InVivoSIM anti-human EGFR (Cetuximab Biosimilar)
Product Description
Specifications
| Isotype | Human IgG1 |
|---|---|
| Recommended Isotype Control(s) | RecombiMAb human IgG1 isotype control, anti-hen egg lysozyme |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Human EGFR (ErbB1) |
| Reported Applications |
in vitro EGFR2+ cell depletion (ADCC assay) in vitro functional assay EGFR blockade in vivo imaging Immunohistochemistry (frozen) Immunofluorescence Flow Cytometry ELISA |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤0.5EU/mg (≤0.0005EU/μg) Determined by LAL assay |
| Aggregation* |
<5% Determined by SEC |
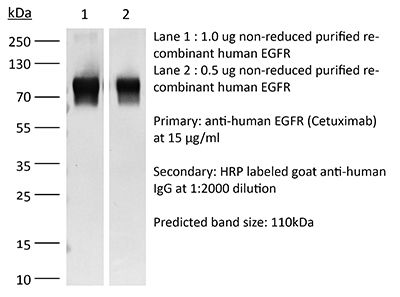
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein A |
| RRID | AB_2894723 |
| Molecular Weight | 150 kDa |
| Murine Pathogen Tests |
Ectromelia/Mousepox Virus: Negative Hantavirus: Negative K Virus: Negative Lactate Dehydrogenase-Elevating Virus: Negative Lymphocytic Choriomeningitis virus: Negative Mouse Adenovirus: Negative Mouse Cytomegalovirus: Negative Mouse Hepatitis Virus: Negative Mouse Minute Virus: Negative Mouse Norovirus: Negative Mouse Parvovirus: Negative Mouse Rotavirus: Negative Mycoplasma Pulmonis: Negative Pneumonia Virus of Mice: Negative Polyoma Virus: Negative Reovirus Screen: Negative Sendai Virus: Negative Theiler’s Murine Encephalomyelitis: Negative |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Gupta S, Pal R, Schmidt EJ, Krishnamoorthy M, Leporati A, Kumar ATN, Bogdanov A (2024). "Miniaturized Fab' imaging probe derived from a clinical antibody: Characterization and imaging in CRISPRi-attenuated mammary tumor models" iScience 27(8):11
PubMed
Clinical imaging-assisted oncosurgical navigation requires cancer-specific miniaturized optical imaging probes. We report a near-infrared (NIR) Fab'-based epidermal growth factor receptor (EGFR)-specific probe carrying 3 NIR fluorophores (Fab'-800CW), which retained high-affinity binding to EGFR ectodomain (equilibrium KD E = 1 nM). Fab'-800CW showed a robust 4-times gain of fluorescence intensity (FI) and a 20% lifetime (FLT) increase under the conditions mimicking intracellular degradation. The probe was tested by using triple-negative breast cancer (TNBC) cell lines obtained by applying CRISPR interference (CRISPRi) effect of EGFR-targeting sgRNA and dCas9-KRAB chimera coexpression in MDA-MB-231 cells (WT cells). FI imaging in cell culture proved a 50% EGFR expression attenuation by CRISPRi. FI imaging in animals harboring attenuated or WT TNBC tumors with ex vivo corroboration identified differences between WT and CRISPRi tumors FI at 30 min post injection. Our results suggest the feasibility of EGFR expression imaging using a Fab'-based probe relevant for imaging-guided cancer surgery.
-
Linde IL, Prestwood TR, Qiu J, Pilarowski G, Linde MH, Zhang X, Shen L, Reticker-Flynn NE, Chiu DK, Sheu LY, Van Deursen S, Tolentino LL, Song WC, Engleman EG (2023). "Neutrophil-activating therapy for the treatment of cancer" Cancer Cell 41(2):356-3
PubMed
Despite their cytotoxic capacity, neutrophils are often co-opted by cancers to promote immunosuppression, tumor growth, and metastasis. Consequently, these cells have received little attention as potential cancer immunotherapeutic agents. Here, we demonstrate in mouse models that neutrophils can be harnessed to induce eradication of tumors and reduce metastatic seeding through the combined actions of tumor necrosis factor, CD40 agonist, and tumor-binding antibody. The same combination activates human neutrophils in vitro, enabling their lysis of human tumor cells. Mechanistically, this therapy induces rapid mobilization and tumor infiltration of neutrophils along with complement activation in tumors. Complement component C5a activates neutrophils to produce leukotriene B4, which stimulates reactive oxygen species production via xanthine oxidase, resulting in oxidative damage and T cell-independent clearance of multiple tumor types. These data establish neutrophils as potent anti-tumor immune mediators and define an inflammatory pathway that can be harnessed to drive neutrophil-mediated eradication of cancer.
