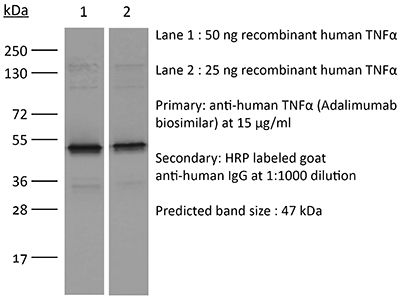
InVivoSIM anti-human TNFα (Adalimumab Biosimilar)
Product Description
Specifications
| Isotype | Human IgG1 |
|---|---|
| Recommended Isotype Control(s) | RecombiMAb human IgG1 isotype control, anti-hen egg lysozyme |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Human TNFα |
| Reported Applications |
in vitro TNFα neutralization in vitro functional assay Flow Cytometry ELISA Immunofluoresence Immunoprecipitation Immunohistochemistry Western Blot |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
<0.5EU/mg (<0.0005EU/μg) Determined by LAL assay |
| Aggregation |
<5% Determined by SEC |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_2894722 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Wang F, Zhou F, Peng J, Chen H, Xie J, Liu C, Xiong H, Chen S, Xue G, Zhou X, Xie Y (2024). "Macrophage Tim-3 maintains intestinal homeostasis in DSS-induced colitis by suppressing neutrophil necroptosis" Redox Biol .
PubMed
T-cell immunoglobulin domain and mucin domain-3 (Tim-3) is a versatile immunomodulator that protects against intestinal inflammation. Necroptosis is a type of cell death that regulates intestinal homeostasis and inflammation. The mechanism(s) underlying the protective role of macrophage Tim-3 in intestinal inflammation is unclear; thus, we investigated whether specific Tim-3 knockdown in macrophages drives intestinal inflammation via necroptosis. Tim-3 protein and mRNA expression were assessed via double immunofluorescence staining and single-cell RNA sequencing (sc-RNA seq), respectively, in the colonic tissues of patients with inflammatory bowel disease (IBD) and healthy controls. Macrophage-specific Tim3-knockout (Tim-3M-KO) mice were generated to explore the function and mechanism of Tim-3 in dextran sodium sulfate (DSS)-induced colitis. Necroptosis was blocked by pharmacological inhibitors of receptor-interacting protein kinase (RIP)1, RIP3, and reactive oxygen species (ROS). Additionally, in vitro experiments were performed to assess the mechanisms of neutrophil necroptosis induced by Tim-3 knockdown macrophages. Although Tim-3 is relatively inactive in macrophages during colon homeostasis, it is highly active during colitis. Compared to those in controls, Tim-3M-KO mice showed increased susceptibility to colitis, higher colitis scores, and increased pro-inflammatory mediator expression. Following the administration of RIP1/RIP3 or ROS inhibitors, a significant reduction in intestinal inflammation symptoms was observed in DSS-treated Tim-3M-KO mice. Further analysis indicated the TLR4/NF-κB pathway in Tim-3 knockdown macrophages mediates the TNF-α-induced necroptosis pathway in neutrophils. Macrophage Tim-3 regulates neutrophil necroptosis via intracellular ROS signaling. Tim-3 knockdown macrophages can recruit neutrophils and induce neutrophil necroptosis, thereby damaging the intestinal mucosal barrier and triggering a vicious cycle in the development of colitis. Our results demonstrate a protective role of macrophage Tim-3 in maintaining gut homeostasis by inhibiting neutrophil necroptosis and provide novel insights into the pathogenesis of IBD.
-
Jarvi NL, Balu-Iyer SV (2023). "A mechanistic marker-based screening tool to predict clinical immunogenicity of biologics" Commun Med (Lond) 3(1):174.
