InVivoMAb anti-mouse RGMb
Product Description
Specifications
| Isotype | Rat IgG2a, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2a isotype control, anti-trinitrophenol |
| Recommended Dilution Buffer | InVivoPure pH 6.0T Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
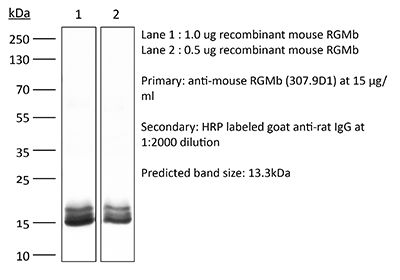
| Immunogen | Recombinant murine RGMb |
| Reported Applications |
in vivo RGMb blockade ELISA Flow cytometry |
| Formulation |
PBS, pH 6.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Xiao Y, Yu S, Zhu B, Bedoret D, Bu X, Francisco LM, Hua P, Duke-Cohan JS, Umetsu DT, Sharpe AH, DeKruyff RH, Freeman GJ (2014). "RGMb is a novel binding partner for PD-L2 and its engagement with PD-L2 promotes respiratory tolerance" J Exp Med 211(5):
PubMed
We report that programmed death ligand 2 (PD-L2), a known ligand of PD-1, also binds to repulsive guidance molecule b (RGMb), which was originally identified in the nervous system as a co-receptor for bone morphogenetic proteins (BMPs). PD-L2 and BMP-2/4 bind to distinct sites on RGMb. Normal resting lung interstitial macrophages and alveolar epithelial cells express high levels of RGMb mRNA, whereas lung dendritic cells express PD-L2. Blockade of the RGMb-PD-L2 interaction markedly impaired the development of respiratory tolerance by interfering with the initial T cell expansion required for respiratory tolerance. Experiments with PD-L2-deficient mice showed that PD-L2 expression on non-T cells was critical for respiratory tolerance, but expression on T cells was not required. Because PD-L2 binds to both PD-1, which inhibits antitumor immunity, and to RGMb, which regulates respiratory immunity, targeting the PD-L2 pathway has therapeutic potential for asthma, cancer, and other immune-mediated disorders. Understanding this pathway may provide insights into how to optimally modulate the PD-1 pathway in cancer immunotherapy while minimizing adverse events.
-
Pérez-Cruz M, Iliopoulou BP, Hsu K, Wu HH, Erkers T, Swaminathan K, Tang SW, Bader CS, Kambham N, Xie B, Dekruyff RH, Freeman GJ, Meyer E (2022). "Immunoregulatory effects of RGMb in gut inflammation" Front Immunol 13:960329.
PubMed
Graft-versus-host disease (GvHD) is a major complication after allogeneic hematopoietic cell transplantation (HCT). Current strategies to prevent GvHD with immunosuppressive drugs carry significant morbidity and may affect the graft-versus-tumor (GVT) effect. Inflammatory bowel disease (IBD) is an intestinal inflammatory condition that affects more than 2 million people in the United States. Current strategies to prevent colitis with immunosuppressive drugs carry significant morbidity. Recently, Repulsive Guidance Molecule b (RGMb) has been identified as part of a signaling hub with neogenin and BMP receptors in mice and humans. In addition, RGMb binds BMP-2/4 in mice and humans as well as PD-L2 in mice. RGMb is expressed in the gut epithelium and by antigen presenting cells, and we found significantly increased expression in mouse small intestine after total body irradiation HCT conditioning. We hypothesized that RGMb may play a role in GvHD and IBD pathogenesis by contributing to mucosal inflammation. Using major-mismatched HCT mouse models, treatment with an anti-RGMb monoclonal antibody (mAb) that blocks the interaction with BMP-2/4 and neogenin prevented GvHD and improved survival compared to isotype control (75% versus 30% survival at 60 days after transplantation). The GVT effect was retained in tumor models. Using an inflammatory bowel disease dextran sulfate sodium model, treatment with anti-RGMb blocking monoclonal antibody but not isotype control prevented colitis and improved survival compared to control (73% versus 33% at 21 days after treatment) restoring gut homeostasis. Anti-RGMb mAb (9D1) treatment decreased IFN-γ and significantly increased IL-5 and IL-10 in the gut of the treated mice compared to the isotype control treated mice.
