InVivoMAb anti-mouse CD16/CD32
Product Description
Specifications
| Isotype | Rat IgG2b, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2b isotype control, anti-keyhole limpet hemocyanin |
| Recommended Dilution Buffer | InVivoPure pH 8.0 Dilution Buffer |
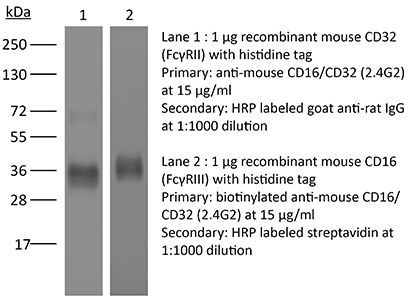
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | BALB/c mouse macrophage cell line J774 |
| Reported Applications |
in vivo Fc receptor blocking Fc receptor blocking Flow cytometry Immunofluorescence |
| Formulation |
PBS, pH 8.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_2736987 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Pasqual, G., et al (2018). "Monitoring T cell-dendritic cell interactions in vivo by intercellular enzymatic labelling" Nature 553(7689): 496-500.
PubMed
Interactions between different cell types are essential for multiple biological processes, including immunity, embryonic development and neuronal signalling. Although the dynamics of cell-cell interactions can be monitored in vivo by intravital microscopy, this approach does not provide any information on the receptors and ligands involved or enable the isolation of interacting cells for downstream analysis. Here we describe a complementary approach that uses bacterial sortase A-mediated cell labelling across synapses of immune cells to identify receptor-ligand interactions between cells in living mice, by generating a signal that can subsequently be detected ex vivo by flow cytometry. We call this approach for the labelling of ‘kiss-and-run’ interactions between immune cells ‘Labelling Immune Partnerships by SorTagging Intercellular Contacts’ (LIPSTIC). Using LIPSTIC, we show that interactions between dendritic cells and CD4(+) T cells during T-cell priming in vivo occur in two distinct modalities: an early, cognate stage, during which CD40-CD40L interactions occur specifically between T cells and antigen-loaded dendritic cells; and a later, non-cognate stage during which these interactions no longer require prior engagement of the T-cell receptor. Therefore, LIPSTIC enables the direct measurement of dynamic cell-cell interactions both in vitro and in vivo. Given its flexibility for use with different receptor-ligand pairs and a range of detectable labels, we expect that this approach will be of use to any field of biology requiring quantification of intercellular communication.
-
Arbelaez, C. A., et al (2015). "IL-7/IL-7 Receptor Signaling Differentially Affects Effector CD4+ T Cell Subsets Involved in Experimental Autoimmune Encephalomyelitis" J Immunol 195(5): 1974-1983.
PubMed
IL-17-producing CD4(+) T (Th17) cells, along with IFN-gamma-expressing Th1 cells, represent two major pathogenic T cell subsets in experimental autoimmune encephalomyelitis (EAE), the animal model of multiple sclerosis (MS). The cytokines and transcription factors involved in the development and effector functions of Th1 and Th17 cells have been largely characterized. Among them, IL-23 is essential for the generation of stable and encephalitogenic Th17 cells and for the development of EAE. The IL-7/IL-7R signaling axis participates in cell survival, and perturbation of this pathway has been associated with enhanced susceptibility to MS. A link between IL-23-driven pathogenic T cells and IL-7/IL-7R signaling has previously been proposed, but has not been formally addressed. In the current study, we showed that Th17 cells from mice with EAE express high levels of IL-7Ralpha compared with Th1 cells. Using mice that constitutively express IL-7Ralpha on T cells, we determined that sustained IL-7R expression in IL-23R-deficient mice could not drive pathogenic T cells and the development of EAE. IL-7 inhibited the differentiation of Th17 cells, but promoted IFN-gamma and GM-CSF secretion in vitro. In vivo IL-7/anti-IL-7 mAb complexes selectively expanded and enhanced the proliferation of CXCR3-expressing Th1 cells, but did not impact Th17 cells and EAE development in wild-type and IL-23R-deficient mice. Importantly, high IL-7 expression was detected in the CNS during EAE and could drive the plasticity of Th17 cells to IFN-gamma-producing T cells. Together, these data address the contribution of IL-23/IL-23R and IL-7/IL-7R signaling in Th17 and Th1 cell dynamics during CNS autoimmunity.
-
Liu, X., et al (2015). "CD47 blockade triggers T cell-mediated destruction of immunogenic tumors" Nat Med 21(10): 1209-1215.