PubMed
Background: The efficacy and safety of therapeutic proteins are undermined by immunogenicity driven by anti-drug antibodies. Immunogenicity risk assessment is critically necessary during drug development, but current methods lack predictive power and mechanistic insight into antigen uptake and processing leading to immune response. A key mechanistic step in T-cell-dependent immune responses is the migration of mature dendritic cells to T-cell areas of lymphoid compartments, and this phenomenon is most pronounced in the immune response toward subcutaneously delivered proteins. Methods: The migratory potential of monocyte-derived dendritic cells is proposed to be a mechanistic marker for immunogenicity screening. Following exposure to therapeutic protein in vitro, dendritic cells are analyzed for changes in activation markers (CD40 and IL-12) in combination with levels of the chemokine receptor CXCR4 to represent migratory potential. Then a transwell assay captures the intensity of dendritic cell migration in the presence of a gradient of therapeutic protein and chemokine ligands. Results: Here, we show that an increased ability of the therapeutic protein to induce dendritic cell migration along a gradient of chemokine CCL21 and CXCL12 predicts higher immunogenic potential. Expression of the chemokine receptor CXCR4 on human monocyte-derived dendritic cells, in combination with activation markers CD40 and IL-12, strongly correlates with clinical anti-drug antibody incidence. Conclusions: Mechanistic understanding of processes driving immunogenicity led to the development of a predictive tool for immunogenicity risk assessment of therapeutic proteins. These predictive markers could be adapted for immunogenicity screening of other biological modalities.
-
Fredon M, Poussard M, Biichlé S, Bonnefoy F, Mantion CF, Seffar E, Renosi F, Bôle-Richard E, Boidot R, Chevrier S, Anna F, Loustau M, Caumartin J, Gonçalves-Venturelli M, Robinet E, Saas P, Deconinck E, Daguidau E, Roussel X, Godet Y, Adotévi O, Ange
PubMed
Chimeric antigen receptor (CAR) T cells express an extracellular domain consisting of a single-chain fragment variable (scFv) targeting a surface tumor-associated antigen. scFv selection should involve safety profiling with evaluation of the efficacy/toxicity balance, especially when the target antigen also is expressed on healthy cells. Here, to assess differences in terms of efficacy and on-target/off-tumor effects, we generated five different CARs targeting CD123 by substituting only the scFv. In in vitro models, T cells engineered to express three of these five CD123 CARs were effectively cytotoxic on leukemic cells without increasing lysis of monocytes or endothelial cells. Using the IncuCyte system, we confirmed the low cytotoxicity of CD123 CAR T cells on endothelial cells. Hematotoxicity evaluation using progenitor culture and CD34 cell lysis showed that two of the five CD123 CAR T cells were less cytotoxic on hematopoietic stem cells. Using a humanized mouse model, we confirmed that CD123- cells were not eliminated by the CD123 CAR T cells. Two CD123 CAR T cells reduced tumor infiltration and increased the overall survival of mice in three in vivo models of blastic plasmacytoid dendritic cell neoplasm. In an aggressive version of this model, bulk RNA sequencing analysis showed that these CD123 CAR T cells upregulated genes associated with cytotoxicity and activation/exhaustion a few days after the injection. Together, these results emphasize the importance of screening different scFvs for the development of CAR constructs to support selection of cells with the optimal risk-benefit ratio for clinical development.
-
Müller T, Tasser C, Tesar M, Fucek I, Schniegler-Mattox U, Koch J, Ellwanger K (2023). "Selection of bispecific antibodies with optimal developability using FcRn‑Ph‑HPLC as an optimized FcRn affinity chromatography method" MAbs 15(1):2245519.
PubMed
A challenge when developing therapeutic antibodies is the identification of candidates with favorable pharmacokinetics (PK) early in development. A key determinant of immunoglobulin (IgG) serum half‑life in vivo is the efficiency of pH-dependent binding to the neonatal Fc receptor (FcRn). Numerous studies have proposed techniques to assess FcRn binding of IgG-based therapeutics in vitro, enabling prediction of serum half-life prior to clinical assessment. FcRn high-performance liquid chromatography (HPLC) assays FcRn binding of therapeutic IgGs across a pH gradient, allowing the correlation of IgG column retention time to the half‑life of a therapeutic IgG in vivo. However, as FcRn retention time cannot be directly compared to an in vivo parameter, modifications to FcRn-HPLC are required to enable interpretation of the data within a physiological context, to provide more accurate estimations of serum half-life. This study presents an important modification to this method, FcRn-pH-HPLC, which reproducibly measures FcRn dissociation pH, allowing correlation with previously established half-lives of therapeutic antibodies. Furthermore, the influence of incorporating various antibody modifications, binding modules, and their orientations within IgGs and bispecifics on FcRn dissociation pH was evaluated using antibodies from the redirected optimized cell killing (ROCK®) platform. Target and effector antigen-binding domain sequences, their presentation format and orientation within a bispecific antibody alter FcRn retention; tested Fc domain modifications and incorporating stabilizing disulfide bonds had minimal effect. This study may inform the generation of mono-, bi- and multi-specific antibodies with tailored half-lives based on FcRn binding properties in vitro, to differentiate antibody-based therapeutic candidates with optimal developability.