-
Lee DH, Ahn H, Sim HI, Choi E, Choi S, Jo Y, Yun B, Song HK, Oh SJ, Denda-Nagai K, Park CS, Irimura T, Park Y, Jin HS (2023). "A CRISPR activation screen identifies MUC-21 as critical for resistance to NK and T cell-mediated cytotoxicity" J Exp Clin
PubMed
Background: Immunotherapy has significantly advanced cancer treatments, but many patients do not respond to it, partly due to immunosuppressive mechanisms used by tumor cells. These cells employ immunosuppressive ligands to evade detection and elimination by the immune system. Therefore, the discovery and characterization of novel immunosuppressive ligands that facilitate immune evasion are crucial for developing more potent anti-cancer therapies. Methods: We conducted gain-of-function screens using a CRISPRa (CRISPR activation) library that covered the entire human transmembrane sub-genome to identify surface molecules capable of hindering NK-mediated cytotoxicity. The immunosuppressive role and mechanism of MUC21 were validated using NK and T cell mediated cytotoxicity assays. Bioinformatics tools were employed to assess the clinical implications of mucin-21 (MUC21) in cancer cell immunity. Results: Our genetic screens revealed that MUC21 expression on cancer cell surfaces inhibits both the cytotoxic activity of NK cells and antibody-dependent cellular cytotoxicity, but not affecting complement-dependent cytotoxicity. Additionally, MUC21 expression hinders T cell activation by impeding antigen recognition, thereby diminishing the effectiveness of the immune checkpoint inhibitor, anti-PD-L1. Moreover, MUC21 expression suppress the antitumor function of both CAR-T cells and CAR-NK cells. Mechanistically, MUC21 facilitates immune evasion by creating steric hindrance, preventing interactions between cancer and immune cells. Bioinformatics analysis revealed elevated MUC21 expression in lung cancer, which correlated with reduced infiltration and activation of cytotoxic immune cells. Intriguingly, MUC21 expression was higher in non-small cell lung cancer (NSCLC) tumors that were non-responsive to anti-PD-(L)1 treatment compared to responsive tumors. Conclusions: These findings indicate that surface MUC21 serves as a potent immunosuppressive ligand, shielding cancer cells from NK and CD8+T cell attacks. This suggests that inhibiting MUC21 could be a promising strategy to improve cancer immunotherapy.
-
Iadonato S, Ovechkina Y, Lustig K, Cross J, Eyde N, Frazier E, Kabi N, Katz C, Lance R, Peckham D, Sridhar S, Talbaux C, Tihista I, Xu M, Guillaudeux T (2023). "A highly potent anti-VISTA antibody KVA12123 - a new immune checkpoint inhibitor and a pr
PubMed
Background: Immune checkpoint therapies have led to significant breakthroughs in cancer patient treatment in recent years. However, their efficiency is variable, and resistance to immunotherapies is common. VISTA is an immune-suppressive checkpoint inhibitor of T cell response belonging to the B7 family and a promising novel therapeutic target. VISTA is expressed in the immuno-suppressive tumor microenvironment, primarily by myeloid lineage cells, and its genetic knockout or antibody blockade restores an efficient antitumor immune response. Methods: Fully human monoclonal antibodies directed against VISTA were produced after immunizing humanized Trianni mice and single B cell sequencing. Anti-VISTA antibodies were evaluated for specificity, cross-reactivity, monocyte and T cell activation, Fc-effector functions, and antitumor efficacy using in vitro and in vivo models to select the KVA12123 antibody lead candidate. The pharmacokinetics and safety profiles of KVA12123 were evaluated in cynomolgus monkeys. Results: Here, we report the development of a clinical candidate anti-VISTA monoclonal antibody, KVA12123. KVA12123 showed high affinity binding to VISTA through a unique epitope distinct from other clinical-stage anti-VISTA monoclonal antibodies. This clinical candidate demonstrated high specificity against VISTA with no cross-reactivity detected against other members of the B7 family. KVA12123 blocked VISTA binding to its binding partners. KVA12123 induced T cell activation and demonstrated NK-mediated monocyte activation. KVA12123 treatment mediated strong single-agent antitumor activity in several syngeneic tumor models and showed enhanced efficacy in combination with anti-PD-1 treatment. This clinical candidate was engineered to improve its pharmacokinetic characteristics and reduce Fc-effector functions. It was well-tolerated in preclinical toxicology studies in cynomolgus monkeys, where hematology, clinical chemistry evaluations, and clinical observations revealed no indicators of toxicity. No cytokines associated with cytokine release syndrome were elevated. Conclusion: These results establish that KVA12123 is a promising drug candidate with a distinct but complementary mechanism of action of the first generation of immune checkpoint inhibitors. This antibody is currently evaluated alone and in combination with pembrolizumab in a Phase 1/2 open-label clinical trial in patients with advanced solid tumors.