-
Stroopinsky D, Liegel J, Bhasin M, Cheloni G, Thomas B, Bhasin S, Panchal R, Ghiasuddin H, Rahimian M, Nahas M, Orr S, Capelletti M, Torres D, Tacettin C, Weinstock M, Bisharat L, Morin A, Mahoney KM, Ebert B, Stone R, Kufe D, Freeman GJ, Rosenblatt
PubMed
We have developed a personalized vaccine whereby patient derived leukemia cells are fused to autologous dendritic cells, evoking a polyclonal T cell response against shared and neo-antigens. We postulated that the dendritic cell (DC)/AML fusion vaccine would demonstrate synergy with checkpoint blockade by expanding tumor antigen specific lymphocytes that would provide a critical substrate for checkpoint blockade mediated activation. Using an immunocompetent murine leukemia model, we examined the immunologic response and therapeutic efficacy of vaccination in conjunction with checkpoint blockade with respect to leukemia engraftment, disease burden, survival and the induction of tumor specific immunity. Mice treated with checkpoint blockade alone had rapid leukemia progression and demonstrated only a modest extension of survival. Vaccination with DC/AML fusions resulted in the expansion of tumor specific lymphocytes and disease eradication in a subset of animals, while the combination of vaccination and checkpoint blockade induced a fully protective tumor specific immune response in all treated animals. Vaccination followed by checkpoint blockade resulted in upregulation of genes regulating activation and proliferation in memory and effector T cells. Long term survivors exhibited increased T cell clonal diversity and were resistant to subsequent tumor challenge. The combined DC/AML fusion vaccine and checkpoint blockade treatment offers unique synergy inducing the durable activation of leukemia specific immunity, protection from lethal tumor challenge and the selective expansion of tumor reactive clones.
-
Park JS, Gazzaniga FS, Wu M, Luthens AK, Gillis J, Zheng W, LaFleur MW, Johnson SB, Morad G, Park EM, Zhou Y, Watowich SS, Wargo JA, Freeman GJ, Kasper DL, Sharpe AH (2023). "Targeting PD-L2-RGMb overcomes microbiome-related immunotherapy resistance"
PubMed
The gut microbiota is a crucial regulator of anti-tumour immunity during immune checkpoint inhibitor therapy. Several bacteria that promote an anti-tumour response to immune checkpoint inhibitors have been identified in mice1-6. Moreover, transplantation of faecal specimens from responders can improve the efficacy of anti-PD-1 therapy in patients with melanoma7,8. However, the increased efficacy from faecal transplants is variable and how gut bacteria promote anti-tumour immunity remains unclear. Here we show that the gut microbiome downregulates PD-L2 expression and its binding partner repulsive guidance molecule b (RGMb) to promote anti-tumour immunity and identify bacterial species that mediate this effect. PD-L1 and PD-L2 share PD-1 as a binding partner, but PD-L2 can also bind RGMb. We demonstrate that blockade of PD-L2-RGMb interactions can overcome microbiome-dependent resistance to PD-1 pathway inhibitors. Antibody-mediated blockade of the PD-L2-RGMb pathway or conditional deletion of RGMb in T cells combined with an anti-PD-1 or anti-PD-L1 antibody promotes anti-tumour responses in multiple mouse tumour models that do not respond to anti-PD-1 or anti-PD-L1 alone (germ-free mice, antibiotic-treated mice and even mice colonized with stool samples from a patient who did not respond to treatment). These studies identify downregulation of the PD-L2-RGMb pathway as a specific mechanism by which the gut microbiota can promote responses to PD-1 checkpoint blockade. The results also define a potentially effective immunological strategy for treating patients who do not respond to PD-1 cancer immunotherapy.
Product Citations
-
Interferon-γ-stimulated antigen-presenting cancer-associated fibroblasts hinder neoadjuvant chemoimmunotherapy efficacy in lung cancer.
In Cell Rep Med on 18 March 2025 by Cao, Z., Meng, Z., et al.
PubMed
Conventional neoadjuvant chemotherapy provides limited benefit for patients with resectable non-small cell lung cancer (NSCLC). Recently, neoadjuvant chemoimmunotherapy (NCIT) has transformed the perioperative management of NSCLC by priming systemic anti-tumor immunity before surgery, yet it remains ineffective for at least 50% of patients. Through single-cell sequencing analysis of our NCIT cohort, we identify that antigen-presenting cancer-associated fibroblasts (apCAFs) can impede the efficacy of NCIT. Using a custom cancer-associated fibroblast biobank, we uncover that interferon (IFN)-γ stimulates apCAF expansion via the JAK1/2-STAT1-IFI6/27 pathway. Mechanistically, apCAFs significantly contribute to PD-L2 expression in the tumor microenvironment (TME), triggering the accumulation of FOXP1+regulatory T cells (Tregs) through the PD-L2-RGMB axis. Reprogramming apCAFs by inhibiting the IFN-γ pathway or blocking the PD-L2-RGMB axis substantially mitigates apCAFs-mediated FOXP1+Tregs' expansion. In summary, we reveal the role of apCAFs in compromising NCIT efficacy and propose applications for anti-PD-L2/RGMB regimens to synergize with anti-PD1 therapies by targeting apCAFs.