Product Citations
-
M1-linked ubiquitination by LUBAC regulates AMPK signalling and the response to energetic stress.
In Cell Death Differ on 13 February 2026 by Elbæk, C. R., Gradinaru, S., et al.
PubMed
Methionine-1 (M1)-linked ubiquitin chains, assembled by the linear ubiquitin chain assembly complex (LUBAC) and disassembled by the deubiquitinase OTULIN, are critical regulators of inflammation and immune homoeostasis. Genetic loss or mutation of the LUBAC subunits HOIP and HOIL-1 or of OTULIN causes autoinflammatory syndromes accompanied by metabolic defects, including amylopectinosis, lipodystrophy, and fatty liver disease. Yet, it remains unclear how LUBAC and OTULIN control metabolic signalling. Here, we demonstrate that LUBAC and OTULIN dynamically regulate the energy-sensing kinase AMPK, a central sensor and switch for cellular and organismal energy balance. LUBAC's activity through the catalytic subunit HOIP is required for full AMPK activation in response to energetic stress, whereas OTULIN antagonises this response. LUBAC and OTULIN form a complex with AMPK, and LUBAC can directly ubiquitinate AMPKα and β subunits in cells and in vitro, establishing AMPK as a bona fide M1-linked ubiquitin substrate. Loss of LUBAC blunts AMPK activation, reduces bioenergetic adaptability, impairs autophagy, and sensitises cells to starvation-induced death, while Drosophila lacking Lubel - the fly orthologue of LUBAC - exhibit defective AMPK activation and reduced survival during starvation. Our findings identify M1-linked ubiquitination as a previously unrecognised regulatory layer controlling AMPK activation, metabolic adaptability, and the cellular response to energetic stress.
-
Evaluation of dasatinib and ponatinib for the control of CD123 CAR-T cell functionalities.
In Mol Ther Oncol on 18 December 2025 by Mantion, C. F., Biichle, S., et al.
PubMed
CD123 CAR-T cells (CAR123) represent a promising therapeutic approach for blastic plasmacytoid dendritic cell neoplasm (BPDCN) and CD123+ acute myeloid leukemia (AML). However, the pro-inflammatory environment resulting from CAR-T cell activation can induce CD123 upregulation on endothelial cells and potential on-target/off-tumor toxicity. We evaluated the capacity of two tyrosine kinase inhibitors (TKIs), dasatinib and ponatinib, to reversibly inhibit CAR-T cell functions. Using different in vitro models of CAR123 co-culture with BPDCN and AML cell lines, we show that both TKIs reduce CAR123 activation phenotype (CD69 and CD25), tumor necrosis factor α (TNF-α) and interferon-γ (IFN-γ) secretion; degranulation (CD107a); and killing of leukemia cells. Moreover, this inhibition was reversible after elimination of the TKIs. However, only dasatinib was effective at clinically relevant concentrations; 50 nM inhibited TNF-α and IFN-γ secretion, with only a slight reduction in cytotoxicity toward leukemia cells and allowed effective control of CAR-T cell cytotoxicity against endothelial cells in relation to the inhibition of cytokine secretion. Thus, dasatinib could be used to minimize potential CAR123 toxicity toward endothelial cells without compromising its anti-leukemic effects. However, a higher dose could be used to completely inhibit CAR-T cell functionality in the event of toxicity.
