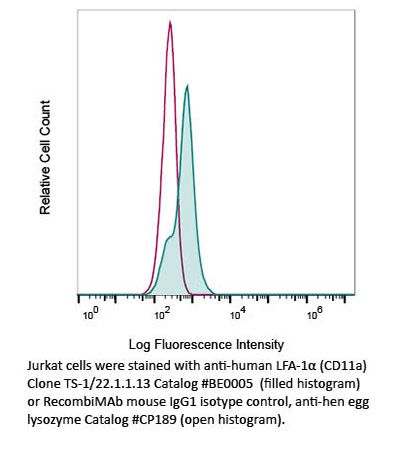
InVivoMAb anti-human LFA-1α (CD11a)
Product Description
Specifications
| Isotype | Mouse IgG1 |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb mouse IgG1 isotype control, unknown specificity |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | Human cytolytic T cells |
| Reported Applications | in vitro LFA-1 neutralization |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107580 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Kitchens, W. H., et al (2012). "Combined costimulatory and leukocyte functional antigen-1 blockade prevents transplant rejection mediated by heterologous immune memory alloresponses" Transplantation 93(10): 997-1005.
PubMed
BACKGROUND: Recent evidence suggests that alloreactive memory T cells are generated by the process of heterologous immunity, whereby memory T cells arising in response to pathogen infection crossreact with donor antigens. Because of their diminished requirements for costimulation during recall, these pathogen-elicited allocrossreactive memory T cells are of particular clinical importance, especially given the emergence of costimulatory blockade as a transplant immunosuppression strategy. METHODS: We used an established model of heterologous immunity involving sequential infection of a naive C57BL/6 recipient with lymphocytic choriomeningitis virus and vaccinia virus, followed by combined skin and bone marrow transplant from a BALB/c donor. RESULTS: We demonstrate that coupling the integrin antagonist anti-leukocyte functional antigen (LFA)-1 with costimulatory blockade could surmount the barrier posed by heterologous immunity in a fully allogeneic murine transplant system. The combined costimulatory and integrin blockade regimen suppressed proliferation of alloreactive memory T cells and attenuated their cytokine effector responses. This combined blockade regimen also promoted the retention of FoxP(3)(+) Tregs in draining lymph nodes. Finally, we show that in an in vitro mixed lymphocyte reaction system using human T cells, the combination of belatacept and anti-LFA-1 was able to suppress cytokine production by alloreactive memory T cells that was resistant to belatacept alone. CONCLUSIONS: As an antagonist against human LFA-1 exists and has been used clinically to treat psoriasis, these findings have significant translational potential for future clinical transplant trials.
Product Citations
-
Exosomal miR-125b-5p deriving from mesenchymal stem cells promotes tubular repair by suppression of p53 in ischemic acute kidney injury.
In Theranostics on 17 April 2021 by Cao, J. Y., Wang, B., et al.
PubMed
Mesenchymal stem cells-derived exosomes (MSC-exos) have attracted great interest as a cell-free therapy for acute kidney injury (AKI). However, the in vivo biodistribution of MSC-exos in ischemic AKI has not been established. The potential of MSC-exos in promoting tubular repair and the underlying mechanisms remain largely unknown. Methods: Transmission electron microscopy, nanoparticle tracking analysis, and western blotting were used to characterize the properties of human umbilical cord mesenchymal stem cells (hucMSCs) derived exosomes. The biodistribution of MSC-exos in murine ischemia/reperfusion (I/R) induced AKI was imaged by the IVIS spectrum imaging system. The therapeutic efficacy of MSC-exos was investigated in renal I/R injury. The cell cycle arrest, proliferation and apoptosis of tubular epithelial cells (TECs) were evaluated in vivo and in HK-2 cells. The exosomal miRNAs of MSC-exos were profiled by high-throughput miRNA sequencing. One of the most enriched miRNA in MSC-exos was knockdown by transfecting miRNA inhibitor to hucMSCs. Then we investigated whether this candidate miRNA was involved in MSC-exos-mediated tubular repair. Results:Ex vivo imaging showed that MSC-exos was efficiently homing to the ischemic kidney and predominantly accumulated in proximal tubules by virtue of the VLA-4 and LFA-1 on MSC-exos surface. MSC-exos alleviated murine ischemic AKI and decreased the renal tubules injury in a dose-dependent manner. Furthermore, MSC-exos significantly attenuated the cell cycle arrest and apoptosis of TECs both in vivo and in vitro. Mechanistically, miR-125b-5p, which was highly enriched in MSC-exos, repressed the protein expression of p53 in TECs, leading to not only the up-regulation of CDK1 and Cyclin B1 to rescue G2/M arrest, but also the modulation of Bcl-2 and Bax to inhibit TEC apoptosis. Finally, inhibiting miR-125b-5p could mitigate the protective effects of MSC-exos in I/R mice. Conclusion: MSC-exos exhibit preferential tropism to injured kidney and localize to proximal tubules in ischemic AKI. We demonstrate that MSC-exos ameliorate ischemic AKI and promote tubular repair by targeting the cell cycle arrest and apoptosis of TECs through miR-125b-5p/p53 pathway. This study provides a novel insight into the role of MSC-exos in renal tubule repair and highlights the potential of MSC-exos as a promising therapeutic strategy for AKI.
-
Eomes-Dependent Loss of the Co-activating Receptor CD226 Restrains CD8+ T Cell Anti-tumor Functions and Limits the Efficacy of Cancer Immunotherapy.
In Immunity on 13 October 2020 by Weulersse, M., Asrir, A., et al.
PubMed
CD8+ T cells within the tumor microenvironment (TME) are exposed to various signals that ultimately determine functional outcomes. Here, we examined the role of the co-activating receptor CD226 (DNAM-1) in CD8+ T cell function. The absence of CD226 expression identified a subset of dysfunctional CD8+ T cells present in peripheral blood of healthy individuals. These cells exhibited reduced LFA-1 activation, altered TCR signaling, and a distinct transcriptomic program upon stimulation. CD226neg CD8+ T cells accumulated in human and mouse tumors of diverse origin through an antigen-specific mechanism involving the transcriptional regulator Eomesodermin (Eomes). Despite similar expression of co-inhibitory receptors, CD8+ tumor-infiltrating lymphocyte failed to respond to anti-PD-1 in the absence of CD226. Immune checkpoint blockade efficacy was hampered in Cd226-/- mice. Anti-CD137 (4-1BB) agonists also stimulated Eomes-dependent CD226 loss that limited the anti-tumor efficacy of this treatment. Thus, CD226 loss restrains CD8+ T cell function and limits the efficacy of cancer immunotherapy.