprint Server for Biology on 6 March 2023 by Mahadevan, K. K., Dyevoich, A. M., et al.
PubMed
Inflammation and tissue damage associated with pancreatitis can precede or occur concurrently with pancreatic ductal adenocarcinoma (PDAC). We demonstrate that in PDAC coupled with pancreatitis (ptPDAC), antigen-presenting type-I conventional dendritic cells (cDC1s) are specifically activated. Immune checkpoint blockade therapy (iCBT) leads to cytotoxic CD8 + T cell activation and eradication of ptPDAC with restoration of lifespan even upon PDAC re-challenge. Such eradication of ptPDAC was reversed following specific depletion of dendritic cells. Employing PDAC antigen-loaded cDC1s as a vaccine, immunotherapy-resistant PDAC was rendered sensitive to iCBT with a curative outcome. Analysis of the T-cell receptor (TCR) sequences in the tumor infiltrating CD8 + T cells following cDC1 vaccination coupled with iCBT identified unique CDR3 sequences with potential therapeutic significance. Our findings identify a fundamental difference in the immune microenvironment and adaptive immune response in PDAC concurrent with, or without pancreatitis, and provides a rationale for combining cDC1 vaccination with iCBT as a potential treatment option.
- IHC,
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Combinatory EHMT and PARP inhibition induces an interferon response and a CD8 T cell-dependent tumor regression in PARP inhibitor-resistant models
Preprint on BioRxiv : the Preprint Server for Biology on 23 February 2023 by Nguyen, L. L., Watson, Z. L., et al.
PubMed
ABSTRACT Euchromatic histone lysine methyltransferases 1 and 2 (EHMT1/2), which catalyze demethylation of histone H3 lysine 9 (H3K9me2), contribute to tumorigenesis and therapy resistance through unknown mechanisms of action. In ovarian cancer, EHMT1/2 and H3K9me2 are directly linked to acquired resistance to poly-ADP-ribose polymerase (PARP) inhibitors and are correlated with poor clinical outcomes. Using a combination of experimental and bioinformatic analyses in several PARP inhibitor resistant ovarian cancer models, we demonstrate that combinatory inhibition of EHMT and PARP is effective in treating PARP inhibitor resistant ovarian cancers. Our in vitro studies show that combinatory therapy reactivates transposable elements, increases immunostimulatory dsRNA formation, and elicits several immune signaling pathways. Our in vivo studies show that both single inhibition of EHMT and combinatory inhibition of EHMT and PARP reduces tumor burden, and that this reduction is dependent on CD8 T cells. Together, our results uncover a direct mechanism by which EHMT inhibition helps to overcome PARP inhibitor resistance and shows how an epigenetic therapy can be used to enhance anti-tumor immunity and address therapy resistance.
- Immunology and Microbiology
Regulatory T cells suppress the formation of potent KLRK1 and IL-7R expressing effector CD8 T cells by limiting IL-2.
In eLife on 27 January 2023 by Tsyklauri, O., Chadimova, T., et al.
PubMed
Regulatory T cells (Tregs) are indispensable for maintaining self-tolerance by suppressing conventional T cells. On the other hand, Tregs promote tumor growth by inhibiting anticancer immunity. In this study, we identified that Tregs increase the quorum of self-reactive CD8+ T cells required for the induction of experimental autoimmune diabetes in mice. Their major suppression mechanism is limiting available IL-2, an essential T-cell cytokine. Specifically, Tregs inhibit the formation of a previously uncharacterized subset of antigen-stimulated KLRK1+ IL-7R+ (KILR) CD8+ effector T cells, which are distinct from conventional effector CD8+ T cells. KILR CD8+ T cells show superior cell-killing abilities in vivo. The administration of agonistic IL-2 immunocomplexes phenocopies the absence of Tregs, i.e., it induces KILR CD8+ T cells, promotes autoimmunity, and enhances antitumor responses in mice. Counterparts of KILR CD8+ T cells were found in the human blood, revealing them as a potential target for immunotherapy. © 2023, Tsyklauri et al.
- FC/FACS,
- Mus musculus (House mouse),
- Cancer Research,
- Immunology and Microbiology
Repurposing nitric oxide donating drugs in cancer therapy through immune modulation.