PubMed
Macrophage phagocytosis of tumor cells mediated by CD47-specific blocking antibodies has been proposed to be the major effector mechanism in xenograft models. Here, using syngeneic immunocompetent mouse tumor models, we reveal that the therapeutic effects of CD47 blockade depend on dendritic cell but not macrophage cross-priming of T cell responses. The therapeutic effects of anti-CD47 antibody therapy were abrogated in T cell-deficient mice. In addition, the antitumor effects of CD47 blockade required expression of the cytosolic DNA sensor STING, but neither MyD88 nor TRIF, in CD11c(+) cells, suggesting that cytosolic sensing of DNA from tumor cells is enhanced by anti-CD47 treatment, further bridging the innate and adaptive responses. Notably, the timing of administration of standard chemotherapy markedly impacted the induction of antitumor T cell responses by CD47 blockade. Together, our findings indicate that CD47 blockade drives T cell-mediated elimination of immunogenic tumors.
-
Peske, J. D., et al (2015). "Effector lymphocyte-induced lymph node-like vasculature enables naive T-cell entry into tumours and enhanced anti-tumour immunity" Nat Commun 6: 7114.
PubMed
The presence of lymph node (LN)-like vasculature in tumours, characterized by expression of peripheral node addressin and chemokine CCL21, is correlated with T-cell infiltration and positive prognosis in breast cancer and melanoma patients. However, mechanisms controlling the development of LN-like vasculature and how it might contribute to a beneficial outcome for cancer patients are unknown. Here we demonstrate that LN-like vasculature is present in murine models of melanoma and lung carcinoma. It enables infiltration by naive T cells that significantly delay tumour outgrowth after intratumoral activation. Development of this vasculature is controlled by a mechanism involving effector CD8 T cells and NK cells that secrete LTalpha3 and IFNgamma. LN-like vasculature is also associated with organized aggregates of B lymphocytes and gp38(+) fibroblasts, which resemble tertiary lymphoid organs that develop in models of chronic inflammation. These results establish LN-like vasculature as both a consequence of and key contributor to anti-tumour immunity.
Product Citations
-
Synthetic super-enhancers enable precision viral immunotherapy.
In Nature on 1 May 2026 by Koeber, U., Matjusaitis, M., et al.
PubMed
Cell-type-specific promoters are used in gene therapy to restrict expression of the therapeutic payload. However, these promoters often have suboptimal strength, selectivity and size. Here, leveraging recent insights into the function of enhancers, we developed synthetic super-enhancers (SSEs) by assembling functionally validated enhancer fragments into multipart arrays. Focusing on the core SOX2-driven and SOX9-driven transcriptional regulatory network in glioblastoma stem cells (GSCs)1, we engineered SSEs with robust activity and high selectivity. Single-cell profiling, biochemical analyses and genome-binding data indicated that SSEs integrate neurodevelopmental and signalling-state transcription factors to trigger the formation of large multimeric complexes of transcription factors. Moreover, GSC-selective expression of a combination of cytotoxic (HSV-TK and ganciclovir) and immunomodulatory (IL-12) payloads, delivered using adeno-associated virus vectors, as a single treatment led to curative outcomes in a mouse model of aggressive glioblastoma. Notably, IL-12 induced an immunological memory that prevented tumour recurrence. The activity and selectivity of the adeno-associated virus and SSE were validated using primary human glioblastoma tissue and normal cortex samples. In summary, SSEs harness the unique core transcriptional programs that define the GSC phenotype and enable precision immune activation. This approach may have broader applications in other contexts when precise control of transgene expression in specific cell states is necessary.
-
Synthetic super-enhancers enable precision viral immunotherapy.
In Nature on 1 May 2026 by Koeber, U., Matjusaitis, M., et al.
