InVivoMAb anti-mouse IFNγ
Product Description
Specifications
| Isotype | Rat IgG1, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG1 isotype control, anti-horseradish peroxidase |
| Recommended Dilution Buffer | InVivoPure pH 8.0 Dilution Buffer |
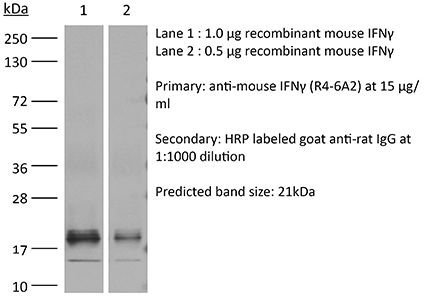
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Partially-purified native mouse IFNγ |
| Reported Applications |
in vivo IFNγ neutralization in vitro IFNγ neutralization |
| Formulation |
PBS, pH 8.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107692 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Conde, P., et al (2015). "DC-SIGN(+) Macrophages Control the Induction of Transplantation Tolerance" Immunity 42(6): 1143-1158.
PubMed
Tissue effector cells of the monocyte lineage can differentiate into different cell types with specific cell function depending on their environment. The phenotype, developmental requirements, and functional mechanisms of immune protective macrophages that mediate the induction of transplantation tolerance remain elusive. Here, we demonstrate that costimulatory blockade favored accumulation of DC-SIGN-expressing macrophages that inhibited CD8(+) T cell immunity and promoted CD4(+)Foxp3(+) Treg cell expansion in numbers. Mechanistically, that simultaneous DC-SIGN engagement by fucosylated ligands and TLR4 signaling was required for production of immunoregulatory IL-10 associated with prolonged allograft survival. Deletion of DC-SIGN-expressing macrophages in vivo, interfering with their CSF1-dependent development, or preventing the DC-SIGN signaling pathway abrogated tolerance. Together, the results provide new insights into the tolerogenic effects of costimulatory blockade and identify DC-SIGN(+) suppressive macrophages as crucial mediators of immunological tolerance with the concomitant therapeutic implications in the clinic.
-
Wensveen, F. M., et al (2015). "NK cells link obesity-induced adipose stress to inflammation and insulin resistance" Nat Immunol 16(4): 376-385.
PubMed
An important cause of obesity-induced insulin resistance is chronic systemic inflammation originating in visceral adipose tissue (VAT). VAT inflammation is associated with the accumulation of proinflammatory macrophages in adipose tissue, but the immunological signals that trigger their accumulation remain unknown. We found that a phenotypically distinct population of tissue-resident natural killer (NK) cells represented a crucial link between obesity-induced adipose stress and VAT inflammation. Obesity drove the upregulation of ligands of the NK cell-activating receptor NCR1 on adipocytes; this stimulated NK cell proliferation and interferon-gamma (IFN-gamma) production, which in turn triggered the differentiation of proinflammatory macrophages and promoted insulin resistance. Deficiency of NK cells, NCR1 or IFN-gamma prevented the accumulation of proinflammatory macrophages in VAT and greatly ameliorated insulin sensitivity. Thus NK cells are key regulators of macrophage polarization and insulin resistance in response to obesity-induced adipocyte stress.
-
Maltby, S., et al (2014). "Production and differentiation of myeloid cells driven by proinflammatory cytokines in response to acute pneumovirus infection in mice" J Immunol 193(8): 4072-4082.
PubMed
Respiratory virus infections are often pathogenic, driving severe inflammatory responses. Most research has focused on localized effects of virus infection and inflammation. However, infection can induce broad-reaching, systemic changes that are only beginning to be characterized. In this study, we assessed the impact of acute pneumovirus infection in C57BL/6 mice on bone marrow hematopoiesis. We hypothesized that inflammatory cytokine production in the lung upregulates myeloid cell production in response to infection. We demonstrate a dramatic increase in the percentages of circulating myeloid cells, which is associated with pronounced elevations in inflammatory cytokines in serum (IFN-gamma, IL-6, CCL2), bone (TNF-alpha), and lung tissue (TNF-alpha, IFN-gamma, IL-6, CCL2, CCL3, G-CSF, osteopontin). Increased hematopoietic stem/progenitor cell percentages (Lineage(-)Sca-I(+)c-kit(+)) were also detected in the bone marrow. This increase was accompanied by an increase in the proportions of committed myeloid progenitors, as determined by colony-forming unit assays. However, no functional changes in hematopoietic stem cells occurred, as assessed by competitive bone marrow reconstitution. Systemic administration of neutralizing Abs to either TNF-alpha or IFN-gamma blocked expansion of myeloid progenitors in the bone marrow and also limited virus clearance from the lung. These findings suggest that acute inflammatory cytokines drive production and differentiation of myeloid cells in the bone marrow by inducing differentiation of committed myeloid progenitors. Our findings provide insight into the mechanisms via which innate immune responses regulate myeloid cell progenitor numbers in response to acute respiratory virus infection.
-
Walsh, K. B., et al (2014). "Animal model of respiratory syncytial virus: CD8+ T cells cause a cytokine storm that is chemically tractable by sphingosine-1-phosphate 1 receptor agonist therapy" J Virol 88(11): 6281-6293.
