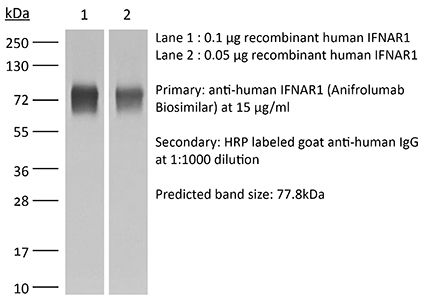
InVivoSIM anti-human IFNAR1 (Anifrolumab Biosimilar)
Product Description
Specifications
| Isotype | Human IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | RecombiMAb human IgG1 (K214R/L234F/L235E/P331S) isotype control, anti-hen egg lysozyme |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Human IFNAR1 |
| Reported Applications |
Functional assays Western blot ELISA |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤0.5EU/mg (≤0.0005EU/μg) Determined by LAL assay |
| Aggregation |
<5% Determined by SEC |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein A |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Product Citations
-
Inflammatory arthritis irAE may represent a unique autoimmune disease primarily driven by T cells but likely not autoantibodies.
In Sci Adv on 3 April 2026 by Zhu, X., Yu, Y., et al.
PubMed
The underlying immunopathogenesis of inflammatory arthritis (IA) immune-related adverse event (irAE) remains obscure. Unlike rheumatoid arthritis (RA), where autoantibodies and B cell dysfunction are central features, the contribution of humoral immunity to IA-irAE is unclear. Here, we performed immunophenotyping of peripheral blood from patients with IA-irAE and compared them with patients with seronegative RA, immune checkpoint inhibition-treated patients without irAE, and healthy controls. IA-irAE was marked with increased cytotoxic gene expression and metabolic activation in T cells and reduced CXCR3 and CCR6 expression in CD4+ T cells. Contrary to seronegative RA, patients with IA-irAE displayed no substantial elevation in autoantibody levels or atypical CD11c+CD21- B cells. IA-irAE was further characterized by elevated levels of interleukin-6 (IL-6), IL-12, and type I interferon, which correlated with the T cell activation phenotypes. Together, our findings define IA-irAE as a disease with certain immunological features distinctive from RA, representing a potentially T cell-driven, autoantibody-independent autoimmunity. These results offer insights into immune tolerance breakdown and therapeutic targeting in irAEs.
-
TNF-α induces type I IFN signalling to suppress neurogenesis and recruit T cells
In bioRxiv on 24 June 2025 by Nissen, T. A. D., Baig, A., et al.
-
Inflammatory arthritis immune related adverse events represent a unique autoimmune disease entity primarily driven by T cells, but likely not autoantibodies
In medRxiv on 6 June 2025 by Zhu, X., Yu, Y., et al.
-
Enhancing the Anticancer Activity of a Carcinoma-Directed Peptide-HLA-I Fusion Protein by Armoring with Mutein IFNα.
In Int J Mol Sci on 29 March 2025 by Samplonius, D. F., van Wijngarden, A. P., et al.
PubMed
Previously, we reported on the peptide-HLA-I fusion protein EpCAM-ReTARGTPR, which allows us to redirect the cytotoxic activity of pre-existing anti-CMV CD8pos T cell immunity to selectively eliminate EpCAMpos cancer cells. EpCAM-ReTARGTPR consists of the CMV pp65-derived peptide TPRVTGGGAM (TPR) fused in tandem with a soluble HLA-B*07:02/β2-microglobulin (β2M) molecule and an EpCAM-directed Fab antibody fragment. To further enhance its anticancer activity, we equipped EpCAM-ReTARGTPR with the immune-potentiating cytokine muteins IL2(H16A,F42A) and IFNαR149A, respectively. Both cytokines are engineered to have attenuated affinity for their respective cytokine receptors. Compared to EpCAM-ReTARGTPR, in vitro treatment of EpCAMpos carcinoma cell lines with EpCAM-ReTARGTPRvIL2 for 24 h increased the cytotoxic activity of PBMCs containing low levels of TPR-specific CD8pos T cells by ~15%, whereas EpCAM-ReTARGTPRIFNαR149A induced an increase of ~50%. Moreover, treatment for 120 h with EpCAM-ReTARGTPRIFNαR149A inhibited the proliferative capacity of the cancer cell lines OvCAR3 and PC3M by ~91% without compromising the viability of the TPR-specific CD8pos T cells and increased their capacity for IFNγ secretion. Importantly, EpCAM-ReTARGTPRIFNαR149A potently induced the elimination of primary EpCAMpos refractory carcinoma cells from a Merkel cell carcinoma (MCC) patient. Taken together, the armoring of the carcinoma-directed peptide-HLA-I fusion protein EpCAM-ReTARGTPR with IFNαR149A potently enhanced the efficacy of pre-existing anti-CMV CD8pos T cell immunity to selectively eliminate EpCAMpos cancer cells.