PubMed
Cell-type-specific promoters are used in gene therapy to restrict expression of the therapeutic payload. However, these promoters often have suboptimal strength, selectivity and size. Here, leveraging recent insights into the function of enhancers, we developed synthetic super-enhancers (SSEs) by assembling functionally validated enhancer fragments into multipart arrays. Focusing on the core SOX2-driven and SOX9-driven transcriptional regulatory network in glioblastoma stem cells (GSCs)1, we engineered SSEs with robust activity and high selectivity. Single-cell profiling, biochemical analyses and genome-binding data indicated that SSEs integrate neurodevelopmental and signalling-state transcription factors to trigger the formation of large multimeric complexes of transcription factors. Moreover, GSC-selective expression of a combination of cytotoxic (HSV-TK and ganciclovir) and immunomodulatory (IL-12) payloads, delivered using adeno-associated virus vectors, as a single treatment led to curative outcomes in a mouse model of aggressive glioblastoma. Notably, IL-12 induced an immunological memory that prevented tumour recurrence. The activity and selectivity of the adeno-associated virus and SSE were validated using primary human glioblastoma tissue and normal cortex samples. In summary, SSEs harness the unique core transcriptional programs that define the GSC phenotype and enable precision immune activation. This approach may have broader applications in other contexts when precise control of transgene expression in specific cell states is necessary.
-
Natural maternal immunity protects neonates from Escherichia coli sepsis.
In Nature on 11 March 2026 by Diep, R. E., Adhikari, U., et al.
PubMed
Escherichia coli is a leading cause of neonatal sepsis, with infection occurring in approximately one in every 1,000 live births1,2. However, with E. coli colonization beginning soon after birth3-5 and defects in neonatal host defence maturation6-9, an alternative consideration is why infection does not occur even more frequently. Here we show that newborn babies with E. coli sepsis have selectively reduced vertically transferred natural antibodies that recognize E. coli, mechanistically explaining their susceptibility to infection. Complementary preclinical studies show that preconceptual intestinal colonization with probiotic E. coli Nissle 1917 (EcN)10 primes anti-E. coli immunoglobulin G (IgG) antibodies with broad cross-reactivity to clinical isolates responsible for neonatal sepsis that override the inherent susceptibility of neonatal mice. Outer membrane protein A (OmpA) is a target of maternal IgG and is also essential for EcN colonization-induced serological immunogenicity. Upon vertical transfer to neonates, colonization-primed anti-E. coli IgG uniquely protects against infection via opsonization, requiring both complement and IgG Fc receptors. Compared with specimens from sex and gestational age-matched healthy control babies without infection, dried blood spot specimens collected one day after birth from 100 babies with E. coli sepsis show consistently reduced IgG titres to pooled E. coli clinical isolates and OmpA, along with impaired IgG-dependent antibacterial opsonization. Together, these results demonstrate that natural infection susceptibility of neonates is efficiently rescued by anti-E. coli IgG and identify defects in pathogen-targeted vertically transferred immunity as a primary risk factor for severe invasive infection in newborn babies.
-
Erythropoietin receptor on cDC1s dictates immune tolerance.
In Nature on 1 February 2026 by Zhang, X., McGinnis, C. S., et al.
PubMed
Type 1 conventional dendritic cells (cDC1s) are unique in their efferocytosis1 and cross-presenting abilities2, resulting in antigen-specific T cell immunity3 or tolerance4-8. However, the mechanisms that underlie cDC1 tolerogenic function remain largely unknown. Here we show that the erythropoietin receptor (EPOR) acts as a critical switch that determines the tolerogenic function of cDC1s and the threshold of antigen-specific T cell responses. In total lymphoid irradiation-induced allograft tolerance9,10, cDC1s upregulate EPOR expression, and conditional knockout of EPOR in cDC1s diminishes antigen-specific induction and expansion of FOXP3+ regulatory T (Treg) cells, resulting in allograft rejection. Mechanistically, EPOR promotes efferocytosis-induced tolerogenic maturation7,11 of splenic cDC1s towards late-stage CCR7+ cDC1s characterized by increased expression of the integrin β8 gene12 (Itgb8), and conditional knockout of Itgb8 in cDC1s impairs tolerance induced by total lymphoid irradiation plus anti-thymocyte serum. Migratory cDC1s in peripheral lymph nodes preferentially express EPOR, and their FOXP3+ Treg cell-inducing capacity is enhanced by erythropoietin. Reciprocally, loss of EPOR enables immunogenic maturation of peripheral lymph node migratory and splenic CCR7+ cDC1s by upregulating genes involved in MHC class II- and class I-mediated antigen presentation, cross-presentation and costimulation. EPOR deficiency in cDC1s reduces tumour growth by enhancing anti-tumour T cell immunity, particularly increasing the generation of precursor exhausted tumour antigen-specific CD8+ T cells13 in tumour-draining lymph nodes and supporting their maintenance within tumours, while concurrently reducing intratumoural Treg cells. Targeting EPOR on cDC1s to induce or inhibit T cell immune tolerance could have potential for treating a variety of diseases.