PubMed
The cytokine storm is an intensified, dysregulated, tissue-injurious inflammatory response driven by cytokine and immune cell components. The cytokine storm during influenza virus infection, whereby the amplified innate immune response is primarily responsible for pulmonary damage, has been well characterized. Now we describe a novel event where virus-specific T cells induce a cytokine storm. The paramyxovirus pneumonia virus of mice (PVM) is a model of human respiratory syncytial virus (hRSV). Unexpectedly, when C57BL/6 mice were infected with PVM, the innate inflammatory response was undetectable until day 5 postinfection, at which time CD8(+) T cells infiltrated into the lung, initiating a cytokine storm by their production of gamma interferon (IFN-gamma) and tumor necrosis factor alpha (TNF-alpha). Administration of an immunomodulatory sphingosine-1-phosphate (S1P) receptor 1 (S1P1R) agonist significantly inhibited PVM-elicited cytokine storm by blunting the PVM-specific CD8(+) T cell response, resulting in diminished pulmonary disease and enhanced survival. IMPORTANCE: A dysregulated overly exuberant immune response, termed a “cytokine storm,” accompanies virus-induced acute respiratory diseases (VARV), is primarily responsible for the accompanying high morbidity and mortality, and can be controlled therapeutically in influenza virus infection of mice and ferrets by administration of sphingosine-1-phosphate 1 receptor (S1P1R) agonists. Here, two novel findings are recorded. First, in contrast to influenza infection, where the cytokine storm is initiated early by the innate immune system, for pneumonia virus of mice (PVM), a model of RSV, the cytokine storm is initiated late in infection by the adaptive immune response: specifically, by virus-specific CD8 T cells via their release of IFN-gamma and TNF-alpha. Blockading these cytokines with neutralizing antibodies blunts the cytokine storm and protects the host. Second, PVM infection is controlled by administration of an S1P1R agonist.
Product Citations
-
Targeting immune cells in the aged brain reveals that engineered cytokine IL-10 enhances neurogenesis and improves cognition.
In Immunity on 10 February 2026 by Navarro Negredo, P., You, J., et al.
PubMed
The immune system could play an important role in the age-related decline in brain function, yet specific immune-based strategies to enhance brain resilience in older individuals are lacking. Here, we combined engineered proteins and direct brain delivery to target immune cell populations within the old brain. We detected T cells with an exhaustion signature in the old brain and targeted them with a potent engineered checkpoint inhibitor (RIPR-PD1). This led to T cell expansion and strong pro-inflammatory responses in many brain cell types, notably microglia. To rescue age-related inflammatory imbalances in microglia, we used the anti-inflammatory cytokine interleukin (IL)-10. IL-10 boosted anti-inflammatory responses in old microglia, but it also triggered pro-inflammatory signaling. An engineered IL-10 variant that uncouples pro- and anti-inflammatory responses positively impacted the transcriptome of multiple cell types, enhanced neurogenesis, and improved cognition in aged mice. Our findings pave the way for immunotherapies for the aged brain.
-
The E3 ligases Itch and WWP2 regulate autoimmune neuroinflammation by controlling TH2 to TH17 cell conversion via interleukin-4-STAT5 axis in mice.
In Nat Commun on 23 January 2026 by Zhao, M., Zhang, C., et al.
PubMed
Multiple sclerosis (MS) is a neurodegenerative autoimmune disease primarily mediated by T helper 17 (TH17) cells. We previously showed that Itch/WWP2 double knockout (DKO) T cells produce high levels of type 2 cytokines, driving spontaneous autoinflammation. Here, we report that DKO TH2-high carrying autoantigen-specific TCR (2D2) develop atypical spontaneous experimental autoimmune encephalomyelitis (EAE), with CD4+ T cells simultaneously producing IL-4 and GM-CSF, directly causing neuroinflammation. Unexpectedly, IL-4 deletion in DKO TH2-high 2D2 mice exacerbates TH17-driven classical EAE, indicating a TH2 to TH17 conversion. Furthermore, we show that the JAK3/STAT5 signaling pathway is critical for maintaining TH2 lineage stability by modulating Blimp1 and c-Maf thereby suppressing TH17 differentiation. Importantly, we find that this phenomenon can also be observed in dupilumab-treated patients with atopic dermatitis who develop psoriasis. Thus, our findings uncover the molecular antagonism and plasticity in the TH2 and TH17 cell programs and identify potential therapeutic targets for modulating TH2 and TH17 cell responses in autoimmune diseases.
-
Potentiating CAR-T bystander killing by enhanced Fas/FasL signaling mitigates antigen escape in heterogeneous tumors
In bioRxiv on 24 September 2025 by Lin, M. J., Chorazeczewski, J. K., et al.
-
Sorafenib-treated Th9 cells exhibit superior anti-tumor effect in lung metastasis.
In Sci Rep on 7 August 2025 by Yuan, F., Wang, H., et al.
PubMed
The lung is the second most common organ of tumor metastasis, with limited treatment options and disappointing treatment outcomes. T helper 9 (Th9) cells have been reported to be effective in the elimination of solid tumors and exhibit superior antitumor properties to T helper 1 (Th1) and T helper 17 (Th17) cells, which makes a potential candidate for Adoptive cell therapy (ACT) against lung metastasis. However, how Th9 cells can be massively expanded in vitro is not clear. Here, we found Sorafenib (Sora) can increase the efficiency of Th9 in vitro through ERK signaling. In addition, Sora-treated Th9 cells exhibit superior anti-tumor effects by reshaping tumor immune microenvironment in lung metastasis mice model. Thus, our findings provide a potential method to expand Th9 cells in vitro for lung metastasis treatment.