Product Citations
-
A monoclonal antibody that inhibits the shedding of CD16a and CD16b and promotes antibody-dependent cellular cytotoxicity against tumors.
In Nat Commun on 11 November 2025 by Bortoleti, B. T. D. S., Quasem, S., et al.
PubMed
CD16a triggers antibody-dependent cellular cytotoxicity (ADCC) and phagocytosis by natural killer (NK) cells and macrophages in anti-tumor immunity. However, CD16a undergoes cleavage by ADAM17 that dampens its anti-tumor immunity. We here develop a monoclonal antibody (F9H4) that binds to CD16a and inhibits its cleavage. F9H4 retains CD16a on the surface of NK cells and macrophages, without triggering or blocking CD16a. F9H4 also binds to and inhibits shedding of CD16b by neutrophils, and inhibits CD16a/b shedding by leukocytes in tumor samples from lung cancer patients. F9H4 promotes ADCC against lung cancer cells that are opsonized by cetuximab, an epidermal growth factor receptor antibody that engages CD16a. F9H4 synergizes with cetuximab to inhibit human lung adenocarcinoma development in immunodeficient mice reconstituted with human NK cells. F9H4 combining with cetuximab also inhibits murine lung carcinoma growth in Fc gamma receptor-humanized mice, and such effect is mediated by NK cells and macrophages. The efficacy of F9H4+cetuximab in lung cancer models is the proof-of-concept for this new approach that promotes anti-tumor functions of Fc-enabled antibodies.
-
Miniaturized Fab' imaging probe derived from a clinical antibody: Characterization and imaging in CRISPRi-attenuated mammary tumor models.
In iScience on 16 August 2024 by Gupta, S., Pal, R., et al.
PubMed
Clinical imaging-assisted oncosurgical navigation requires cancer-specific miniaturized optical imaging probes. We report a near-infrared (NIR) Fab'-based epidermal growth factor receptor (EGFR)-specific probe carrying 3 NIR fluorophores (Fab'-800CW), which retained high-affinity binding to EGFR ectodomain (equilibrium KDE = 1 nM). Fab'-800CW showed a robust 4-times gain of fluorescence intensity (FI) and a 20% lifetime (FLT) increase under the conditions mimicking intracellular degradation. The probe was tested by using triple-negative breast cancer (TNBC) cell lines obtained by applying CRISPR interference (CRISPRi) effect of EGFR-targeting sgRNA and dCas9-KRAB chimera coexpression in MDA-MB-231 cells (WT cells). FI imaging in cell culture proved a 50% EGFR expression attenuation by CRISPRi. FI imaging in animals harboring attenuated or WT TNBC tumors with ex vivo corroboration identified differences between WT and CRISPRi tumors FI at 30 min post injection. Our results suggest the feasibility of EGFR expression imaging using a Fab'-based probe relevant for imaging-guided cancer surgery.
-
A highly potent anti-VISTA antibody KVA12123 - a new immune checkpoint inhibitor and a promising therapy against poorly immunogenic tumors.
In Front Immunol on 28 December 2023 by Iadonato, S., Ovechkina, Y., et al.
PubMed
Immune checkpoint therapies have led to significant breakthroughs in cancer patient treatment in recent years. However, their efficiency is variable, and resistance to immunotherapies is common. VISTA is an immune-suppressive checkpoint inhibitor of T cell response belonging to the B7 family and a promising novel therapeutic target. VISTA is expressed in the immuno-suppressive tumor microenvironment, primarily by myeloid lineage cells, and its genetic knockout or antibody blockade restores an efficient antitumor immune response.
-
Neutrophil-activating therapy for the treatment of cancer.
In Cancer Cell on 13 February 2023 by Linde, I. L., Prestwood, T. R., et al.
PubMed
Despite their cytotoxic capacity, neutrophils are often co-opted by cancers to promote immunosuppression, tumor growth, and metastasis. Consequently, these cells have received little attention as potential cancer immunotherapeutic agents. Here, we demonstrate in mouse models that neutrophils can be harnessed to induce eradication of tumors and reduce metastatic seeding through the combined actions of tumor necrosis factor, CD40 agonist, and tumor-binding antibody. The same combination activates human neutrophils in vitro, enabling their lysis of human tumor cells. Mechanistically, this therapy induces rapid mobilization and tumor infiltration of neutrophils along with complement activation in tumors. Complement component C5a activates neutrophils to produce leukotriene B4, which stimulates reactive oxygen species production via xanthine oxidase, resulting in oxidative damage and T cell-independent clearance of multiple tumor types. These data establish neutrophils as potent anti-tumor immune mediators and define an inflammatory pathway that can be harnessed to drive neutrophil-mediated eradication of cancer.