InVivoMAb anti-mouse TSLP
Product Description
Specifications
| Isotype | Rat IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
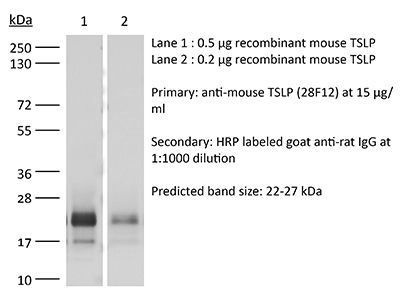
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Purified murine TSLP |
| Reported Applications |
in vivo TSLP neutralization in vitro TSLP neutralization |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Purification | Protein G |
| RRID | AB_2927516 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Stier, M. T., et al (2016). "Respiratory syncytial virus infection activates IL-13-producing group 2 innate lymphoid cells through thymic stromal lymphopoietin" J Allergy Clin Immunol 138(3): 814-824 e811.
PubMed
BACKGROUND: Respiratory syncytial virus (RSV) is a major health care burden with a particularly high worldwide morbidity and mortality rate among infants. Data suggest that severe RSV-associated illness is in part caused by immunopathology associated with a robust type 2 response. OBJECTIVE: We sought to determine the capacity of RSV infection to stimulate group 2 innate lymphoid cells (ILC2s) and the associated mechanism in a murine model. METHODS: Wild-type (WT) BALB/c, thymic stromal lymphopoietin receptor (TSLPR) knockout (KO), or WT mice receiving an anti-TSLP neutralizing antibody were infected with the RSV strain 01/2-20. During the first 4 to 6 days of infection, lungs were collected for evaluation of viral load, protein concentration, airway mucus, airway reactivity, or ILC2 numbers. Results were confirmed with 2 additional RSV clinical isolates, 12/11-19 and 12/12-6, with known human pathogenic potential. RESULTS: RSV induced a 3-fold increase in the number of IL-13-producing ILC2s at day 4 after infection, with a concurrent increase in total lung IL-13 levels. Both thymic stromal lymphopoietin (TSLP) and IL-33 levels were increased 12 hours after infection. TSLPR KO mice did not mount an IL-13-producing ILC2 response to RSV infection. Additionally, neutralization of TSLP significantly attenuated the RSV-induced IL-13-producing ILC2 response. TSLPR KO mice displayed reduced lung IL-13 protein levels, decreased airway mucus and reactivity, attenuated weight loss, and similar viral loads as WT mice. Both 12/11-19 and 12/12-6 similarly induced IL-13-producing ILC2s through a TSLP-dependent mechanism. CONCLUSION: These data demonstrate that multiple pathogenic strains of RSV induce IL-13-producing ILC2 proliferation and activation through a TSLP-dependent mechanism in a murine model and suggest the potential therapeutic targeting of TSLP during severe RSV infection.
-
Mosconi, I., et al (2013). "Intestinal bacteria induce TSLP to promote mutualistic T-cell responses" Mucosal Immunol 6(6): 1157-1167.
PubMed
Thymic stromal lymphopoietin (TSLP) is constitutively expressed in the intestine and is known to regulate inflammation in models of colitis. We show that steady-state TSLP expression requires intestinal bacteria and has an important role in limiting the expansion of colonic T helper type 17 (Th17) cells. Inappropriate expansion of the colonic Th17 cells occurred in response to an entirely benign intestinal microbiota, as determined following the colonization of germ-free C57BL/6 or TSLPR(-/-) mice with the altered Schaedler flora (ASF). TSLP-TSLPR (TSLP receptor) interactions also promoted the expansion of colonic Helios(-)Foxp3(+) regulatory T cells, necessary for the control of inappropriate Th17 responses following ASF bacterial colonization. In summary, these data reveal an important role for TSLP-TSLPR signaling in promoting steady-state mutualistic T-cell responses following intestinal bacterial colonization.
-
Sokol, C. L., et al (2008). "A mechanism for the initiation of allergen-induced T helper type 2 responses" Nat Immunol 9(3): 310-318.
PubMed
Both metazoan parasites and simple protein allergens induce T helper type 2 (TH2) immune responses, but the mechanisms by which the innate immune system senses these stimuli are unknown. In addition, the cellular source of cytokines that control TH2 differentiation in vivo has not been defined. Here we showed that basophils were activated and recruited to the draining lymph nodes specifically in response to TH2-inducing allergen challenge. Furthermore, we demonstrate that the basophil was the accessory cell type required for TH2 induction in response to protease allergens. Finally, we show that basophils were directly activated by protease allergens and produced TH2-inducing cytokines, including interleukin 4 and thymic stromal lymphopoietin, which are involved in TH2 differentiation in vivo.
Product Citations
-
Activation of TSLP-IL-9 Axis Hinders the Antifibrotic Effect of ST2 Deficiency in Pulmonary Fibrosis.
In Int J Mol Sci on 5 December 2025 by Atamas, S. P., Lockatell, V., et al.
PubMed
Previous studies have suggested that activation of the IL-33/ST2 axis as well as elevated expression of the full-length IL-33 precursor acting in an ST2-independent fashion both contribute to pulmonary fibrosis. The protective effect of genetic ST2 deficiency on pulmonary fibrosis is known to be partial, with unclear mechanisms preventing a more complete protection. Here, we report that ST2 deficiency failed to fully protect the lungs from excess collagen accumulation after the profibrotic bleomycin injury and simultaneously facilitated elevations in pulmonary levels of a previously suggested profibrotic mediator, IL-9, as well as a known activator of IL-9 expression, TSLP. Pulmonary CD4+ T cells were the main producers of IL-9. Neutralizing antibody-mediated in vivo blockade of TSLP potently attenuated pulmonary levels of both IL-9 and collagen in the bleomycin injury model in wild-type and particularly ST2-deficient mice. All these observations were markedly pronounced in mice with single deficiency of ST2 and the overall pattern of findings was also preserved in mice with dual deficiency of ST2 and IL-33. It was concluded that the antifibrotic effect of ST2 deficiency is hindered by the simultaneous activation of the TSLP-IL-9 axis in experimental bleomycin-induced pulmonary fibrosis. These findings inform further development of antifibrotic therapies.