In Journal of Experimental & Clinical Cancer Research : CR on 14 January 2023 by Li, C. Y., Anuraga, G., et al.
PubMed
Nitric oxide-releasing drugs are used for cardiovascular diseases; however, their effects on the tumor immune microenvironment are less clear. Therefore, this study explored the impact of nitric oxide donors on tumor progression in immune-competent mice. The effects of three different nitric oxide-releasing compounds (SNAP, SNP, and ISMN) on tumor growth were studied in tumor-bearing mouse models. Three mouse tumor models were used: B16F1 melanoma and LL2 lung carcinoma in C57BL/6 mice, CT26 colon cancer in BALB/c mice, and LL2 lung carcinoma in NOD/SCID mice. After nitric oxide treatment, splenic cytokines and lymphocytes were analyzed by cytokine array and flow cytometry, and tumor-infiltrating lymphocytes in the TME were analyzed using flow cytometry and single-cell RNA sequencing. Low doses of three exogenous nitric oxide donors inhibited tumor growth in two immunocompetent mouse models but not in NOD/SCID immunodeficient mice. Low-dose nitric oxide donors increase the levels of splenic cytokines IFN-γ and TNF-α but decrease the levels of cytokines IL-6 and IL-10, suggesting an alteration in Th2 cells. Nitric oxide donors increased the number of CD8+ T cells with activation gene signatures, as indicated by single-cell RNA sequencing. Flow cytometry analysis confirmed an increase in infiltrating CD8+ T cells and dendritic cells. The antitumor effect of nitric oxide donors was abolished by depletion of CD8+ T cells, indicating the requirement for CD8+ T cells. Tumor inhibition correlated with a decrease in a subtype of protumor macrophages and an increase in a subset of Arg1-positive macrophages expressing antitumor gene signatures. The increase in this subset of macrophages was confirmed by flow cytometry analysis. Finally, the combination of low-dose nitric oxide donor and cisplatin induced an additive cancer therapeutic effect in two immunocompetent animal models. The enhanced therapeutic effect was accompanied by an increase in the cells expressing the gene signature of NK cell. Low concentrations of exogenous nitric oxide donors inhibit tumor growth in vivo by regulating T cells and macrophages. CD8+ T cells are essential for antitumor effects. In addition, low-dose nitric oxide donors may be combined with chemotherapeutic drugs in cancer therapy in the future. © 2023. The Author(s).
- Cancer Research,
- Immunology and Microbiology
Tumor Cell-Intrinsic SETD2 Deficiency Reprograms Neutrophils to Foster Immune Escape in Pancreatic Tumorigenesis.
In Advanced Science (Weinheim, Baden-Wurttemberg, Germany) on 1 January 2023 by Niu, N., Shen, X., et al.
PubMed
Genetic and epigenetic alterations play central roles in shaping the immunosuppressive tumor microenvironment (TME) to evade immune surveillance. The previous study shows that SETD2-H3K36me3 loss promotes KRAS-induced pancreatic tumorigenesis. However, little is known about its role in remodeling the TME and immune evasion. Here, it is shown that SETD2 deficiency can reprogram neutrophils to an immunosuppressive phenotype, thereby promoting immune escape during pancreatic tumor progression. By comprehensive profiling of the intratumoral immune cells, neutrophils are identified as the subset with the most significant changes upon Setd2 loss. Setd2-deficient pancreatic tumor cells directly enhance neutrophil recruitment and reprogramming, thereby inhibiting the cytotoxicity of CD8+ T cells to foster tumor progression. Mechanistically, it is revealed that Setd2-H3K36me3 loss leads to ectopic gain of H3K27me3 to downregulate Cxadr expression, which boosts the PI3K-AKT pathway and excessive expression of CXCL1 and GM-CSF, thereby promoting neutrophil recruitment and reprogramming toward an immunosuppressive phenotype. The study provides mechanistic insights into how tumor cell-intrinsic Setd2 deficiency strengthens the immune escape during pancreatic tumorigenesis, which may offer potential therapeutic implications for pancreatic cancer patients with SETD2 deficiency. © 2022 The Authors. Advanced Science published by Wiley-VCH GmbH.