-
Validation of the i-Tracker Drug and Total Anti-Drug Antibody CLIA Assays on IDS-iSYS for Therapeutic Drug Monitoring in Adalimumab- and Infliximab-Treated Patients.
In Diagnostics (Basel) on 25 September 2025 by Dossou, A. S., Kang, S., et al.
PubMed
Background/Objectives: Adalimumab and Infliximab are biologics used to treat autoimmune diseases. Monitoring drug and anti-drug antibody (ADA) levels in patients helps optimize treatment. However, current quantitation methodologies for drug and total (free and drug-bound) ADAs often involve multi-step workflows. Automated systems can streamline the process. The i-Tracker chemiluminescent immunoassays (CLIA) are cartridge-based kits for quantifying serum levels of drugs such as Adalimumab, Infliximab, and associated ADAs. Herein, we aimed to establish performance characteristics of the i-Tracker Adalimumab, Infliximab, and total ADAs in serum on the random-access analyzer IDS-iSYS and to compare patient results with an electrochemiluminescent immunoassay (ECLIA)-based reference method. Methods: Remnant serum specimens, calibration material, or spiked serum were used to evaluate assay linearity, precision, functional sensitivity, and accuracy on the IDS-iSYS analyzer and to perform the method comparison. Results: The assays displayed linearity, accuracy, and up to 8% imprecision across clinically relevant analyte ranges. Compared to the reference method, the drug assays exhibited a strong linear fit (correlation coefficient > 0.95) with <±1.0 µg/mL mean bias. The total anti-Adalimumab assay demonstrated over 85% qualitative agreement. The total anti-Infliximab assay, however, showed higher detection rate of ADAs in Infliximab-treated patient specimens, yielding < 60% negative agreement with the reference method. Although i-Tracker total ADA assays exhibited drug sensitivity, they still detected ADAs in supratherapeutic drug concentrations. Conclusions: The i-Tracker assays demonstrated robust analytical performance, suggesting potential for clinical application. The method comparison underscored functional differences with the reference method, an important consideration when transitioning assay formats for monitoring Adalimumab- and Infliximab-treated patients.
-
TBK1 Targeting Is Identified as a Therapeutic Strategy to Enhance CAR T-Cell Efficacy Using Patient-Derived Organotypic Tumor Spheroids.
In Cancer Immunol Res on 3 February 2025 by Sun, Y., Maggs, L., et al.
PubMed
Novel therapeutic strategies are needed to improve the efficacy of chimeric antigen receptor (CAR) T cells as a treatment of solid tumors. Multiple tumor microenvironmental factors are thought to contribute to resistance to CAR T-cell therapy in solid tumors, and appropriate model systems to identify and examine these factors using clinically relevant biospecimens are limited. In this study, we examined the activity of B7-H3-directed CAR T cells (B7-H3.CAR-T) using 3D microfluidic cultures of patient-derived organotypic tumor spheroids (PDOTS) and then confirmed the activity of B7-H3.CAR T cells in PDOTS. Although B7-H3 expression in PDOTS was associated with B7-H3.CAR-T sensitivity, mechanistic studies revealed dynamic upregulation of co-inhibitory receptors on CAR T-cells following target cell encounter that led to CAR T-cell dysfunction and limited efficacy against B7-H3-expressing tumors. PD-1 blockade restored CAR T-cell activity in monotypic and organotypic tumor spheroids with improved tumor control and upregulation of effector cytokines. Given the emerging role of TANK-binding kinase 1 (TBK1) as an immune evasion gene, we examined the effect of TBK1 inhibition on CAR T-cell efficacy. Similar to PD-1 blockade, TBK1 inhibition restored CAR T-cell activity in monotypic and organotypic tumor spheroids, prevented CAR T-cell dysfunction, and enhanced CAR T-cell proliferation. Inhibition or deletion of TBK1 also enhanced the sensitivity of cancer cells to immune-mediated killing. Taken together, our results demonstrate the feasibility and utility of ex vivo profiling of CAR T cells using PDOTS and suggest that targeting TBK1 could be used to enhance CAR T-cell efficacy by overcoming tumor-intrinsic and -extrinsic resistance mechanisms.